Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Zr determination with isotope dilution ICP-MS
Zr determination with isotope dilution ICP-MSAsai, Shiho; Hanzawa, Yukiko; Konda, Miki; Suzuki, Daisuke; Magara, Masaaki; Kimura, Takaumi; Ishihara, Ryo*; Saito, Kyoichi*; Yamada, Shinsuke*; Hirota, Hideyuki*
Talanta, 185, p.98 - 105, 2018/08
Times Cited Count:9 Percentile:30.63(Chemistry, Analytical)Estimating the risks associated with radiation from long-lived fission products (LLFP) in radioactive waste is essential to ensure the long-term safety of potential disposal sites. In this study, the amount of  Zr, a LLFP, was determined by ICP-MS after separating Zr from a spent nuclear fuel solution using a microvolume anion-exchange cartridge (TEDA cartridge). The TEDA cartridge achieved highly selective separation of Zr regardless of its small bed volume of 0.08 cm
Zr, a LLFP, was determined by ICP-MS after separating Zr from a spent nuclear fuel solution using a microvolume anion-exchange cartridge (TEDA cartridge). The TEDA cartridge achieved highly selective separation of Zr regardless of its small bed volume of 0.08 cm . The time taken to complete the Zr separation was 1.2 min with a flow rate of 1.5 mL/min, which was 10 times faster than that for a conventional anion-exchange resin column. Almost all the other elements were removed, leading to accurate measurement of
. The time taken to complete the Zr separation was 1.2 min with a flow rate of 1.5 mL/min, which was 10 times faster than that for a conventional anion-exchange resin column. Almost all the other elements were removed, leading to accurate measurement of  Zr. The result connects experimental value to theoretical prediction provided by ORIGEN2, which requires verification. With the measured value, we demonstrated that the theoretical value is reliable enough to estimate radiation risks.
Zr. The result connects experimental value to theoretical prediction provided by ORIGEN2, which requires verification. With the measured value, we demonstrated that the theoretical value is reliable enough to estimate radiation risks.
Aoyagi, Noboru; Palladino, G.*; Nagasaki, Shinya*; Kimura, Takaumi
Bulletin of the Chemical Society of Japan, 91(6), p.882 - 890, 2018/06
Times Cited Count:3 Percentile:10.51(Chemistry, Multidisciplinary)The speciation and coordination geometries of M(III)-citrate complexes in aqueous solutions, where M denotes Eu, Tb, Lu, or Cm, are studied using potentiometric titration, nuclear magnetic resonance spectroscopies. Their photophysical properties are also characterized by time-resolved fluorescence spectra. The formation constants of mononuclear, dinuclear, and trinuclear Lu-citrate species were determined by potentiometric titration in 3.00 M NaClO aqueous media. Terminal carboxylic conformation in trinuclear complexes comprised both the five- and six-membered rings at different exchanging rates. Hydration states evaluated for Eu
aqueous media. Terminal carboxylic conformation in trinuclear complexes comprised both the five- and six-membered rings at different exchanging rates. Hydration states evaluated for Eu ions are the chemical formula of [Eu(Cit)
ions are the chemical formula of [Eu(Cit) (H
(H O)
O)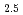 ]
] and [Eu
and [Eu (OH)
(OH) (Cit)
(Cit) (H
(H O)
O)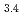 ]
]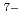 . These complexes in aqueous solution have geometrical similarity to the crystal structures in the literature. Furthermore, the entity of the hetero-trinuclear complex induces the intramolecular energy transfer from Tb
. These complexes in aqueous solution have geometrical similarity to the crystal structures in the literature. Furthermore, the entity of the hetero-trinuclear complex induces the intramolecular energy transfer from Tb to Eu
to Eu . The incorporation of Cm
. The incorporation of Cm into these homo/hetero-trinuclear citrate complexes proved to be a successful trial to probe the formation of actinide polymer at a trace level.
into these homo/hetero-trinuclear citrate complexes proved to be a successful trial to probe the formation of actinide polymer at a trace level.
 on Na-montmorillonite studied by time-resolved laser fluorescence spectroscopy and surface complexation modeling
on Na-montmorillonite studied by time-resolved laser fluorescence spectroscopy and surface complexation modelingSasaki, Takayuki*; Ueda, Kenyo*; Saito, Takumi; Aoyagi, Noboru; Kobayashi, Taishi*; Takagi, Ikuji*; Kimura, Takaumi; Tachi, Yukio
Journal of Nuclear Science and Technology, 53(4), p.592 - 601, 2016/04
Times Cited Count:15 Percentile:77.12(Nuclear Science & Technology)The influences of pH and the concentrations of Eu and NaNO
and NaNO on the sorption of Eu
on the sorption of Eu to Na-montmorillonite were investigated through batch sorption measurements and time-resolved laser fluorescence spectroscopy (TRLFS). The pH had a little effect on the distribution coefficients (Kd) in 0.01 M NaNO
to Na-montmorillonite were investigated through batch sorption measurements and time-resolved laser fluorescence spectroscopy (TRLFS). The pH had a little effect on the distribution coefficients (Kd) in 0.01 M NaNO , whereas the Kd strongly depended on pH at 1 M NaNO
, whereas the Kd strongly depended on pH at 1 M NaNO . A cation exchange model combined with a one-site non-electrostatic surface complexation model was successfully applied to the measured Kd. The TRLFS spectra of Eu
. A cation exchange model combined with a one-site non-electrostatic surface complexation model was successfully applied to the measured Kd. The TRLFS spectra of Eu sorbed were processed by parallel factor analysis (PARAFAC), which corresponded to one outer-sphere (factor A) and two inner-sphere (factor B and C) complexes. It turned out that factors A and B correspond to Eu
sorbed were processed by parallel factor analysis (PARAFAC), which corresponded to one outer-sphere (factor A) and two inner-sphere (factor B and C) complexes. It turned out that factors A and B correspond to Eu sorbed by ion exchange sites and inner-sphere complexation with hydroxyl groups of the edge faces, respectively. Factor C became dominant at relatively high pH and ionic strength and likely correspond to the precipitation of Eu(OH)
sorbed by ion exchange sites and inner-sphere complexation with hydroxyl groups of the edge faces, respectively. Factor C became dominant at relatively high pH and ionic strength and likely correspond to the precipitation of Eu(OH) on the surface.
on the surface.
 Np content in spent nuclear fuel
Np content in spent nuclear fuelAsai, Shiho; Hanzawa, Yukiko; Konda, Miki; Suzuki, Daisuke; Magara, Masaaki; Kimura, Takaumi; Ishihara, Ryo*; Saito, Kyoichi*; Yamada, Shinsuke*; Hirota, Hideyuki*
Analytical Chemistry, 88(6), p.3149 - 3155, 2016/03
Times Cited Count:8 Percentile:26.70(Chemistry, Analytical)Neptunium-237 ( Np) is one of the major long-lived radionuclides found in spent nuclear fuel. To evaluate the long-term safety of a HLW repository, the
Np) is one of the major long-lived radionuclides found in spent nuclear fuel. To evaluate the long-term safety of a HLW repository, the  Np content in spent nuclear fuel must be determined. In this study, micro-volume anion-exchange porous polymer disk-packed cartridges were prepared for Am-Np separation, which is required prior to the measurement of
Np content in spent nuclear fuel must be determined. In this study, micro-volume anion-exchange porous polymer disk-packed cartridges were prepared for Am-Np separation, which is required prior to the measurement of  Np with ICP-MS. Disks with a volume of 0.08 cm
Np with ICP-MS. Disks with a volume of 0.08 cm were cut out from porous sheets having triethylenediamine (TEDA)-containing polymer chains densely attached on the pore surface. The resulting TEDA-introduced disk cartridge was applied to a spent nuclear fuel sample. The chemical yield of Np was 90.4%, which is sufficiently high for ICP-MS measurement of
were cut out from porous sheets having triethylenediamine (TEDA)-containing polymer chains densely attached on the pore surface. The resulting TEDA-introduced disk cartridge was applied to a spent nuclear fuel sample. The chemical yield of Np was 90.4%, which is sufficiently high for ICP-MS measurement of  Np. Compared with the conventional separation technique using commercially available anion-exchange resin columns, the time required to adsorb, wash and elute Np using the TEDA-introduced disk cartridge was reduced by 75%.
Np. Compared with the conventional separation technique using commercially available anion-exchange resin columns, the time required to adsorb, wash and elute Np using the TEDA-introduced disk cartridge was reduced by 75%.
Kimura, Takaumi
Hosha Kagaku No Jiten, p.245 - 246, 2015/09
no abstracts in English
 -bis(thiocyanato)aurate(I) complexes in ionic liquids
-bis(thiocyanato)aurate(I) complexes in ionic liquidsAoyagi, Noboru; Shinha, Yusuke*; Ikeda, Atsushi*; Haga, Yoshinori; Shimojo, Kojiro; Brooks, N. R.*; Izuoka, Akira*; Naganawa, Hirochika; Kimura, Takaumi; Binnemans, K.*
Crystal Growth & Design, 15(3), p.1422 - 1429, 2015/03
Times Cited Count:12 Percentile:64.25(Chemistry, Multidisciplinary)The photochemistry of a gold(I) thiocyanate complex has been investigated to determine the coordination structure in both the solid and liquid states. The coordination geometries of the supramolecular complex and the concomitant exciplex have mainly been analyzed by crystallographic analysis and X-ray absorption spectroscopy. The Au-S bond distance and Au-Au separation of the compound in the ground state and in the phosphorescent excited state were compared. Upon irradiation with UV light, the excimeric interactions were enhanced, resulting in a contraction of the Au-Au aurophilic distance. A broad luminescence spectrum was observed for the one-dimensional chain suprastructure. The time-resolved luminescence spectra indicated the entity of several oligomeric species in the crude liquid without neutral solvent molecules. In addition, EXAFS spectroscopy exhibited a slight change in the nearest Au-S distance due to the photo-switched transformation. The deformation of the (Au-Au)* exciplexes was not apparently promoted in the liquid state with the asymmetrical imidazoium cations having a non-local charge distribution in the present observation. This is plausibly due to a flexible molecular structure and a property in the liquid state. In conclusion, the photo-excited nature of the pseudo-one dimensional complexes is emerged in controlling bond distances among the supramolecular networks.


Saito, Takumi; Aoyagi, Noboru; Kimura, Takaumi
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1129 - 1132, 2015/02
Times Cited Count:16 Percentile:75.78(Chemistry, Analytical)Time-resolved laser-induced fluorescence spectroscopy (TRLFS) is a powerful speciation technique for fluorescent metal ions and can be further improved by combining with multi-mode factor analysis such as parallel factor analysis (PARAFAC). This study demonstrates the applicability of TRLFS combined with PARAFAC for the speciation of uranyl (UO
 ) in the presence of silicic acid (Si(OH)
) in the presence of silicic acid (Si(OH) ). A series of TRLFS data with varied Si(OH)
). A series of TRLFS data with varied Si(OH) concentration was processed by PARAFAC, resulting in three factors corresponding to free UO
concentration was processed by PARAFAC, resulting in three factors corresponding to free UO
 , UO
, UO SiO(OH)
SiO(OH)
 , and UO
, and UO OH
OH . The stability constant of UO
. The stability constant of UO SiO(OH)
SiO(OH)
 was further optimized, based on the intensity profiles of the factors.
was further optimized, based on the intensity profiles of the factors.
Aoyagi, Noboru; Watanabe, Masayuki; Kirishima, Akira*; Sato, Nobuaki*; Kimura, Takaumi
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1095 - 1098, 2015/02
Times Cited Count:6 Percentile:42.34(Chemistry, Analytical)Kimura, Takaumi
Genshiryoku, Ryoshi, Kakuyugo Jiten, 3, p.52 - 55, 2014/12
no abstracts in English
Kitatsuji, Yoshihiro; Otobe, Haruyoshi; Kimura, Takaumi; Kihara, Sorin*
Electrochimica Acta, 141, p.6 - 12, 2014/09
Times Cited Count:5 Percentile:9.00(Electrochemistry)Reduction processes of U(VI) in weakly acidic solutions were investigated based on electrochemical and spectrophotometric measurements. A reversible one-electron reduction of U(VI) to U(V) and a further irreversible reduction of U(V) were observed voltammetrically at a gold microdisk electrode in solutions of pH from 2.0 to 3.5. Aggregates of U(IV) were formed as a deposit on the electrode and a colloid in the bulk solution, when the electrolysis was carried out at a gold gauze electrode, even though the potential applied was that available for the first one-electron reduction wave of U(VI) observed. It was elucidated that the aggregate was produced by the combination of the one-electron reduction to U(V) and the disproportionation of U(V) producing U(IV) and U(VI). The aggregate enhanced the rate of the disproportionation of U(V), and hence the reduction current of U(VI) increased abruptly when a definite amount of aggregate was formed on the electrode, in the solution, or both.
Sasaki, Yuji; Kitatsuji, Yoshihiro; Sugo, Yumi; Tsubata, Yasuhiro; Suzuki, Tomoya; Kimura, Takaumi; Morita, Yasuji
Proceedings of 20th International Solvent Extraction Conference (ISEC 2014), p.431 - 435, 2014/09
The eight amide extractants including podand-types, which introduce oxygen, nitrogen and sulfur donor atoms in their central frame, are synthesized and their D values for actinide extraction are compared. The central frame affect greatly on their extractability and it is clear that diglycolamide (DGA) has the highest extractability among extractants used here. In addition, DGA having the different length of alkyl chain or phenyl groups attached to amidic N atoms are obtained and the effect of these substituents is studied. Due to the steric hindrance or hydrogen bond, DGA with long and branched alkyl chains, phenyl group, and proton give relatively low D values.
Kimura, Takaumi
Hosha Kagaku, (29), p.26 - 33, 2014/04
no abstracts in English
 -ray irradiation
-ray irradiationArisaka, Makoto; Watanabe, Masayuki; Sugo, Yumi; Kobayashi, Kumiko*; Kanao, Osamu*; Kimura, Takaumi
Journal of Nuclear Science and Technology, 51(4), p.457 - 464, 2014/04
Times Cited Count:3 Percentile:21.88(Nuclear Science & Technology)Toward the development of a practical separation method for trivalent actinides and lanthanides, we used extraction chromatography with alkyl-pyridinedicarboxyamides as the extractant. The results confirmed that the performance degradation of the adsorbent caused by contact with HNO and/or irradiation with
and/or irradiation with  rays would be very small during the operation of column chromatography. The optimal conditions for the column separation were also determined: eluent, 5M HNO
rays would be very small during the operation of column chromatography. The optimal conditions for the column separation were also determined: eluent, 5M HNO ; flow rate, 0.1 mL/min.
; flow rate, 0.1 mL/min.
Okubo, Ayako; Obata, Hajime*; Magara, Masaaki; Kimura, Takaumi; Ogawa, Hiroshi*
Analytica Chimica Acta, 804, p.120 - 125, 2013/12
Times Cited Count:2 Percentile:6.97(Chemistry, Analytical)This work introduces a novel method of recovery of iron hydroxide using a DIAION CR-20 chelating resin column to determine Th isotopes in seawater with a sector field (SF) inductively coupled plasma mass spectrometer (ICP-MS). Thorium isotopes in seawater were coprecipitated with iron hydroxide, and this precipitate was sent to chelating resin column. The chelating column quantitatively collected 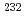 Th with iron hydroxide in seawater at flow rates of 20-25 mL/min. Based on this flow rate, a 5 L sample was processed within 3-4 h.
Th with iron hydroxide in seawater at flow rates of 20-25 mL/min. Based on this flow rate, a 5 L sample was processed within 3-4 h.
Arisaka, Makoto; Watanabe, Masayuki; Kimura, Takaumi
Radiochimica Acta, 101(11), p.711 - 717, 2013/11
Times Cited Count:3 Percentile:24.16(Chemistry, Inorganic & Nuclear)Four new alkyl-pyridinedicarboxyamides (R-PDA; R = butyl, octyl, decyl, dodecyl) were synthesized and used as extractants for partitioning minor actinides from high level radioactive waste using extraction chromatography. The R-PDAs were successfully impregnated into XAD resins (Amberlite). The prepared adsorbents, R-PDA/XAD, exhibited moderate adsorptions of both Am(III) and Eu(III), and separation between Am(III) and Eu(III), over a high HNO concentration range (3-5 M). Deterioration of the prepared adsorbent as a result of contact with HNO
concentration range (3-5 M). Deterioration of the prepared adsorbent as a result of contact with HNO solution was prevented by using R-PDAs containing longer alkyl groups. The equilibrium data for Eu(III) were analyzed using Langmuir and Freundlich isotherm models. The adsorption of Eu(III) ions onto the R-PDA/XAD was fitted better to the Langmuir isotherm than to the Freundlich isotherm. The maximum adsorption capacity was determined to be 24.2 mg Eu/g (R-PDA/XAD).
solution was prevented by using R-PDAs containing longer alkyl groups. The equilibrium data for Eu(III) were analyzed using Langmuir and Freundlich isotherm models. The adsorption of Eu(III) ions onto the R-PDA/XAD was fitted better to the Langmuir isotherm than to the Freundlich isotherm. The maximum adsorption capacity was determined to be 24.2 mg Eu/g (R-PDA/XAD).
Aoyagi, Noboru; Saito, Takumi*; Kimura, Takaumi
Bunseki, 2013(9), p.536 - 542, 2013/09
no abstracts in English
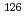 Sn content in spent nuclear fuel sample
Sn content in spent nuclear fuel sampleAsai, Shiho; Toshimitsu, Masaaki; Hanzawa, Yukiko; Suzuki, Hideya; Shinohara, Nobuo; Inagawa, Jun; Okumura, Keisuke; Hotoku, Shinobu; Kimura, Takaumi; Suzuki, Kensuke*; et al.
Journal of Nuclear Science and Technology, 50(6), p.556 - 562, 2013/06
Times Cited Count:11 Percentile:61.40(Nuclear Science & Technology)The 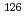 Sn content in a spent nuclear fuel solution was determined by ICP-MS for its inventory estimation in high-level radioactive waste. An irradiated UO
Sn content in a spent nuclear fuel solution was determined by ICP-MS for its inventory estimation in high-level radioactive waste. An irradiated UO fuel was used as a sample to evaluate the reliability of the methodology. Prior to the measurement, Sn was separated from
fuel was used as a sample to evaluate the reliability of the methodology. Prior to the measurement, Sn was separated from 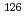 Te, which causes major isobaric interference in the determination of
Te, which causes major isobaric interference in the determination of 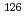 Sn content, along with highly radioactive coexisting elements using an anion-exchange column. The absence of counts attributed to Te in the Sn-containing effluent indicates that Te was completely removed. After washing, Sn retained on the column was readily eluted with 1 M HNO
Sn content, along with highly radioactive coexisting elements using an anion-exchange column. The absence of counts attributed to Te in the Sn-containing effluent indicates that Te was completely removed. After washing, Sn retained on the column was readily eluted with 1 M HNO . The isotope ratios of Sn were successfully determined and showed good agreement with those obtained through ORIGEN2 calculations. The results reported in this paper are the first experimental values of
. The isotope ratios of Sn were successfully determined and showed good agreement with those obtained through ORIGEN2 calculations. The results reported in this paper are the first experimental values of 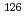 Sn content in the spent nuclear fuel solution originating in spent nuclear fuel irradiated at a nuclear power plant in Japan.
Sn content in the spent nuclear fuel solution originating in spent nuclear fuel irradiated at a nuclear power plant in Japan.
Esaka, Fumitaka; Magara, Masaaki; Kimura, Takaumi
Journal of Analytical Atomic Spectrometry, 28(5), p.682 - 688, 2013/05
Times Cited Count:10 Percentile:48.11(Chemistry, Analytical)A fission track technique and automated particle measurement (APM) were used as particle screening methods to select uranium containing particles, prior to the ICP-MS analysis. In the ICP-MS combined with the fission track technique, the uranium isotope abundance ratios were successfully determined for individual particles with diameters ranging from 1.2 to 2.4  m in a certified reference material. The analysis of the real inspection swipe sample gave the
m in a certified reference material. The analysis of the real inspection swipe sample gave the  U/
U/ U isotope abundance ratios between 0.0246 and 0.0376. When the APM was used as the particle screening method for the real inspection swipe sample, the
U isotope abundance ratios between 0.0246 and 0.0376. When the APM was used as the particle screening method for the real inspection swipe sample, the  U/
U/ U isotope abundance ratios ranging from 0.0259 to 0.0432 were obtained. From these result, it is indicated that the performance of APM to identify uranium containing particles with higher
U isotope abundance ratios ranging from 0.0259 to 0.0432 were obtained. From these result, it is indicated that the performance of APM to identify uranium containing particles with higher  U abundance is greater than that of the fission track technique.
U abundance is greater than that of the fission track technique.
Esaka, Fumitaka; Magara, Masaaki; Suzuki, Daisuke; Miyamoto, Yutaka; Kimura, Takaumi
Hyomen Kagaku, 34(3), p.125 - 130, 2013/03
Analysis of individual particles containing nuclear materials in environmental samples gives important information on the origins. This paper describes analytical techniques of individual particles containing nuclear materials such as uranium and plutonium. A scanning electron microscope (SEM) combined with an X-ray detector, solid track detectors, and secondary ion mass spectrometry (SIMS) were used to identify particles containing uranium and/or plutonium in environmental samples. Isotope ratios for these particles were successfully determined with SIMS, thermal ionization mass spectrometry (TIMS) or inductively coupled plasma mass spectrometry (ICPMS). The complementary use of these methods is effective to characterize individual particles rapidly, precisely and accurately.
 radiolysis of the mixture of mordenite and seawater
radiolysis of the mixture of mordenite and seawaterKumagai, Yuta; Kimura, Atsushi; Taguchi, Mitsumasa; Nagaishi, Ryuji; Yamagishi, Isao; Kimura, Takaumi
Journal of Nuclear Science and Technology, 50(2), p.130 - 138, 2013/02
Times Cited Count:35 Percentile:91.41(Nuclear Science & Technology)