Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Shiga, Hideaki*; Taki, Junichi*; Washiyama, Koshin*; Yamamoto, Jumpei*; Kinase, Sakae; Okuda, Koichi*; Kinuya, Seigo*; Watanabe, Naoto*; Tonami, Hisao*; Koshida, Kichiro*; et al.
PLOS ONE (Internet), 8(2), p.e57671_1 - e57671_8, 2013/02
Times Cited Count:18 Percentile:68.67(Multidisciplinary Sciences)Kinase, Sakae; Washiyama, Koshin*; Shiga, Hideaki*; Taki, Junichi*; Nakanishi, Yusuke*; Koshida, Kichiro*; Miwa, Takaki*; Kinuya, Seigo*; Amano, Ryohei*
KEK Proceedings 2012-7, p.35 - 40, 2012/10
no abstracts in English
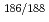 Re-labeled antibody (A7) for radioimmunotherapy with rhenium(I) tricarbonyl core as a chelate site
Re-labeled antibody (A7) for radioimmunotherapy with rhenium(I) tricarbonyl core as a chelate siteOgawa, Kazuma*; Kawashima, Hidekazu*; Kinuya, Seigo*; Shiba, Kazuhiro*; Onoguchi, Masahisa*; Kimura, Hiroyuki*; Hashimoto, Kazuyuki; Odani, Akira*; Saji, Hideo*
Annals of Nuclear Medicine, 23(10), p.843 - 848, 2009/12
Times Cited Count:9 Percentile:31.42(Radiology, Nuclear Medicine & Medical Imaging)Rhenium is one of the most valuable elements for internal radiotherapy because 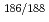 Re have favorable physical characteristics. However, there are problems when proteins such as antibodies are used as carriers of
Re have favorable physical characteristics. However, there are problems when proteins such as antibodies are used as carriers of 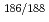 Re. Labeling methods require the complicated processes. Therefore, we planned the preparation by a simple method and evaluation of a stable
Re. Labeling methods require the complicated processes. Therefore, we planned the preparation by a simple method and evaluation of a stable 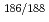 Re-labeled antibody. For this purpose, we selected
Re-labeled antibody. For this purpose, we selected 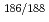 Re(I) tricarbonyl complex as a chelating site. A7 was used as a model protein.
Re(I) tricarbonyl complex as a chelating site. A7 was used as a model protein. 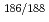 Re-labeled A7 was prepared by directly reacting a
Re-labeled A7 was prepared by directly reacting a 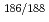 Re(I) tricarbonyl precursor with A7.
Re(I) tricarbonyl precursor with A7. 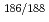 Re-(CO)
Re-(CO) -A7 were prepared with radiochemical yields of 23-28%. After purification,
-A7 were prepared with radiochemical yields of 23-28%. After purification, 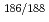 Re-(CO)
Re-(CO) -A7 showed a radiochemical purity of over 95%. In biodistribution experiments,
-A7 showed a radiochemical purity of over 95%. In biodistribution experiments, 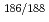 Re-labeled A7 showed high uptakes in the tumor.
Re-labeled A7 showed high uptakes in the tumor.
 Re-complex-conjugated bisphosphonate for the palliation of metastatic bone pain in an animal model
Re-complex-conjugated bisphosphonate for the palliation of metastatic bone pain in an animal modelOgawa, Kazuma*; Mukai, Takahiro*; Asano, Daigo*; Kawashima, Hidekazu*; Kinuya, Seigo*; Shiba, Kazuhiro*; Hashimoto, Kazuyuki; Mori, Hirofumi*; Saji, Hideo*
Journal of Nuclear Medicine, 48(1), p.122 - 127, 2007/01
We developed a highly stable rhenium-186 ( Re)-MAG3 complex-conjugated bisphosphonate, (
Re)-MAG3 complex-conjugated bisphosphonate, ( Re-MAG3-HBP), for the treatment of painful bone metastases. This agent showed a superior biodistribution as a bone-seeking agent in normal mice when compared with
Re-MAG3-HBP), for the treatment of painful bone metastases. This agent showed a superior biodistribution as a bone-seeking agent in normal mice when compared with  Re-HEDP. In this study, we evaluated the therapeutic effects of
Re-HEDP. In this study, we evaluated the therapeutic effects of  Re-MAG3-HBP using an animal model of bone metastasis. In the rats treated with
Re-MAG3-HBP using an animal model of bone metastasis. In the rats treated with  Re-HEDP, tumor growth was comparable to that in untreated rats. In contrast, when
Re-HEDP, tumor growth was comparable to that in untreated rats. In contrast, when  Re-MAG3-HBP was administered, tumor growth was significantly inhibited. Allodynia induced by bone metastasis was attenuated by treatment with
Re-MAG3-HBP was administered, tumor growth was significantly inhibited. Allodynia induced by bone metastasis was attenuated by treatment with  Re-MAG3-HBP or
Re-MAG3-HBP or  Re-HEDP, but
Re-HEDP, but  Re-MAG3-HBP tended to be more effective. These results indicate that
Re-MAG3-HBP tended to be more effective. These results indicate that  Re-MAG3-HBP could be useful as a therapeutic agent for the palliation of metastatic bone pain.
Re-MAG3-HBP could be useful as a therapeutic agent for the palliation of metastatic bone pain.
 Re-MAG3-A7 anti-colorectal cancer monoclonal antibody; Comparison with
Re-MAG3-A7 anti-colorectal cancer monoclonal antibody; Comparison with  I-counterpart
I-counterpartKinuya, Seigo*; Yokoyama, Kunihiko*; Kobayashi, Katsutoshi; Motoishi, Shoji; Onoma, Katsuyuki; Watanabe, Naoto*; Shuke, Noriyuki*; Bunko, Hisashi*; Nichigishi, Takatoshi*; Tonami, Norihisa*
Annals of Nuclear Medicine, 15(3), p.199 - 202, 2001/06
Times Cited Count:9 Percentile:31.12(Radiology, Nuclear Medicine & Medical Imaging)no abstracts in English
 Re-labeled monoclonal antibody
Re-labeled monoclonal antibodyKinuya, Seigo*; Yokoyama, Kunihiko*; Kudo, Miho*; Kasahara, Yoshihito*; Kobayashi, Katsutoshi; Motoishi, Shoji; Onoma, Katsuyuki; Bunko, Hisashi*; Nichigishi, Takatoshi*; Tonami, Norihisa*
Journal of Nuclear Medicine, 42(4), p.596 - 600, 2001/04
no abstracts in English
 Re-labeled antibody (A7) by a simple method using rhenium(I) tricarbonyl complex
Re-labeled antibody (A7) by a simple method using rhenium(I) tricarbonyl complexOgawa, Kazuma*; Kawashima, Hidekazu*; Kinuya, Seigo*; Yoshimoto, Mitsuyoshi*; Shiba, Kazuhiro*; Kimura, Hiroyuki*; Hashimoto, Kazuyuki; Mori, Hirofumi*; Saji, Hideo*
no journal, ,
 Re is one of the most useful radionuclides for internal radiotherapy. However, there is a problem when protein such as antibody is used as a carrier of
Re is one of the most useful radionuclides for internal radiotherapy. However, there is a problem when protein such as antibody is used as a carrier of  Re. The labeling method using bifunctional chelating agents require the conjugation of
Re. The labeling method using bifunctional chelating agents require the conjugation of  Re-complex to protein after radiolabeling with the bifunctional chelating agent. Then, we planned the preparation of a stable
Re-complex to protein after radiolabeling with the bifunctional chelating agent. Then, we planned the preparation of a stable  Re-labeled protein by a simple method. A7 monoclonal antibody was labeled by reacting
Re-labeled protein by a simple method. A7 monoclonal antibody was labeled by reacting  Re(I) tricarbonyl precursor with A7 directly.
Re(I) tricarbonyl precursor with A7 directly.  Re labeled A7 was prepared with radiochemical yield of 23%. After purification,
Re labeled A7 was prepared with radiochemical yield of 23%. After purification,  Re labeled A7 showed radiochemical purity over 98%. After 24 hours of incubation, about 93% of
Re labeled A7 showed radiochemical purity over 98%. After 24 hours of incubation, about 93% of  Re-A7 remained intact, which indicates
Re-A7 remained intact, which indicates  Re-A7 is stable in vitro. In biodistribution experiment, 11.2% of the injected dose/g of
Re-A7 is stable in vitro. In biodistribution experiment, 11.2% of the injected dose/g of  Re-A7 accumulated in the tumor at 24 hours postinjection, and tumor to blood ratio was over 1.0 at the same time.
Re-A7 accumulated in the tumor at 24 hours postinjection, and tumor to blood ratio was over 1.0 at the same time.
Kinase, Sakae; Washiyama, Koshin*; Shiga, Hideaki*; Taki, Junichi*; Nakanishi, Yusuke*; Koshida, Kichiro*; Miwa, Takaki*; Kinuya, Seigo*; Amano, Ryohei*
no journal, ,
no abstracts in English