Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Nakajima, Kenji; Kawakita, Yukinobu; Ito, Shinichi*; Abe, Jun*; Aizawa, Kazuya; Aoki, Hiroyuki; Endo, Hitoshi*; Fujita, Masaki*; Funakoshi, Kenichi*; Gong, W.*; et al.
Quantum Beam Science (Internet), 1(3), p.9_1 - 9_59, 2017/12
The neutron instruments suite, installed at the spallation neutron source of the Materials and Life Science Experimental Facility (MLF) at the Japan Proton Accelerator Research Complex (J-PARC), is reviewed. MLF has 23 neutron beam ports and 21 instruments are in operation for user programs or are under commissioning. A unique and challenging instrumental suite in MLF has been realized via combination of a high-performance neutron source, optimized for neutron scattering, and unique instruments using cutting-edge technologies. All instruments are/will serve in world-leading investigations in a broad range of fields, from fundamental physics to industrial applications. In this review, overviews, characteristic features, and typical applications of the individual instruments are mentioned.
 resolution
resolutionUemura, Takuya*; Kita, Akiko*; Watanabe, Yoshihiko*; Adachi, Motoyasu; Kuroki, Ryota; Morimoto, Yukio*
Biochemical and Biophysical Research Communications, 469(2), p.158 - 163, 2016/01
Times Cited Count:16 Percentile:50.44(Biochemistry & Molecular Biology)Futakawa, Masatoshi; Kogawa, Hiroyuki; Hasegawa, Shoichi; Ikeda, Yujiro; Riemer, B.*; Wendel, M.*; Haines, J.*; Bauer, G.*; Naoe, Takashi; Okita, Kohei*; et al.
Journal of Nuclear Materials, 377(1), p.182 - 188, 2008/06
Times Cited Count:28 Percentile:85.1(Materials Science, Multidisciplinary)no abstracts in English
Hasegawa, Ken*; Koide, Kaoru*; Muneto, Masaru*; Nagasaki, Yasushi*; Nakano, Katsushi; Yamagishi, Akiko*; Okita, Masatoshi*
JNC TN7400 2005-020, 199 Pages, 1994/04
None
Okita, Masatoshi*; Muneto, Masaru*; Hasegawa, Ken*; Nagasaki, Yasushi*; Koide, Kaoru*; Yamagishi, Akiko*; Semba, Tsuyoshi
JNC TN7400 2005-018, 241 Pages, 1992/04
None
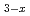 K
K H(SeO
H(SeO )
) by single crystal neutron diffraction
by single crystal neutron diffractionKiyanagi, Ryoji; Matsuo, Yasumitsu*; Ohara, Takashi; Kawasaki, Takuro; Oikawa, Kenichi; Kaneko, Koji; Tamura, Itaru; Hanashima, Takayasu*; Munakata, Koji*; Nakao, Akiko*; et al.
no journal, ,
The materials represented as M H(XO
H(XO )
) is known to exhibit a super protonic conductivity at rather low temperature. The protonic conductivity emerges upon a structural phase transition, and the phase transition temperature varies depending on the elements at M and X. In addition, the protonic conductivity also varies depending on the elements, but the origin has not been uncovered. In order to clarify the relation between the structure and the protonic conductivity including the phase transition mechanism, Solid solutions of super protonic conductors, Rb
is known to exhibit a super protonic conductivity at rather low temperature. The protonic conductivity emerges upon a structural phase transition, and the phase transition temperature varies depending on the elements at M and X. In addition, the protonic conductivity also varies depending on the elements, but the origin has not been uncovered. In order to clarify the relation between the structure and the protonic conductivity including the phase transition mechanism, Solid solutions of super protonic conductors, Rb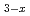 K
K H(SeO
H(SeO )
) (x=0, 1, 2, 3), were structurally investigated by single crystal neutron diffraction. The structure analyses showed evident linear relation between the lattice parameters and x. Meanwhile, K was found to preferably reside at one of the two M sites. A apparent relation between the occupancy of K at one site and the transition temperature and the hydrogen-bond length were demonstrated.
(x=0, 1, 2, 3), were structurally investigated by single crystal neutron diffraction. The structure analyses showed evident linear relation between the lattice parameters and x. Meanwhile, K was found to preferably reside at one of the two M sites. A apparent relation between the occupancy of K at one site and the transition temperature and the hydrogen-bond length were demonstrated.
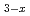 K
K H(SeO
H(SeO )
)
Kiyanagi, Ryoji; Matsuo, Yasumitsu*; Ohara, Takashi; Kawasaki, Takuro; Oikawa, Kenichi; Kaneko, Koji; Tamura, Itaru; Nakao, Akiko*; Hanashima, Takayasu*; Munakata, Koji*; et al.
no journal, ,
The materials represented as M H(XO
H(XO )
) are known to exhibit super protonic conductivity at rather low temperature, but the mechanism of the super protonic phase transition and the protonic conduction have not been clarified. In this study, a mixed material, Rb
are known to exhibit super protonic conductivity at rather low temperature, but the mechanism of the super protonic phase transition and the protonic conduction have not been clarified. In this study, a mixed material, Rb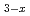 K
K H(SeO
H(SeO )
) , was investigated by means of conductivity measurements and neutron diffraction. The neutron diffraction experiments were conducted on BL18 at J-PARC/MLF. The conductivity measurements revealed that the phase transition temperature significantly changes below x=2. Meanwhile, the neutron structure analyses revealed that K atoms exclusively occupy one of two non-equivalent sites for M atoms below x=2. These observations are indicative of the close relationship between the phase transition and the internal atomic arrangement.
, was investigated by means of conductivity measurements and neutron diffraction. The neutron diffraction experiments were conducted on BL18 at J-PARC/MLF. The conductivity measurements revealed that the phase transition temperature significantly changes below x=2. Meanwhile, the neutron structure analyses revealed that K atoms exclusively occupy one of two non-equivalent sites for M atoms below x=2. These observations are indicative of the close relationship between the phase transition and the internal atomic arrangement.
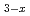 K
K H(SeO
H(SeO )
)
Kiyanagi, Ryoji; Matsuo, Yasumitsu*; Ohara, Takashi; Kawasaki, Takuro; Oikawa, Kenichi; Kaneko, Koji; Tamura, Itaru; Hanashima, Takayasu*; Munakata, Koji*; Nakao, Akiko*; et al.
no journal, ,
M H(SeO
H(SeO )
) forms a material group that exhibits superprotonic conductivity at rather low temperature. The superprotonic conductivity manifests itself upon a structural phase transition. The phase transition temperature varies depending on the elements of M and X. In order to clarify the relation between the crystal structure and the phase transition, Rb
forms a material group that exhibits superprotonic conductivity at rather low temperature. The superprotonic conductivity manifests itself upon a structural phase transition. The phase transition temperature varies depending on the elements of M and X. In order to clarify the relation between the crystal structure and the phase transition, Rb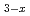 K
K H(SeO
H(SeO )
) was investigated by means of a conductivity measurement and single crystal neutron structure analyses. The phase transition temperature was found to be non-linear to the variation of x by the conductivity measurement. The structure analyses revealed that the site occupancy of K at one of two M sites non-linearly varied with respect to x. This suggest that a close relation between the M site and the superprotonic phase transition.
was investigated by means of a conductivity measurement and single crystal neutron structure analyses. The phase transition temperature was found to be non-linear to the variation of x by the conductivity measurement. The structure analyses revealed that the site occupancy of K at one of two M sites non-linearly varied with respect to x. This suggest that a close relation between the M site and the superprotonic phase transition.
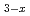 K
K H(SeO
H(SeO )
)
Kiyanagi, Ryoji; Matsuo, Yasumitsu*; Ohara, Takashi; Kawasaki, Takuro; Oikawa, Kenichi; Kaneko, Koji; Tamura, Itaru; Hanashima, Takayasu*; Munakata, Koji*; Nakao, Akiko*; et al.
no journal, ,
The materials, M H(XO
H(XO )
) (M=alkaline metal, X=Se, S), exhibit high protonic conductivities in a relatively low temperature region. The high protonic conductivities emerge upon a structural phase transition and the phase transition temperature (Tc) varies depending on the elements of M and X. In this study, mixed materials, Rb
(M=alkaline metal, X=Se, S), exhibit high protonic conductivities in a relatively low temperature region. The high protonic conductivities emerge upon a structural phase transition and the phase transition temperature (Tc) varies depending on the elements of M and X. In this study, mixed materials, Rb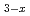 K
K H(SeO
H(SeO )
) , were studied by means of conductivity measurements and single crystal neutron structure analyses in order to clarify how the elements at the M site affect the Tc from the structural aspect. The conductivity measurements revealed that the Tc decreased as the K content increased. The variation of the Tc was found to be non-linear with respect to the K content. The structure analyses showed that the K ions preferred to occupy one of two M sites and the occupancy of the K ion at this site non-linearly varied. In addition, the SeO
, were studied by means of conductivity measurements and single crystal neutron structure analyses in order to clarify how the elements at the M site affect the Tc from the structural aspect. The conductivity measurements revealed that the Tc decreased as the K content increased. The variation of the Tc was found to be non-linear with respect to the K content. The structure analyses showed that the K ions preferred to occupy one of two M sites and the occupancy of the K ion at this site non-linearly varied. In addition, the SeO tetrahedron became more distorted as the K content increased, suggesting the close relationship between the distortion and the Tc.
tetrahedron became more distorted as the K content increased, suggesting the close relationship between the distortion and the Tc.
 H(SeO
H(SeO )
)
Kiyanagi, Ryoji; Matsuo, Yasumitsu*; Ishikawa, Yoshihisa*; Noda, Yukio*; Ohara, Takashi; Kawasaki, Takuro; Oikawa, Kenichi; Kaneko, Koji; Tamura, Itaru; Hanashima, Takayasu*; et al.
no journal, ,
The materials represented as M H(XO
H(XO )
) (M=alkali metals, X=Se, S) are known to exhibit high protonic conductivities at relatively low temperature. Although the high protonic conductivity is considered to be due to the disorder of the hydrogen bonds in the materials, details have not been well understood. In addition, clarification of the phase transition to the high proton conducting phase is important in order to understand the realization of the high protonic conductivity. In this study, a high temperature neutron structural study was carried out on Rb
(M=alkali metals, X=Se, S) are known to exhibit high protonic conductivities at relatively low temperature. Although the high protonic conductivity is considered to be due to the disorder of the hydrogen bonds in the materials, details have not been well understood. In addition, clarification of the phase transition to the high proton conducting phase is important in order to understand the realization of the high protonic conductivity. In this study, a high temperature neutron structural study was carried out on Rb H(SeO
H(SeO )
) , and the solid solution of Rb
, and the solid solution of Rb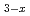 K
K H(SeO
H(SeO )
) were also investigated by means of conductivity measurements and neutron structural studies. The high temperature neutron structure analyses revealed that the protons in the high proton conducting phase were two dimensionally distributed, which is considered to be the direct observation of the conducting protons. The solid solution exhibited non-linear decreases of the phase transition temperatures as the K concentration increased. The K ions were found to prefer to occupy one of two possible sites, of which occupancies apparently have close relationship with the phase transition temperature.
were also investigated by means of conductivity measurements and neutron structural studies. The high temperature neutron structure analyses revealed that the protons in the high proton conducting phase were two dimensionally distributed, which is considered to be the direct observation of the conducting protons. The solid solution exhibited non-linear decreases of the phase transition temperatures as the K concentration increased. The K ions were found to prefer to occupy one of two possible sites, of which occupancies apparently have close relationship with the phase transition temperature.
Uemura, Takuya*; Kita, Akiko*; Adachi, Motoyasu; Kuroki, Ryota; Morimoto, Yukio*
no journal, ,
no abstracts in English