Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Narukawa, Takafumi; Kondo, Keietsu; Fujimura, Yuki; Kakiuchi, Kazuo; Udagawa, Yutaka; Nemoto, Yoshiyuki
Journal of Nuclear Materials, 587, p.154736_1 - 154736_8, 2023/12
Times Cited Count:1 Percentile:63.33(Materials Science, Multidisciplinary) 10
10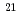 W/cm
W/cm
Skobelev, I. Yu.*; Ryazantsev, S. N.*; Kulikov, R. K.*; Sedov, M. V.*; Filippov, E. D.*; Pikuz, S. A.*; Asai, Takafumi*; Kanasaki, Masato*; Yamauchi, Tomoya*; Jinno, Satoshi; et al.
Photonics (Internet), 10(11), p.1250_1 - 1250_11, 2023/11
Times Cited Count:0 Percentile:0.00(Optics)It is challenging to clearly distinguish the impacts of the optical field and collisional ionization in the evolution of the charge state of a plasma produced when matter interacts with high-intensity laser pulses. In this work, time-dependent calculations of plasma kinetics are used to show that it is possible only when low-density gaseous targets with sufficiently small clusters are used. In the case of Ar plasma, the upper limit of the cluster radius was estimated to be 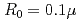 m.
m.
Narukawa, Takafumi; Kondo, Keietsu; Fujimura, Yuki; Kakiuchi, Kazuo; Udagawa, Yutaka; Nemoto, Yoshiyuki
Journal of Nuclear Materials, 582, p.154467_1 - 154467_12, 2023/08
Times Cited Count:3 Percentile:92.52(Materials Science, Multidisciplinary)Kimura, Atsushi; Taguchi, Mitsumasa; Kondo, Takafumi*; Yang, J.*; Nagaishi, Ryuji; Yoshida, Yoichi*; Hirota, Koichi
Radiation Physics and Chemistry, 79(11), p.1159 - 1164, 2010/11
Times Cited Count:3 Percentile:23.88(Chemistry, Physical)Much of this past research has focused on finding more effective reactions between charged reactive species in room temperature ionic liquid (RTILs), which have a coulombic field. Reactions in RTILs involving the charged reactive species, particularly solvated electrons, have been studied by the techniques of radiation chemistry. The reaction behavior of halophenols in RTILs were investigated by  ray and pulsed electron radiolysis. The yield of CP decomposition in the RTILs was almost the same as that of solvated electrons formation. The yield of CP decomposition and phenol formation in the RTILs was low in the presence of electron scavengers such as nitrous oxide, indicating that solvated electrons primarily contributed to the decomposition of CP. The type of halogen group with the exception of fluorine had a negligible effect on the decomposition of halophenols in the RTILs. Therefore, RTILs could be applied as an reaction media for the reduction by ionizing radiation.
ray and pulsed electron radiolysis. The yield of CP decomposition in the RTILs was almost the same as that of solvated electrons formation. The yield of CP decomposition and phenol formation in the RTILs was low in the presence of electron scavengers such as nitrous oxide, indicating that solvated electrons primarily contributed to the decomposition of CP. The type of halogen group with the exception of fluorine had a negligible effect on the decomposition of halophenols in the RTILs. Therefore, RTILs could be applied as an reaction media for the reduction by ionizing radiation.
Kondo, Takafumi*; Asano, Akira*; Yang, J.*; Norizawa, Kimihiro*; Takahashi, Kenji*; Taguchi, Mitsumasa; Nagaishi, Ryuji; Kato, Ryuji*; Yoshida, Yoichi*
Radiation Physics and Chemistry, 78(12), p.1157 - 1160, 2009/12
Times Cited Count:28 Percentile:85.57(Chemistry, Physical)Spectrum and kinetic behavior of solvated electrons (e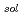
 ) in alkyl ammonium ionic liquids (ILs),
) in alkyl ammonium ionic liquids (ILs), 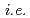 DEMMA-TFSI, DEMMA-BF4, TMPA-TFSI, PP13-TFSI, P13-TFSI and P14-TFSI were investigated by pulse radiolysis method. The e
DEMMA-TFSI, DEMMA-BF4, TMPA-TFSI, PP13-TFSI, P13-TFSI and P14-TFSI were investigated by pulse radiolysis method. The e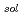
 in the ILs have same absorption peak at 1100 nm with a molar absorption coefficient of 1.5-2.3
in the ILs have same absorption peak at 1100 nm with a molar absorption coefficient of 1.5-2.3 10
10 dm
dm mol
mol cm
cm . The reaction rate constant of e
. The reaction rate constant of e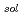
 with Pyrene (Py) was 1.5-3.5
with Pyrene (Py) was 1.5-3.5 10
10 dm
dm mol
mol s
s . These values were about one order of magnitude higher than that of diffusion controlled limit, which was calculated from measured viscosity. The dry electron (e
. These values were about one order of magnitude higher than that of diffusion controlled limit, which was calculated from measured viscosity. The dry electron (e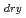
 ) reacts with Py, and its rate constant in DEMMA-TFSI was 7.9
) reacts with Py, and its rate constant in DEMMA-TFSI was 7.9 10
10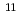 dm
dm mol
mol s
s , three orders of magnitude higher than that of the e
, three orders of magnitude higher than that of the e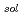
 reactions.
reactions.
Kimura, Atsushi; Taguchi, Mitsumasa; Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*; Hirota, Koichi
Radiation Physics and Chemistry, 77(10-12), p.1253 - 1257, 2008/10
Times Cited Count:11 Percentile:58.97(Chemistry, Physical)Asano, Akira*; Yang, J.*; Kondo, Takafumi*; Norizawa, Kimihiro*; Nagaishi, Ryuji; Takahashi, Kenji*; Yoshida, Yoichi*
Radiation Physics and Chemistry, 77(10-12), p.1244 - 1247, 2008/10
Times Cited Count:25 Percentile:82.34(Chemistry, Physical)The molar extinction coefficient and the radiolytic yield of solvated electrons in ionic liquid of DEMMA-TFSI were estimated respectively to be 2.3 10
10 L mol
L mol cm
cm and 0.8
and 0.8 10
10 mol J
mol J by observing the reaction and kinetics of solvated electron and dry electron in the ionic liquid. The solvated electron in DEMMA-TFSI has a very broad absorption spectrum peaking at 1100 nm and reacts slowly with pyrene with a rate constant of 3.5
by observing the reaction and kinetics of solvated electron and dry electron in the ionic liquid. The solvated electron in DEMMA-TFSI has a very broad absorption spectrum peaking at 1100 nm and reacts slowly with pyrene with a rate constant of 3.5 10
10 L mol
L mol s
s , while the dry electron is captured rapidly by pyrene with C
, while the dry electron is captured rapidly by pyrene with C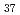 value of 0.065 mol L
value of 0.065 mol L .
.
Yang, J.*; Kondo, Takafumi*; Norizawa, Kimihiro*; Nagaishi, Ryuji; Taguchi, Mitsumasa; Takahashi, Kenji*; Kato, Ryuji*; Anishchik, S. V.*; Yoshida, Yoichi*; Tagawa, Seiichi*
Radiation Physics and Chemistry, 77(10-12), p.1233 - 1238, 2008/10
Times Cited Count:25 Percentile:82.34(Chemistry, Physical)First observation of picosecond dynamics in ionic liquid of DEMMA-TFSI in radiation chemistry was reported. It is found that the electron produced by ionization is solvated to full solvation in ionic liquid with a rate constant of 3.9 10
10 s
s . The yield of solvated electrons in picosecond time region is 1.2
. The yield of solvated electrons in picosecond time region is 1.2 10
10 mol J
mol J . The dry electron in ionic liquid reacts rapidly with biphenyl and pyrene with a rate constant of 3.8 to 7.9
. The dry electron in ionic liquid reacts rapidly with biphenyl and pyrene with a rate constant of 3.8 to 7.9 10
10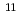 L mol
L mol s
s . The geminate ion recombination in n-dodecane and n-hexane was also observed by monitoring the transient absorptions at 523 nm.
. The geminate ion recombination in n-dodecane and n-hexane was also observed by monitoring the transient absorptions at 523 nm.
Kondo, Takafumi*; Yang, J.*; Kan, Koichi*; Yoshida, Yoichi*; Shibata, Hiromi*; Taguchi, Mitsumasa; Kojima, Takuji
JAEA-Review 2007-060, JAEA Takasaki Annual Report 2006, P. 160, 2008/03
no abstracts in English
Kondo, Takafumi*; Yang, J.*; Kan, Koichi*; Yoshida, Yoichi*; Shibata, Hiromi*; Taguchi, Mitsumasa; Kojima, Takuji
JAEA-Review 2006-042, JAEA Takasaki Annual Report 2005, P. 141, 2007/02
no abstracts in English
Yoshida, Yoichi*; Yang, J.*; Kondo, Takafumi*; Seki, Shuhei*; Kozawa, Takahiro*; Tagawa, Seiichi*; Shibata, Hiromi*; Taguchi, Mitsumasa; Kojima, Takuji; Namba, Hideki
JAEA-Review 2005-001, TIARA Annual Report 2004, p.183 - 185, 2006/01
A heavy-ion-pulse radiolysis technology was developed using a single-photon-counting system. In the system, the ion beam was injected a thin scintillator before irradiating the sample. The light emitted from the scintillator by the ion irradiation was used as analyzing source to detect the absorption of primary species in water. Measurement of time-dependent absorption of hydrated electrons in water was achieved using the system, which demonstrates the usefulness of this technique.
Asano, Akira*; Kondo, Takafumi*; Yang, J.*; Taguchi, Mitsumasa; Nagaishi, Ryuji; Takahashi, Kenji*; Yoshida, Yoichi*
no journal, ,
The spectrum and kinetic behavior of solvated electrons in DEMMA-TFSI and DEMMA-BF ionic liquids (ILs) were investigated by using nanosecond pulse radiolysis technique. The reactions of the solvated electrons with pyrene were studied. The molar extinction coefficient and G value of the solvated electrons in ILs were estimated to be 2.2
ionic liquids (ILs) were investigated by using nanosecond pulse radiolysis technique. The reactions of the solvated electrons with pyrene were studied. The molar extinction coefficient and G value of the solvated electrons in ILs were estimated to be 2.2 10
10 M
M cm
cm (at 1100nm) and 0.9#100eV
(at 1100nm) and 0.9#100eV . The formation of the solvated electrons in IL on picosecond time scale was observed by picosecond pulse radiolysis.
. The formation of the solvated electrons in IL on picosecond time scale was observed by picosecond pulse radiolysis.
Kondo, Takafumi*; Yang, J.*; Kan, Koichi*; Yoshida, Yoichi*; Shibata, Hiromi*; Taguchi, Mitsumasa; Kojima, Takuji
no journal, ,
no abstracts in English
Aoyama, Takafumi; Okazaki, Koki*; Ito, Chikara; Iguchi, Tetsuo*; Watanabe, Kenichi*; Takegawa, Nobuyuki*; Kondo, Yutaka*
no journal, ,
no abstracts in English
Kondo, Takafumi*; Yang, J.*; Kan, Koichi*; Norizawa, Kimihiro*; Shibata, Hiromi*; Taguchi, Mitsumasa; Kurashima, Satoshi; Hirota, Koichi
no journal, ,
no abstracts in English
Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*; Nagaishi, Ryuji; Taguchi, Mitsumasa; Takahashi, Kenji*; Kato, Ryuji*
no journal, ,
The effect of the coexistence water on the solvated electrons in ammonium ionic liquids was studied by the electron pulse radiolysis method. The optical absorption band of the solvation electron decreased by coexistence of water, and new absorption band like a hydrated electron were generated in the medium wavelength region between solvated electron and hydrated electron. Furthermore, if quantity of water increase in the hydrophilic ionic liquid, it was found that the spectrum changed into the hydrated electron.
Kimura, Atsushi; Taguchi, Mitsumasa; Hirota, Koichi; Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*
no journal, ,
no abstracts in English
Kondo, Takafumi*; Yang, J.*; Taguchi, Mitsumasa; Kurashima, Satoshi; Kan, Koichi*; Yoshida, Yoichi*; Shibata, Hiromi*; Hirota, Koichi
no journal, ,
no abstracts in English
Nagaishi, Ryuji; Aoyagi, Noboru; Shinha, Yusuke; Taguchi, Mitsumasa; Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*
no journal, ,
Radiolytic behavior of ionic liquids of thiocyanate salts was studied by using the ns-pulse radiolysis technique. The transient absorption of thiocyante dimer radical ion was observed in the liquids, and the rise was within the width of electron pulse. The maximum absorbance of dimer in the liquids was equivalent to that in aqueous solutions, where the dimer is formed from the reaction of thiocyanate ion with OH, and increased linearly with increasing the thiocyanate concentration in the liquids. These indicate that one of the ionization sites of liquids should be thiocyanate ion.
Kondo, Takafumi*; Yang, J.*; Kan, Koichi*; Yoshida, Yoichi*; Shibata, Hiromi*; Kurashima, Satoshi; Taguchi, Mitsumasa; Hirota, Koichi
no journal, ,
no abstracts in English