Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Oe, Kazuhiro*; Attallah, M. F.*; Asai, Masato; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke*; Kasamatsu, Yoshitaka*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1317 - 1320, 2015/02
Times Cited Count:10 Percentile:60.68(Chemistry, Analytical)A new technique for continuous dissolution of nuclear reaction products transported by a gas-jet system was developed for superheavy element (SHE) chemistry. In this technique, a hydrophobic membrane is utilized to separate an aqueous phase from the gas phase. With this technique, the dissolution efficiencies of short-lived radionuclides of 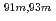 Mo and
Mo and 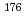 W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
Oe, Kazuhiro; Tsukada, Kazuaki; Asai, Masato; Sato, Tetsuya; Toyoshima, Atsushi; Miyashita, Sunao; Nagame, Yuichiro; Sch del, M.; Kaneya, Yusuke*; Lerum, H. V.*; et al.
del, M.; Kaneya, Yusuke*; Lerum, H. V.*; et al.
no journal, ,
We are planning to investigate the redox potentials of element 106, seaborgium, with a combination of the rapid liquid-liquid extraction apparatus SISAK and a flow electrolytic column. In order to combine these two apparatuses, we need to reduce the liquid flow-rate of the SISAK system. In this study, a new degasser, which works with a lower flow rate, was developed for SISAK. It separates the gas-liquid mixture using a hydrophobic Teflon membrane (only the gas can go through the membrane). Using this new degasser, dissolution efficiencies of gas-jet transported products were measured. At the typical flow rate for the SISAK system (0.4 mL/s), a dissolution efficiency of approximately 80% was obtained. A high yield of around 80% was also observed at a flow rate of 0.1 mL/s with a mixer for the gas-liquid mixing.
Oe, Kazuhiro; Tsukada, Kazuaki; Asai, Masato; Sato, Tetsuya; Toyoshima, Atsushi; Miyashita, Sunao; Nagame, Yuichiro; Sch del, M.; Kaneya, Yusuke*; Lerum, H. V.*; et al.
del, M.; Kaneya, Yusuke*; Lerum, H. V.*; et al.
no journal, ,
We are planning to investigate the redox behavior of element 106, seaborgium (Sg). For the Sg study, we newly developed a continuous dissolution apparatus which dissolves the gas-jet transported nuclides produced in nuclear reactions into aqueous solution. The new apparatus has a hydrophobic membrane filter for separation of aqueous solution from the gas. We investigated the dissolution efficiencies with the apparatus for short-lived nuclides of molybdenum and tungsten which are lighter homologues of Sg. In the meeting, the dependence of the efficiencies on the aqueous- and gas-flow rates will be reported.
Miyashita, Sunao; Toyoshima, Atsushi; Oe, Kazuhiro*; Asai, Masato; Sato, Tetsuya; Tsukada, Kazuaki; Nagame, Yuichiro; Sch del, M.; Kaneya, Yusuke; Haba, Hiromitsu*; et al.
del, M.; Kaneya, Yusuke; Haba, Hiromitsu*; et al.
no journal, ,
Solvent extraction of  Mo and
Mo and 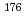 W using 4-isopropyltropolone (Hinokitiol, HT) was investigated. Extraction mechanism of Mo and W with HT was examined by slope analysis. The slopse of the distribution ratio of Mo and W vs. [HT] in logarithmic scale are 1.88 and 1.54, respectively.
W using 4-isopropyltropolone (Hinokitiol, HT) was investigated. Extraction mechanism of Mo and W with HT was examined by slope analysis. The slopse of the distribution ratio of Mo and W vs. [HT] in logarithmic scale are 1.88 and 1.54, respectively.
Miyashita, Sunao; Toyoshima, Atsushi; Oe, Kazuhiro*; Sato, Tetsuya; Asai, Masato; Tsukada, Kazuaki; Nagame, Yuichiro; Sch del, M.; Kaneya, Yusuke; Haba, Hiromitsu*; et al.
del, M.; Kaneya, Yusuke; Haba, Hiromitsu*; et al.
no journal, ,
The extraction behavior of hexavalent and reduced Mo and W were investigated when aqueous phase was 0.1 M HCl/0.9 M LiCl solution. The D value of Mo was changed from 10 to 1 when applied potential was near 0 to -0.2 V. On the other hand, the D value of W was not changed at all applied potentials in this experimental condition. Those results indicated that Mo was reduced by FEC, and the extraction behavior of reduced Mo was different from hexavalent Mo. In the case of W, W was not reduced or the D values of reduced W was same as hexavalent W.
Omtvedt, J. P.*; Oe, Kazuhiro; Lerum, H. V.*; Toyoshima, Atsushi; Haba, Hiromitsu*; Tsukada, Kazuaki; Kratz, J. V.*; Nagame, Yuichiro; Sch del, M.
del, M.
no journal, ,
We are planning to investigate the redox potentials of element 106, seaborgium, with a combination of the automated liquid-liquid extraction system SISAK and a flow electrolytic column. In order to combine these two apparatuses, we need to reduce the liquid flow-rate of the SISAK system. In this study, a new degasser, which works with a lower flow rate, was developed for SISAK. It separates the gas-liquid mixture using a hydrophobic Teflon membrane (only the gas can go through the membrane). Using this new degasser, dissolution efficiencies of gas-jet transported products were measured. A high yield of around 80% was observed at a flow rate of 0.1 mL/s with a mixer for the gas-liquid mixing. This result has never been achieved with the conventional SISAK degasser. This is an important step forward with respect to combining the SISAK system with a flow electrolytic column for performing redox experiments on Sg.
Toyoshima, Atsushi; Asai, Masato; Attallah, M. F.*; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke; Kasamatsu, Yoshitaka*; et al.
no journal, ,
Towards electrolytic reduction of Sg, batch-wise electrolytic reduction of carrier-free  Mo and
Mo and 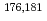 W radiotracers was studied using a flow electrolytic column (FEC). The electrolyzed samples from a FEC were chemically analyzed by solvent extraction with TOA and HDEHP to separate and identify reduced species from the stable Mo(VI) and W(VI) ones based on their different extraction behavior.
W radiotracers was studied using a flow electrolytic column (FEC). The electrolyzed samples from a FEC were chemically analyzed by solvent extraction with TOA and HDEHP to separate and identify reduced species from the stable Mo(VI) and W(VI) ones based on their different extraction behavior.  Mo and
Mo and 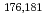 W were applied as radiotracers. We also performed cyclic voltammetry and UV/Vis absorption spectrometry of macro amounts of Mo and W in acidic solutions to obtain information on redox reactions of these elements under given conditions. In the conference, the present status of the preparatory reduction experiments with Mo and W will be presented.
W were applied as radiotracers. We also performed cyclic voltammetry and UV/Vis absorption spectrometry of macro amounts of Mo and W in acidic solutions to obtain information on redox reactions of these elements under given conditions. In the conference, the present status of the preparatory reduction experiments with Mo and W will be presented.
Toyoshima, Atsushi; Miyashita, Sunao*; Oe, Kazuhiro*; Kitayama, Yuta*; Lerum, H. V.*; Goto, Naoya*; Kaneya, Yusuke; Komori, Yukiko*; Mitsukai, Akina*; Vascon, A.; et al.
no journal, ,
no abstracts in English
Toyoshima, Atsushi; Oe, Kazuhiro*; Asai, Masato; Attallah, M. F.*; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Kaneko, Masashi*; Kaneya, Yusuke; Kasamatsu, Yoshitaka*; et al.
no journal, ,
Due to short half-lives less than 10 s and extremely low production rates, transactinide elements heavier than seaborgium (Sg) are produced on an atom per hour scale. Therefore, a continuous rapid chemistry assembly is required to study aqueous-phase chemistry of these heaviest elements. In the present study, we started developments of a continuous chemistry assembly. Our first attempt was made in on-line experiments with Mo and W, lighter homologs of Sg, to optimize a chemistry assembly consisting of a newly developed membrane degasser as an interface between gas-jet and aqueous phase, a flow electrolytic column apparatus utilized to control oxidation states of Mo and W ions, and the continuous liquid-liquid extraction apparatus of SISAK for separation. In the conference, present status of the developments will be presented.
Toyoshima, Atsushi; Miyashita, Sunao*; Asai, Masato; Sato, Tetsuya; Kaneya, Yusuke; Tsukada, Kazuaki; Kitatsuji, Yoshihiro; Nagame, Yuichiro; Sch del, M.; Lerum, H. V.*; et al.
del, M.; Lerum, H. V.*; et al.
no journal, ,
To carry out a continuous reduction experiment of Sg with the low production rates and the short half-life, we are developing a new chemistry assembly consisting of a membrane degasser (MDG), a flow electrolytic column (FEC), the continuous liquid-liquid extraction apparatus, and the liquid scintillation counting system (SISAK). Recently, we have begun preparatory studies with Mo and W isotopes. Aqueous solution dissolving Mo and W was successfully separated from a carrier gas. Redox couples of Mo(VI)/Mo(V) and W(VI)/W(V) in HCl have been characterized for their macro amounts. Extraction behavior of Mo(VI) and W(VI) between toluene containing hinokitiol (HT) and HCl was successfully observed by a batch method. On-line extractions of short-lived Mo and W were also carried out using SISAK and MDG. In the symposium, our present status of the preparation with Mo and W will be presented.