Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
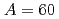
Tamii, Atsushi*; Pellegri, L.*; S derstr
derstr m, P.-A.*; Allard, D.*; Goriely, S.*; Inakura, Tsunenori*; Khan, E.*; Kido, Eiji*; Kimura, Masaaki*; Litvinova, E.*; et al.
m, P.-A.*; Allard, D.*; Goriely, S.*; Inakura, Tsunenori*; Khan, E.*; Kido, Eiji*; Kimura, Masaaki*; Litvinova, E.*; et al.
European Physical Journal A, 59(9), p.208_1 - 208_21, 2023/09
Times Cited Count:7 Percentile:76.15(Physics, Nuclear)no abstracts in English
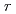 -process nucleosynthesis
-process nucleosynthesisBhattacharyya, A.*; Datta, U.*; Rahaman, A.*; Chakraborty, S.*; Aumann, T.*; Beceiro-Novo, S.*; Boretzky, K.*; Caesar, C.*; Carlson, B. V.*; Catford, W. N.*; et al.
Physical Review C, 104(4), p.045801_1 - 045801_14, 2021/10
Times Cited Count:7 Percentile:64.07(Physics, Nuclear)no abstracts in English
Yamashiki, Yosuke*; Maehara, Hiroyuki*; Airapetian, V.*; Notsu, Yuta*; Sato, Tatsuhiko; Notsu, Shota*; Kuroki, Ryusuke*; Murashima, Keiya*; Sato, Hiroaki*; Namekata, Kosuke*; et al.
Astrophysical Journal, 881(2), p.114_1 - 114_24, 2019/08
Times Cited Count:38 Percentile:80.35(Astronomy & Astrophysics)The impact of Stellar flares on extrasolar planetary systems has been discussed and argued, especially whether there is a potential impact on their life systems. Here, we propose a comprehensive evaluation system for stellar flares, focusing on Stellar Proton Events (SPE) on selected extrasolar planets with hypothetical atmospheres and oceans. This is done by cross-linking KIC flare-observed and flare-estimated stars by their start pots that are directly linked with the Monte Carlo simulation system PHITS through the exoplanetary database system ExoKyoto. The estimated dose at ground level for each planetary surface did not exceed the critical dose for complex animals.
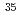 Al via Coulomb breakup
Al via Coulomb breakupChakraborty, S.*; Datta, U.*; Aumann, T.*; Beceiro-Novo, S.*; Boretzky, K.*; Caesar, C.*; Carlson, B. V.*; Catford, W. N.*; Chartier, M.*; Cortina-Gil, D.*; et al.
Physical Review C, 96(3), p.034301_1 - 034301_9, 2017/09
Times Cited Count:4 Percentile:31.80(Physics, Nuclear)no abstracts in English
 )
) ]
] and the crystal structure of Na
and the crystal structure of Na [U(CO
[U(CO )
) ]
] 12H
12H O
OHennig, C.*; Ikeda, Atsushi; Emmerling, F.*; Kraus, W.*; Bernhard, G.*
Dalton Transactions, 39(15), p.3744 - 3750, 2010/04
Times Cited Count:40 Percentile:83.80(Chemistry, Inorganic & Nuclear)The coordination of the limiting U(IV) carbonate species in aqueous solution was investigated by comparing its structure parameters with those of the complex preserved in a crystal structure. The U(IV) carbonate complex in the crystal structure was identified as a [U(CO )
) ]
] anionic complex. This monomeric anion complex forms a network with charge compensating Na+ cations and H
anionic complex. This monomeric anion complex forms a network with charge compensating Na+ cations and H O ligands. U L3-edge EXAFS spectra were collected from the solid Na
O ligands. U L3-edge EXAFS spectra were collected from the solid Na [U(CO
[U(CO )
) ]
] 12H
12H O and the corresponding solution. The obtained data indicate the identity of the [U(CO
O and the corresponding solution. The obtained data indicate the identity of the [U(CO )
) ]
] complex in solid and solution states. The high negative charge of the [U(CO
complex in solid and solution states. The high negative charge of the [U(CO )
) ]
] anionic complex is compensated by Na+ cations. In the solid state the Na+ cations form a bridging network between the [U(CO
anionic complex is compensated by Na+ cations. In the solid state the Na+ cations form a bridging network between the [U(CO )
) ]
] anion complex, while in the liquid state they seem to be located closer at the anionic complex anion.
anion complex, while in the liquid state they seem to be located closer at the anionic complex anion.
Hennig, C.*; Kraus, W.*; Emmerling, F.*; Ikeda, Atsushi; Scheinost, A. C.*
Inorganic Chemistry, 47(5), p.1634 - 1638, 2008/02
Times Cited Count:29 Percentile:73.65(Chemistry, Inorganic & Nuclear)Uranium(IV) sulfate species in an aqueous solution and in the solid state has been investigated by extended X-ray absorption fine structure and X-ray diffraction. The coordination polyhedron was found to comprise unidentate and bidentate sulfate ions, and water molecules both in the aqueous solution phase and in the solid state.
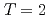 mirrors
mirrors 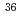 Ca and
Ca and 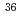 S; A Test for isospin symmetry of shell gaps at the driplines
S; A Test for isospin symmetry of shell gaps at the driplinesDoornenbal, P.*; Reiter, P.*; Grawe, H.*; Otsuka, Takaharu*; Al-Khatib, A.*; Banu, A.*; Beck, T.*; Becker, F.*; Bednarczyk, P.*; Benzoni, G.*; et al.
Physics Letters B, 647(4), p.237 - 242, 2007/04
Times Cited Count:35 Percentile:86.80(Astronomy & Astrophysics)The first excited state of 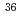 Ca was measured at GSI for the first time. The measured
Ca was measured at GSI for the first time. The measured 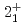 energy is found to be 3015(16) keV, which is lower than its mirror nucleus
energy is found to be 3015(16) keV, which is lower than its mirror nucleus 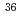 S by as large as 276 keV. The structure of those nuclei is studied by the shell model. It is found that those nuclei can be well described by the
S by as large as 276 keV. The structure of those nuclei is studied by the shell model. It is found that those nuclei can be well described by the 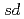 valence space. The large energy shift between them is caused by the Thomas-Ehrman effect. We presented that the energy shift in the
valence space. The large energy shift between them is caused by the Thomas-Ehrman effect. We presented that the energy shift in the 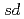 shell region can be explained by the shell model with a phenomenological treatment of the Thomas-Ehrman effect.
shell region can be explained by the shell model with a phenomenological treatment of the Thomas-Ehrman effect.