Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 O
O glass
glassBrazhkin, V. V.*; Akola, J.*; Katayama, Yoshinori; Kohara, Shinji*; Kondrin, M. V.*; Lyapin, A. G.*; Lyapin, S. G.*; Tricot, G.*; Yagafarov, O.
Journal of Materials Chemistry, 21(28), p.10442 - 10447, 2011/07
Times Cited Count:18 Percentile:48.14(Chemistry, Physical)P O
O compound is an archetypical glass-forming oxide with a high hygroscopicity. We found that the quenching from the P
compound is an archetypical glass-forming oxide with a high hygroscopicity. We found that the quenching from the P O
O melt under ultrahigh pressures enables obtaining densified P
melt under ultrahigh pressures enables obtaining densified P O
O glasses with a residual densification up to 12% at normal conditions. These glasses have a low hygroscopicity and can exist under air conditions for several weeks. An examination of the structure of the new form of P
glasses with a residual densification up to 12% at normal conditions. These glasses have a low hygroscopicity and can exist under air conditions for several weeks. An examination of the structure of the new form of P O
O glass reveals a cardinal decrease of the volume of nanovoids in the glassy matrix.
glass reveals a cardinal decrease of the volume of nanovoids in the glassy matrix.
 -As
-As S
S realgar under high pressure
realgar under high pressureTuktabiev, M. A.*; Popova, S. V.*; Brazhkin, V. V.*; Lyapin, A. G.*; Katayama, Yoshinori
Journal of Physics; Condensed Matter, 21(38), p.385401_1 - 385401_7, 2009/08
Times Cited Count:19 Percentile:58.21(Physics, Condensed Matter)The energy-dispersive X-ray diffraction technique has been employed to study the structure and equation of state of realgar As S
S under pressures up to 8 GPa at room temperature. We have obtained pressure dependences of the unit cell parameters and volume for the monoclinic structure of realgar. At a pressure of around 7 GPa, realgar showed a polymorph transition to a new molecular phase with a supposedly orthorhombic structure. The phase transition is likely to be associated with the removal of a monoclinic distortion in the unit cell.
under pressures up to 8 GPa at room temperature. We have obtained pressure dependences of the unit cell parameters and volume for the monoclinic structure of realgar. At a pressure of around 7 GPa, realgar showed a polymorph transition to a new molecular phase with a supposedly orthorhombic structure. The phase transition is likely to be associated with the removal of a monoclinic distortion in the unit cell.
 and ZnCl
and ZnCl halogenides under pressure
halogenides under pressureBrazhkin, V. V.*; Lyapin, A. G.*; Popova, S. V.*; Katayama, Yoshinori; Saito, Hiroyuki; Utsumi, Wataru
Journal of Physics; Condensed Matter, 19(24), p.246104_1 - 246104_10, 2007/06
Times Cited Count:23 Percentile:66.30(Physics, Condensed Matter)Liquid AlCl and ZnCl
and ZnCl halogenides are studied by
halogenides are studied by 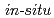 high-pressure high-temperature X-ray diffraction measurements. These halogenides have a quasi-molecular network structure in liquid state at normal pressure. They are intermediate between pure covalent and ionic melts. The present study is indicative of a rapid and strong breakdown of an intermediate range order in a tetrahedral network of melts for the initial pressure range and points to rather sharp transitions in liquids with the formation of a short-range order structure similar to ionic melt structures. Such a two-stage structural transformation under pressure is the general feature for a wide class of simple melts, including most of the pseudo-covalent halogenides.
high-pressure high-temperature X-ray diffraction measurements. These halogenides have a quasi-molecular network structure in liquid state at normal pressure. They are intermediate between pure covalent and ionic melts. The present study is indicative of a rapid and strong breakdown of an intermediate range order in a tetrahedral network of melts for the initial pressure range and points to rather sharp transitions in liquids with the formation of a short-range order structure similar to ionic melt structures. Such a two-stage structural transformation under pressure is the general feature for a wide class of simple melts, including most of the pseudo-covalent halogenides.
Brazhkin, V. V.*; Ekimov, E. A.*; Lyapin, A. G.*; Popova, S. V.*; Rakhmanina, A. V.*; Stishov, S. M.*; Lebedev, V. M.*; Katayama, Yoshinori; Kato, Kenichi*
Physical Review B, 74(14), p.140502_1 - 140502_4, 2006/10
Times Cited Count:58 Percentile:86.83(Materials Science, Multidisciplinary)Using two different high-pressure techniques, we have prepared boron-doped diamonds with atomic concentration of the dopant ranging from 0.04% to 4% (from 7 10
10 to 7
to 7 10
10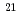 atom/cm
atom/cm ) and studied the lattice constants and thermal expansion of the diamonds in the temperature range from 90 to 300 K. Both sets of samples demonstrate the same increasing concentration dependence of the lattice parameter with maximum shift of the lattice constant about 0.011
) and studied the lattice constants and thermal expansion of the diamonds in the temperature range from 90 to 300 K. Both sets of samples demonstrate the same increasing concentration dependence of the lattice parameter with maximum shift of the lattice constant about 0.011 . We have established an abnormally high thermal expansion of the heavily boron-doped superconducting diamonds with respect to the undoped ones and a nearly linear correlation between lattice constant and critical temperature of the superconducting transition.
. We have established an abnormally high thermal expansion of the heavily boron-doped superconducting diamonds with respect to the undoped ones and a nearly linear correlation between lattice constant and critical temperature of the superconducting transition.
 , AlCl
, AlCl ) under pressure
) under pressureBrazhkin, V. V.*; Katayama, Yoshinori; Lyapin, A. G.*; Popova, S. V.*; Inamura, Yasuhiro*; Saito, Hiroyuki; Utsumi, Wataru
JETP Letters, 82(11), p.713 - 718, 2005/12
Times Cited Count:11 Percentile:54.98(Physics, Multidisciplinary)The results of investigating the phase diagrams of ZnCl and AlCl
and AlCl halides, as well as the structure of the short range order of the corresponding melts under pressures up to 6.5 GPa, by the method of energy-dispersive X-ray diffraction are reported. When a ZnCl
halides, as well as the structure of the short range order of the corresponding melts under pressures up to 6.5 GPa, by the method of energy-dispersive X-ray diffraction are reported. When a ZnCl crystal is compressed, a phase transition occurs from the
crystal is compressed, a phase transition occurs from the  phase (HgI
phase (HgI structure type) to the
structure type) to the  phase (distorted CdI
phase (distorted CdI structure, WTe
structure, WTe type). The structural studies of the liquid state of ZnCl
type). The structural studies of the liquid state of ZnCl and AlCl
and AlCl indicate that the intermediate-range order decreases rapidly in the tetrahedral network of both melts as the pressure increases to 1.8 and 2.3 GPa for ZnCl
indicate that the intermediate-range order decreases rapidly in the tetrahedral network of both melts as the pressure increases to 1.8 and 2.3 GPa for ZnCl and AlCl
and AlCl , respectively. With further compression, the transitions in both melts occur with a change in the structure of the short-range order and with an increase in the coordination number. In this case, the transition in AlCl
, respectively. With further compression, the transitions in both melts occur with a change in the structure of the short-range order and with an increase in the coordination number. In this case, the transition in AlCl occurs at about 4 GPa and is a sharp first order transition, whereas the transition in ZnCl
occurs at about 4 GPa and is a sharp first order transition, whereas the transition in ZnCl occurs more smoothly in a pressure range of 2-4 GPa with a maximum intensity near 3 GPa. Thus the AlCl
occurs more smoothly in a pressure range of 2-4 GPa with a maximum intensity near 3 GPa. Thus the AlCl and ZnCl
and ZnCl compounds exemplify the existence of two phenomena, gradual decay of intermidiate-range structural correlations and a sharper liquid-liquid coordination transition.
compounds exemplify the existence of two phenomena, gradual decay of intermidiate-range structural correlations and a sharper liquid-liquid coordination transition.
 O
O under high pressure
under high pressureBrazhkin, V. V.*; Katayama, Yoshinori; Inamura, Yasuhiro; Kondrin, M. V.*; Lyapin, A. G.*; Popova, S. V.*; Voloshin, R. N.*
JETP Letters, 78(6), p.393 - 397, 2003/06
Times Cited Count:32 Percentile:78.08(Physics, Multidisciplinary)We present in situ (X-ray diffraction) and ex situ (quenching) structural studies of crystalline, liquid, and glassy B O
O up to 9 GPa and 1700 K, drawing equilibrium and nonequilibrium phase diagrams of B
up to 9 GPa and 1700 K, drawing equilibrium and nonequilibrium phase diagrams of B O
O , Particularly, we have determined the melting curve, the stability regions for crystalline B
, Particularly, we have determined the melting curve, the stability regions for crystalline B O
O and B
and B O
O II modifications, the regions of transformations, such as densification or crystallization, for both the liquid and glassy states, including the region of sharp first-order-like transition in liquid B
II modifications, the regions of transformations, such as densification or crystallization, for both the liquid and glassy states, including the region of sharp first-order-like transition in liquid B O
O to a high-density phase near 7 GPa. Quenching experiments also show that the transition to the high-density liquid can occur at much lower pressures in nonstoichiometric melts with an excess of boron. B
to a high-density phase near 7 GPa. Quenching experiments also show that the transition to the high-density liquid can occur at much lower pressures in nonstoichiometric melts with an excess of boron. B O
O is the first glassformer whose transformations in the disordered state have been comparatively studied for both liquid and glassy phases.
is the first glassformer whose transformations in the disordered state have been comparatively studied for both liquid and glassy phases.