Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Dei, Shuntaro; Shibata, Masahito*; Negishi, Kumi*; Sugiura, Yuki; Amano, Yuki; Bateman, K.*; Wilson, J.*; Yokoyama, Tatsunori; Kagami, Saya; Takeda, Masaki; et al.
Results in Earth Sciences (Internet), 3, p.100097_1 - 100097_16, 2025/12
Interactions between cement and host rock in geological repositories for radioactive waste will result in a chemically disturbed zone, which may potentially affect the long-term safety. This paper investigates the chemical evolution at the interface between cement (Ordinary Portland Cement: OPC and Low Alkaline Cement: LAC) and mudstone after 11 years of in situ reactions at the Horonobe Underground Research Laboratory. The study combines various analytical techniques to identify the key reactions at the cement-rock interface, including cement dissolution, precipitation of secondary minerals such as calcite and C-(A-)S-H phases, cation exchange in montmorillonite and reduced porosity in mudstone. The study also highlights the effects of cement-mudstone interactions on radionuclide migration, such as reduction of diffusivity due to reduced porosity and enhancement of sorption due to incorporation into secondary minerals in the altered mudstone.
Aoyama, Takahito; Choudhary, S.*; Pandaleon, A.*; Burns, J. T.*; Kokaly, M.*; Restis, J.*; Ross, J.*; Kelly, R. G.*
Corrosion, 81(6), p.609 - 621, 2025/06
 Co
Co )Sn studied by
)Sn studied by  SR
SRCai, Y.*; Yoon, S.*; Sheng, Q.*; Zhao, G.*; Seewald, E. F.*; Ghosh, S.*; Ingham, J.*; Pasupathy, A. N.*; Queiroz, R.*; Lei, H.*; et al.
Physical Review B, 111(21), p.214412_1 - 214412_17, 2025/06
Times Cited Count:0Wilson, J.*; Sasamoto, Hiroshi; Tachi, Yukio; Kawama, Daisuke*
Applied Clay Science, 275, p.107862_1 - 107862_15, 2025/05
Times Cited Count:0High-Level Radioactive Waste (HLW) repositories include iron or steel-based containers/overpack and bentonite buffers. Over the last 25 years or so, research efforts have attempted to elucidate the nature of iron-bentonite interactions, especially the potential for the deleterious alteration of the swelling clay component (smectite), to iron-rich layer silicates, some of which lack the capacity for intracrystalline swelling. This could result in a reduction or loss in swelling pressure in the bentonite buffer which is designed to protect waste containers from shear forces and also acts to restrict water and solute transport, as part of an engineered barrier system. Most data on iron-bentonite interactions come from experimental and geochemical modelling studies, as natural analogue data are lacking. The data suggests that there is the potential for the development of an iron-rich bentonite alteration zone with smectite (generally present as the aluminous montmorillonite type) undergoing alteration to iron-rich solids, including layer silicates and steel corrosion products such as green rust or magnetite. The evidence available is complex, arguably incomplete, with many potential complex couplings. Many uncertainties remain despite efforts taken over the last 25 years, but plausible scenarios for iron-bentonite interactions have been identified and possible implications for buffer properties have been suggested.
Kato, Masaru*; Zheng, J.*; Deng, Y.*; Saito, Fumie*; Unuma, Yuki*; Oka, Sayuki*; Tamura, Kazuhisa; Yagi, Ichizo*
ACS Catalysis, 15(10), p.7710 - 7719, 2025/04
Times Cited Count:0 Percentile:0.00(Chemistry, Physical) hydridosilicate at high pressures; A Bridge to BaSiH
hydridosilicate at high pressures; A Bridge to BaSiH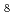 polyhydride
polyhydrideBeyer, D. C.*; Spektor, K.*; Vekilova, O. Y.*; Grins, J.*; Barros Brant Carvalho, P. H.*; Leinbach, L. J.*; Sannemo-Targama, M.*; Bhat, S.*; Baran, V.*; Etter, M.*; et al.
ACS Omega (Internet), 10(15), p.15029 - 15035, 2025/04
Times Cited Count:0 Percentile:0.00(Chemistry, Multidisciplinary)Hydridosilicates featuring SiH octahedral moieties represent a rather new class of compounds with potential properties relating to hydrogen storage and hydride ion conductivity. Here, we report on the new representative BaSiH
octahedral moieties represent a rather new class of compounds with potential properties relating to hydrogen storage and hydride ion conductivity. Here, we report on the new representative BaSiH obtained from reacting the Zintl phase hydride BaSiH
obtained from reacting the Zintl phase hydride BaSiH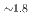 with H
with H fluid at pressures above 4 GPa and subsequent decompression to ambient pressure. It consists of complex SiH
fluid at pressures above 4 GPa and subsequent decompression to ambient pressure. It consists of complex SiH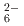 ions, which are octahedrally coordinated by Ba
ions, which are octahedrally coordinated by Ba counterions. The arrangement of Ba and Si atoms deviates only slightly from an ideal fcc NaCl structure. IR and Raman spectroscopy showed SiH
counterions. The arrangement of Ba and Si atoms deviates only slightly from an ideal fcc NaCl structure. IR and Raman spectroscopy showed SiH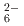 bending and stretching modes in the ranges 800-1200 and 1400-1800 cm
bending and stretching modes in the ranges 800-1200 and 1400-1800 cm , respectively. BaSiH
, respectively. BaSiH is thermally stable up to 95
is thermally stable up to 95 C above which decomposition into BaH
C above which decomposition into BaH and Si takes place. DFT calculations indicated a direct band gap of 2.5 eV. The discovery of BaSiH
and Si takes place. DFT calculations indicated a direct band gap of 2.5 eV. The discovery of BaSiH consolidates the compound class of hydridosilicates, accessible from hydrogenations of silicides at gigapascal pressures (
consolidates the compound class of hydridosilicates, accessible from hydrogenations of silicides at gigapascal pressures (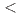 10 GPa). The structural properties of BaSiH
10 GPa). The structural properties of BaSiH suggest that it presents an intermediate (or precursor) for further hydrogenation at considerably higher pressures to the predicted superconducting polyhydride BaSiH
suggest that it presents an intermediate (or precursor) for further hydrogenation at considerably higher pressures to the predicted superconducting polyhydride BaSiH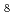 .
.
Hu, F.-F.*; Qin, T.-Y.*; Ao, N.*; Xu, P. G.; Su, Y. H.; Parker, J. D.*; Shinohara, Takenao; Shobu, Takahisa; Kang, G.-Z.*; Ren, M.-M.; et al.
Journal of Traffic and Transportation Engineering, 25(2), p.75 - 93, 2025/04
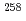 Md produced in the
Md produced in the  He +
He + 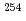 Es reaction
Es reactionNishio, Katsuhisa; Hirose, Kentaro; Makii, Hiroyuki; Orlandi, R.; Kean, K. R.*; Tsukada, Kazuaki; Toyoshima, Atsushi*; Asai, Masato; Sato, Tetsuya; Chiera, N. M.*; et al.
Physical Review C, 111(4), p.044609_1 - 044609_12, 2025/04
Times Cited Count:0 Percentile:0.00(Physics, Nuclear) rays in the
rays in the 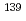 La(
La(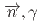 )
)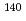 La reaction
La reactionOkuizumi, Mao*; Auton, C. J.*; Endo, Shunsuke; Fujioka, Hiroyuki*; Hirota, Katsuya*; Ino, Takashi*; Ishizaki, Kohei*; Kimura, Atsushi; Kitaguchi, Masaaki*; Koga, Jun*; et al.
Physical Review C, 111(3), p.034611_1 - 034611_6, 2025/03
Times Cited Count:0 Percentile:0.00(Physics, Nuclear)Lee, J.; Rossi, F.; Kodama, Yu; Hironaka, Kota; Koizumi, Mitsuo; Sano, Tadafumi*; Matsuo, Yasunori*; Hori, Junichi*
Annals of Nuclear Energy, 211, p.111017_1 - 111017_7, 2025/02
Times Cited Count:0 Percentile:0.00(Nuclear Science & Technology)Naeem, M.*; Ma, Y.*; Tian, J.*; Kong, H.*; Romero-Resendiz, L.*; Fan, Z.*; Jiang, F.*; Gong, W.; Harjo, S.; Wu, Z.*; et al.
Materials Science & Engineering A, 924, p.147819_1 - 147819_10, 2025/02
Times Cited Count:1 Percentile:0.00(Nanoscience & Nanotechnology)Yoon, J.-Y.*; Takeuchi, Yutaro*; Takechi, Ryota*; Han, J.*; Uchiyama, Tomohiro*; Yamane, Yuta*; Kanai, Shun*; Ieda, Junichi; Ohno, Hideo*; Fukami, Shunsuke*
Nature Communications (Internet), 16, p.1171_1 - 1171_8, 2025/02
Times Cited Count:0 Percentile:0.00(Multidisciplinary Sciences)Nozaki, Yukio*; Sukegawa, Hiroaki*; Watanabe, Shinichi*; Yunoki, Seiji*; Horaguchi, Taisuke*; Nakayama, Hayato*; Yamanoi, Kazuto*; Wen, Z.*; He, C.*; Song, J.*; et al.
Science and Technology of Advanced Materials, 26(1), p.2428153_1 - 2428153_39, 2025/02
Times Cited Count:0 Percentile:0.00(Materials Science, Multidisciplinary)Metcalfe, R.*; Benbow, S. J.*; Kawama, Daisuke*; Tachi, Yukio
Science of the Total Environment, 958, p.177690_1 - 177690_17, 2025/01
Uplifting fractured granitic rocks occur in substantial areas of countries such as Japan. A repository site would be selected in such an area only if it is possible to make a safety case, accounting for the changing conditions during uplift. The safety case must include robust arguments that chemical processes in the rocks around the repository will contribute sufficiently to minimise radiological doses to biosphere receptors. To provide confidence in the safety arguments, numerical models need to be sufficiently realistic, but also parameterised conservatively (pessimistically). However, model development is challenging because uplift involves many complex couplings between groundwater flow, chemical reactions between water and rock, and changing rock properties. The couplings would affect radionuclide mobilisation and retardation, by influencing diffusive radionuclide fluxes between groundwater flowing in fractures and effectively immobile porewater in the rock matrix and radionuclide partitioning between water and solid phases, via: (i) mineral precipitation/dissolution; (ii) mineral alteration; and (iii) sorption/desorption. It is difficult to represent all this complexity in numerical models while showing that they are parameterised conservatively. Here we present a modelling approach, illustrated by simulation cases for some exemplar radioelements, to identify realistically conservative process conceptualisations and model parameterisations.
Nagata, Mitsuhiro; Fukuda, Shoma; Sueoka, Shigeru; Nakajima, Toru; Yokoyama, Tatsunori; Wall, C. J.*; Danhara, Toru*; Iwano, Hideki*; Tagami, Takahiro*
Fuisshon, Torakku Nyusureta, (37), p.11 - 13, 2024/12
In recent years, advances in analytical technology have made it possible to measure the U-Th-Pb dating of young geological units (e.g.  1 Ma). However, there are very few young standard samples available for evaluation. In this study, we attempted U-Pb dating using isotope dilution Thermal Ionization Mass Spectrometry (ID-TIMS) on young zircon samples (TRG04 and OGPK). As a result, we obtained preliminary U-Pb ages of 2.6654
1 Ma). However, there are very few young standard samples available for evaluation. In this study, we attempted U-Pb dating using isotope dilution Thermal Ionization Mass Spectrometry (ID-TIMS) on young zircon samples (TRG04 and OGPK). As a result, we obtained preliminary U-Pb ages of 2.6654  0.0016 Ma and 1.1266
0.0016 Ma and 1.1266  0.0014 Ma, respectively.
0.0014 Ma, respectively.
Liu, P.-F.*; Li, X.*; Li, J.*; Zhu, J.*; Tong, Z.*; Kofu, Maiko*; Nirei, Masami; Xu, J.*; Yin, W.*; Wang, F.*; et al.
National Science Review, 11(12), p.nwae216_1 - nwae216_10, 2024/12
Times Cited Count:13 Percentile:94.32(Multidisciplinary Sciences)Xia, C.-J.*; Maruyama, Toshiki; Yasutake, Nobutoshi*; Tatsumi, Toshitaka*
Physical Review D, 110(11), p.114024_1 - 114024_14, 2024/12
Times Cited Count:1 Percentile:35.96(Astronomy & Astrophysics) neutron diffraction
neutron diffractionNaeem, M.*; Ma, Y.*; Knowles, A. J.*; Gong, W.; Harjo, S.; Wang, X.-L.*; Romero Resendiz, L.*; 6 of others*
Materials Science & Engineering A, 916, p.147374_1 - 147374_8, 2024/11
Times Cited Count:3 Percentile:57.76(Nanoscience & Nanotechnology)Watanabe, Tomoaki; Suyama, Kenya; Tada, Kenichi; Ferrer, R. M.*; Hykes, J.*; Wemple, C. A.*
Nuclear Science and Engineering, 198(11), p.2230 - 2239, 2024/11
Times Cited Count:1 Percentile:51.66(Nuclear Science & Technology)A new nuclear data library for the advanced lattice physics code CASMO5 has been prepared based on JENDL-5. In JENDL-5, many essential nuclides for conventional LWR analysis have also been modified based on state-of-the-art evaluations. The new JENDL-5-based CASMO5 library was prepared by replacing as much of the nuclear data of the current CASMO5 ENDF/B-VII.1-based library as possible with JENDL-5. This study verified and validated the new library. Verifications were performed based on the OECD/NEA burnup credit criticality safety benchmark phase III-C, and the calculated k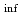 and fuel compositions of the BWR fuel assembly were compared with reported benchmark results. Comparison with the MCNP6.2 result was also performed using the same benchmark model. In addition, the TCA critical experiment and Takahama-3 post-irradiation experiment were used for validation. The results indicate that the new library performs well and is comparable to the ENDF/B-VII.1-based library in predictions of reactivity and fuel compositions for LWR systems.
and fuel compositions of the BWR fuel assembly were compared with reported benchmark results. Comparison with the MCNP6.2 result was also performed using the same benchmark model. In addition, the TCA critical experiment and Takahama-3 post-irradiation experiment were used for validation. The results indicate that the new library performs well and is comparable to the ENDF/B-VII.1-based library in predictions of reactivity and fuel compositions for LWR systems.
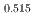 Gd
Gd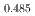 Mn
Mn O
O
Ishii, Yuta*; Sakakura, Terutoshi*; Ishikawa, Yoshihisa*; Kiyanagi, Ryoji; Lustikova, J.*; Aoyama, Takuya*; Ogushi, Kenya*; Wakabayashi, Yusuke*; Kimura, Hiroyuki*; Noda, Yukio*
Physical Review B, 110(18), p.184404_1 - 184404_7, 2024/11
Times Cited Count:0 Percentile:0.00(Materials Science, Multidisciplinary)