Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Matsuda, Shohei; Yokoyama, Keiichi
Isotope News, (786), p.6 - 9, 2023/04
no abstracts in English
Matsuda, Shohei; Nakashima, Nobuaki*; Yokoyama, Keiichi; Taniguchi, Seiji*; Chosrowjan, H.*; Somekawa, Toshihiro*; Yatsuhashi, Tomoyuki*
Chemical Physics Letters, 802, p.139759_1 - 139759_6, 2022/09
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)no abstracts in English
 molecular dynamics simulations reveal the hydration structure of the radium(II) ion
molecular dynamics simulations reveal the hydration structure of the radium(II) ionYamaguchi, Akiko; Nagata, Kojiro*; Kobayashi, Keita; Tanaka, Kazuya; Kobayashi, Toru; Tanida, Hajime; Shimojo, Kojiro; Sekiguchi, Tetsuhiro; Kaneta, Yui; Matsuda, Shohei; et al.
iScience (Internet), 25(8), p.104763_1 - 104763_12, 2022/08
Times Cited Count:9 Percentile:68.46(Multidisciplinary Sciences)no abstracts in English
Matsuda, Shohei; Yokoyama, Keiichi; Yaita, Tsuyoshi; Kobayashi, Toru; Kaneta, Yui; Simonnet, M.; Sekiguchi, Tetsuhiro; Honda, Mitsunori; Shimojo, Kojiro; Doi, Reisuke; et al.
Science Advances (Internet), 8(20), p.eabn1991_1 - eabn1991_11, 2022/05
Times Cited Count:6 Percentile:58.16(Multidisciplinary Sciences)no abstracts in English
Yamaguchi, Akiko; Nagata, Kojiro*; Tanaka, Kazuya; Kobayashi, Keita; Kobayashi, Toru; Shimojo, Kojiro; Tanida, Hajime; Sekiguchi, Tetsuhiro; Kaneta, Yui; Matsuda, Shohei; et al.
Hosha Kagaku, (45), p.28 - 30, 2022/03
no abstracts in English
Nakashima, Nobuaki*; Yatsuhashi, Tomoyuki*; Sakota, Kenji*; Iwakura, Izumi*; Hashimoto, Sena*; Yokoyama, Keiichi; Matsuda, Shohei
Chemical Physics Letters, 752, p.137570_1 - 137570_5, 2020/08
Times Cited Count:1 Percentile:5(Chemistry, Physical)Photo-redox reactions between Eu and Eu
and Eu ions are induced by laser irradiation in alcoholic solution. Efficiency, wavelength dependence, and laser-power dependence are investigated with three different lasers. Nano second laser pulses at a wavelength of 308 nm is found to cause one-photon redox reactions with a quantum yield around 0.5. Nano second laser pulses at a wavelength of 394 nm induces two-photon reduction of Eu
ions are induced by laser irradiation in alcoholic solution. Efficiency, wavelength dependence, and laser-power dependence are investigated with three different lasers. Nano second laser pulses at a wavelength of 308 nm is found to cause one-photon redox reactions with a quantum yield around 0.5. Nano second laser pulses at a wavelength of 394 nm induces two-photon reduction of Eu to form Eu
to form Eu . When the pulse energy is 5 mJ, the quantum yield is measured to be 0.015. Although the quantum yield is one order of magnitude lower than that of the one photon reduction, reduction phenomena can be easily observed under the moderate laser field strength. Because of the two-photon nature, there should be a room to improve the efficiency by increasing the laser field strength.
. When the pulse energy is 5 mJ, the quantum yield is measured to be 0.015. Although the quantum yield is one order of magnitude lower than that of the one photon reduction, reduction phenomena can be easily observed under the moderate laser field strength. Because of the two-photon nature, there should be a room to improve the efficiency by increasing the laser field strength.
 as a source of atmospheric I
as a source of atmospheric I
Watanabe, Kosuke*; Matsuda, Shohei; Cuevas, C. A.*; Saiz-Lopez, A.*; Yabushita, Akihiro*; Nakano, Yukio*
ACS Earth and Space Chemistry (Internet), 3(4), p.669 - 679, 2019/04
Times Cited Count:8 Percentile:43.59(Chemistry, Multidisciplinary)The photooxidation of aqueous iodide ions (I
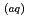 ) at sea surface results in the emission of gaseous iodine molecules (I
) at sea surface results in the emission of gaseous iodine molecules (I
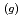 ) into the atmosphere. It plays a certain role in the transport of iodine from ocean to the atmosphere in the natural cycle of iodine. In this study, we determined the photooxidation parameters, the molar absorption coefficient (
) into the atmosphere. It plays a certain role in the transport of iodine from ocean to the atmosphere in the natural cycle of iodine. In this study, we determined the photooxidation parameters, the molar absorption coefficient (
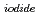 (
( )) and the photooxidative quantum yields (
)) and the photooxidative quantum yields (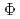
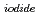 (
( )) of I
)) of I
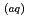 , in the range of 290-500 nm. Through the investigation of the influence of pH and dissolved oxygen (DO) on
, in the range of 290-500 nm. Through the investigation of the influence of pH and dissolved oxygen (DO) on 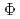
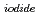 (
( ), the subsequent emission rates of I
), the subsequent emission rates of I
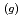 following the photooxidation of I
following the photooxidation of I
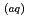 in deionized water solution (pH 5.6, DO 7.8 mg L
in deionized water solution (pH 5.6, DO 7.8 mg L ) and artificial seawater solution (pH 8.0, DO 7.0 mg L
) and artificial seawater solution (pH 8.0, DO 7.0 mg L ) were estimated. A global chemistry-climate model employed herein to assess the I
) were estimated. A global chemistry-climate model employed herein to assess the I
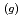 ocean emission on a global scale indicated that the photooxidation of I
ocean emission on a global scale indicated that the photooxidation of I
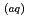 by solar light can enhance the atmospheric iodine budget by up to
by solar light can enhance the atmospheric iodine budget by up to  8% over some oceanic regions.
8% over some oceanic regions.
 by multiphoton excitation in alcohol solutions
by multiphoton excitation in alcohol solutionsMatsuda, Shohei; Nakashima, Nobuaki*; Yokoyama, Keiichi; Yatsuhashi, Tomoyuki*; Chosrowjan, H.*; Taniguchi, Seiji*; Somekawa, Toshihiro*
no journal, ,
The photoredox reactions of lanthanides as a photochemical separative technique proposed in the 1970s are expected to be applied for recovery and recycling of rare metals and nuclear fuel reprocessing. In this study, Eu ions in methanol and ethanol solutions were reduced to Eu
ions in methanol and ethanol solutions were reduced to Eu ions upon irradiation with nanosecond laser pulses. Eu
ions upon irradiation with nanosecond laser pulses. Eu was observed when the laser wavelength was tuned to f-f transition bands at 394 and 465 nm. The formation rate of Eu
was observed when the laser wavelength was tuned to f-f transition bands at 394 and 465 nm. The formation rate of Eu was proportional to the square of the incident laser intensity, indicating that the photoreduction is induced by a two-photon process. The efficiency of the photoreduction in ethanol was higher than that in methanol.
was proportional to the square of the incident laser intensity, indicating that the photoreduction is induced by a two-photon process. The efficiency of the photoreduction in ethanol was higher than that in methanol.
Yokoyama, Keiichi; Matsuda, Shohei
no journal, ,
The scheme of cesium isotope separation proposed by us needs low-frequency terahertz wave, which often suffers from significant loss during wave traveling due to the diffraction. To avoid this difficulty, it may be helpful to design an experiment involving interaction between matter and terahertz wave right after the generation. With this regards, we conducted an experiment to measure terahertz waveform right after the generation with the OH1 crystal, which is known to have high efficiency in both THz generation and electro-optic effect. As a result, we found that the waveform can be measured without bother from the pump pulse, by using reflection of terahertz wave inside the OH1 crystal.
Matsuda, Shohei; Nakashima, Nobuaki*; Yokoyama, Keiichi; Yatsuhashi, Tomoyuki*; Chosrowjan, H.*; Taniguchi, Seiji*; Somekawa, Toshihiro*
no journal, ,
The photochemical reactions of lanthanide and actinide ions are expected to be utilized for recovery and recycling of rare earths, and nuclear fuel reprocessing. Heading towards research for implementation of photochemical purification, a unified evaluation of the efficiency of multiphoton processes is required. In this study, we have evaluated the fluence dependence of photoreduction of Eu(III) more precisely than before by measuring the beam profile of the excitation laser. The reduction rate of Eu(III) Eu(II) is proportional to the square of the laser fluence, indicating that the photoreduction of Eu(III) is induced by a two-photon process.
Eu(II) is proportional to the square of the laser fluence, indicating that the photoreduction of Eu(III) is induced by a two-photon process.
Matsuda, Shohei; Yokoyama, Keiichi; Yaita, Tsuyoshi; Kobayashi, Toru; Nakashima, Nobuaki*
no journal, ,
One of the spectroscopic properties for lanthanides and actinides is that they have f-f transitions in the visible region of their spectra. Those absorption appears at specific wavelength and has narrow line width even in the liquid phase owing to shielded f electrons. It has been reported that reduction reactions of Sm , Eu
, Eu , and Yb
, and Yb are induced by multiphoton excitation through each f-f transition depending on conditions. This photochemical process may be applicable to mutual separation of f elements. In this study, we tried to optically control the oxidation state of americium. As a result, we found that trivalent americium (Am
are induced by multiphoton excitation through each f-f transition depending on conditions. This photochemical process may be applicable to mutual separation of f elements. In this study, we tried to optically control the oxidation state of americium. As a result, we found that trivalent americium (Am ) in nitric acid solution is oxidized to pentavalent one (AmO
) in nitric acid solution is oxidized to pentavalent one (AmO
 ) during nanosecond pulsed-laser irradiation at 503 nm corresponding to the
) during nanosecond pulsed-laser irradiation at 503 nm corresponding to the  L
L
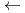
 F
F transition of Am
transition of Am . Identification and quantification of the reactants and products were performed by absorption spectroscopy. Excitation wavelength dependence and fluence dependence of the oxidation suggest that it is initiated by a resonance-enhanced multiphoton process.
. Identification and quantification of the reactants and products were performed by absorption spectroscopy. Excitation wavelength dependence and fluence dependence of the oxidation suggest that it is initiated by a resonance-enhanced multiphoton process.
Matsuda, Shohei; Yokoyama, Keiichi; Yaita, Tsuyoshi; Kobayashi, Toru; Nakashima, Nobuaki*
no journal, ,
We have recently realized the oxidation of trivalent americium (Am ) in nitric acid by multiphoton excitation via an f-f transition. In this study, an effect of nitrate ion (NO
) in nitric acid by multiphoton excitation via an f-f transition. In this study, an effect of nitrate ion (NO
 ) on the Am
) on the Am photooxidation have been investigated. Our results of NO
photooxidation have been investigated. Our results of NO
 activity dependence of the Am
activity dependence of the Am photooxidation and density functional theory calculation indicate that the complex of Am
photooxidation and density functional theory calculation indicate that the complex of Am with NO
with NO
 , in particular one-nitrate complex, contributes to it.
, in particular one-nitrate complex, contributes to it.
Yamaguchi, Akiko; Nagata, Kojiro*; Tanaka, Kazuya; Kobayashi, Keita; Okumura, Masahiko; Kobayashi, Toru; Shimojo, Kojiro; Tanida, Hajime; Sekiguchi, Tetsuhiro; Kaneta, Yui; et al.
no journal, ,
no abstracts in English
Yamaguchi, Akiko; Nagata, Kojiro*; Tanaka, Kazuya; Kobayashi, Keita; Okumura, Masahiko; Kobayashi, Toru; Shimojo, Kojiro; Tanida, Hajime; Sekiguchi, Tetsuhiro; Kaneta, Yui; et al.
no journal, ,
no abstracts in English