Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 /MOX fuel oxidation under severe light water reactor accident conditions
/MOX fuel oxidation under severe light water reactor accident conditionsLiu, J.; Miwa, Shuhei; Karasawa, Hidetoshi; Osaka, Masahiko
Nuclear Materials and Energy (Internet), 37, p.101532_1 - 101532_5, 2023/12
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology)Mohamad, A. B.; Nakajima, Kunihisa; Miwa, Shuhei; Osaka, Masahiko
Journal of Nuclear Science and Technology, 60(3), p.215 - 222, 2023/03
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology)Miwa, Shuhei; Karasawa, Hidetoshi; Nakajima, Kunihisa; Kino, Chiaki*; Suzuki, Eriko; Imoto, Jumpei
JAEA-Data/Code 2021-022, 32 Pages, 2023/01
The improved model for cesium (Cs) chemisorption onto stainless steel (SS) in the fission product (FP) chemistry database named ECUME was incorporated into the severe accident (SA) analysis code SAMPSON for the more accurate estimation of Cs distribution within nuclear reactor vessels in the TEPCO's Fukushima Daiichi Nuclear Power Station (1F). The SAMPSON with the improved model was verified based on the analysis results reproducing the experimental results which were subjected to the modeling of Cs chemisorption behavior. Then, the experiment in the facility with the temperature gradient tube to simulate SA conditions such as temperature decrease and aerosol formation was analyzed to confirm availability of the improved model to the analysis of Cs chemisorption onto SS. The SAMPSON with the improved model successfully reproduced the experimental results, which indicates that the improved model and the analytical method such as setting a method of node-junction, models of aerosol formation and the calculation method of saturated CsOH vapor pressure can be applicable to the analysis of Cs chemisorption behavior. As the information on water-solubility of Cs deposits was also prerequisite to estimate the Cs distribution in the 1F because Cs can be transported through aqueous phase after the SA, the water-solubility of chemisorbed Cs compounds was investigated. The chemisorbed compounds on SS304 have been identified to CsFeO at 873 K to 973 K with higher water-solubility, CsFeSiO
at 873 K to 973 K with higher water-solubility, CsFeSiO at 973 K to 1273 K and Cs
at 973 K to 1273 K and Cs Si
Si O
O at 1073 K to 1273 K with lower water-solubility. From these results, the water-solubility of chemisorbed Cs compounds can be estimated according to the SA analysis conditions such as temperature in the reactor and the CsOH concentration affecting the amount of chemisorbed Cs.
at 1073 K to 1273 K with lower water-solubility. From these results, the water-solubility of chemisorbed Cs compounds can be estimated according to the SA analysis conditions such as temperature in the reactor and the CsOH concentration affecting the amount of chemisorbed Cs.
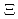
 atoms (J-PARC E03, E07 and future)
atoms (J-PARC E03, E07 and future)Yamamoto, Takeshi; Fujita, Manami; Gogami, Toshiyuki*; Harada, Takeshi*; Hayakawa, Shuhei*; Hosomi, Kenji; Ichikawa, Yudai; Ishikawa, Yuji*; Kamata, K.*; Kanauchi, H.*; et al.
EPJ Web of Conferences, 271, p.03001_1 - 03001_5, 2022/11
Liu, J.; Nakajima, Kunihisa; Miwa, Shuhei; Shirasu, Noriko; Osaka, Masahiko
Journal of Nuclear Science and Technology, 59(4), p.484 - 490, 2022/04
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology)Rizaal, M.; Miwa, Shuhei; Suzuki, Eriko; Imoto, Jumpei; Osaka, Masahiko; Gou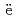 llo, M.*
llo, M.*
ACS Omega (Internet), 6(48), p.32695 - 32708, 2021/12
Times Cited Count:1 Percentile:6.77(Chemistry, Multidisciplinary)Liu, J.; Miwa, Shuhei; Nakajima, Kunihisa; Osaka, Masahiko
Nuclear Materials and Energy (Internet), 26, p.100916_1 - 100916_6, 2021/03
Times Cited Count:2 Percentile:31.78(Nuclear Science & Technology)Miyahara, Naoya; Miwa, Shuhei; Gou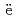 llo, M.*; Imoto, Jumpei; Horiguchi, Naoki; Sato, Isamu*; Osaka, Masahiko
llo, M.*; Imoto, Jumpei; Horiguchi, Naoki; Sato, Isamu*; Osaka, Masahiko
Journal of Nuclear Science and Technology, 57(12), p.1287 - 1296, 2020/12
Times Cited Count:5 Percentile:53.85(Nuclear Science & Technology)In order to clarify the cesium iodide (CsI) transport behavior with a focus on the mechanisms of gaseous iodine formation in the reactor coolant system of LWR under a severe accident condition, a reproductive experiment of CsI transport behavior was conducted using a facility equipped with a thermal gradient tube. Various analyses on deposits and airborne materials during transportation could elucidate two mechanisms for the gaseous iodine formation. One was the gaseous phase chemical reaction in Cs-I-O-H system at relatively high-temperature region, which led to gaseous iodine transport to the lower temperature region without any further changes in gas species due to the kinetics limitation effects. The other one was the chemical reactions related to condensed phase of CsI, namely those of CsI deposits on walls with surface of stainless steel to form Cs CrO
CrO compound and CsI aerosol particles with steam, which were newly found in this study.
compound and CsI aerosol particles with steam, which were newly found in this study.
Miwa, Shuhei; Nakajima, Kunihisa; Miyahara, Naoya; Nishioka, Shunichiro; Suzuki, Eriko; Horiguchi, Naoki; Liu, J.; Miradji, F.; Imoto, Jumpei; Afiqa, B. M.; et al.
Mechanical Engineering Journal (Internet), 7(3), p.19-00537_1 - 19-00537_11, 2020/06
We constructed the fission product (FP) chemistry database named ECUME for LWR severe accident. This version of ECUME is equipped with dataset of the chemical reactions and their kinetics constants for the reactions of cesium(Cs)-iodine(I)-boron(B)-molybdenum(Mo)-oxygen(O)-hydrogen(H) system in gas phase, the elemental model for the high temperature chemical reaction of Cs with stainless steel applied as the structural material in a reactor, and thermodynamic data for CsBO vapor species and solids of Cs
vapor species and solids of Cs Si
Si O
O and CsFeSiO
and CsFeSiO for these chemical reactions. The ECUME will provide estimation of Cs distribution due to the evaluation of effects of interaction with BWR control material B and stainless steel on Cs behavior in the Fukushima Daiichi Nuclear Power Station.
for these chemical reactions. The ECUME will provide estimation of Cs distribution due to the evaluation of effects of interaction with BWR control material B and stainless steel on Cs behavior in the Fukushima Daiichi Nuclear Power Station.
Miwa, Shuhei; Takase, Gaku; Imoto, Jumpei; Nishioka, Shunichiro; Miyahara, Naoya; Osaka, Masahiko
Journal of Nuclear Science and Technology, 57(3), p.291 - 300, 2020/03
Times Cited Count:7 Percentile:55.67(Nuclear Science & Technology)For the evaluation of transport behavior of control material boron in a severe accident of BWR from the viewpoint of chemical effects on cesium and iodine behavior, boron chemistry during transportation in the high temperature region above 400 K was experimentally investigated. The heating tests of boron oxide specimen were conducted using the dedicated experimental apparatus reproducing fission product release and transport in steam atmosphere. Released boron oxide vapor was deposited above 1,000 K by the condensation onto stainless steel. The boron deposits and/or vapors significantly reacted with stainless steel above 1,000 K and formed the stable iron-boron mixed oxide (FeO) BO
BO . These results indicate that released boron from degraded BWR control blade in a severe accident could remain in the high temperature region such as a Reactor Pressure Vessel. Based on these results, it can be said that the existence of boron deposits in the high temperature region would decrease the amount of transported cesium vapors from a Reactor Pressure Vessel due to possible formation of low volatile cesium borate compounds by the reaction of boron deposits with cesium vapors.
. These results indicate that released boron from degraded BWR control blade in a severe accident could remain in the high temperature region such as a Reactor Pressure Vessel. Based on these results, it can be said that the existence of boron deposits in the high temperature region would decrease the amount of transported cesium vapors from a Reactor Pressure Vessel due to possible formation of low volatile cesium borate compounds by the reaction of boron deposits with cesium vapors.
Liu, J.; Miyahara, Naoya; Miwa, Shuhei; Takano, Masahide; Hidaka, Akihide; Osaka, Masahiko
Journal of Nuclear Materials, 527, p.151819_1 - 151819_7, 2019/12
Times Cited Count:2 Percentile:21.58(Materials Science, Multidisciplinary)To evaluate the effect of each constituent element on the evaporation rate of ruthenium (Ru) from fission-produced alloy precipitates, the oxidation and evaporation behaviors of metallic Ru, molybdenum (Mo), palladium (Pd), rhodium (Rh) and Mo-Ru-Pd-Rh alloy powders were investigated by thermogravimetric analysis under oxidizing atmospheres from 1473 to 1723 K. The findings led to the following conclusions: (1) The quick oxidation of Mo into condensed Mo oxides can effectively suppress the oxidation and evaporation of Ru in alloy powders; (2) After the complete evaporation of Mo, the evaporation loss rate of Ru would be directly influenced by the Ru activity in the Ru-Pd-Rh alloys, which is determined by the composition of alloys.
Miwa, Shuhei; Miyahara, Naoya; Nakajima, Kunihisa; Nishioka, Shunichiro; Suzuki, Eriko; Horiguchi, Naoki; Liu, J.; Miradji, F.; Imoto, Jumpei; Afiqa, B. M.; et al.
Proceedings of 27th International Conference on Nuclear Engineering (ICONE-27) (Internet), 8 Pages, 2019/05
We constructed the first version of fission product (FP) chemistry database named ECUME for LWR severe accident. The first version of ECUME is equipped with dataset of the chemical reactions and their kinetics constants for the reactions of cesium(Cs)-iodine(I)-boron(B)-molybdenum(Mo)-oxygen(O)-hydrogen(H) system in gas phase, the elemental model for the high temperature chemical reaction of Cs with stainless steel, and thermodynamic data for CsBO vapor species and solids of Cs
vapor species and solids of Cs Si
Si O
O and CsFeSiO
and CsFeSiO . The ECUME will provide more accurate estimation of Cs distribution due to the evaluation of effects of interaction with BWR control material B and stainless steel on Cs behavior in the Fukushima Daiichi Nuclear Power Station.
. The ECUME will provide more accurate estimation of Cs distribution due to the evaluation of effects of interaction with BWR control material B and stainless steel on Cs behavior in the Fukushima Daiichi Nuclear Power Station.
Mohamad, A.*; Nakajima, Kunihisa; Suzuki, Eriko; Miwa, Shuhei; Osaka, Masahiko; Oishi, Yuji*; Muta, Hiroaki*; Kurosaki, Ken*
Proceedings of International Topical Workshop on Fukushima Decommissioning Research (FDR 2019) (Internet), 4 Pages, 2019/05
In the accident of Fukushima Daiichi Nuclear Power Station, formation of a volatile SrCl could have occurred by the sea-water injection into the core. This can cause the release of non-volatile group Sr from the fuel to induce chemical reactions with reactor structural materials, such as stainless steel and Zircaloy (Zry) cladding. Such reactions could cause the changes in distribution of Sr in the reactor. Chemical reactions between Sr species and Zry were therefore investigated experimentally. As the result, it can be said that Sr vapor species were chemically trapped right after the release from fuel. This trapping effect of Sr by Zry-cladding implies a possibility of preferable Sr retention in the oxide phase of debris.
could have occurred by the sea-water injection into the core. This can cause the release of non-volatile group Sr from the fuel to induce chemical reactions with reactor structural materials, such as stainless steel and Zircaloy (Zry) cladding. Such reactions could cause the changes in distribution of Sr in the reactor. Chemical reactions between Sr species and Zry were therefore investigated experimentally. As the result, it can be said that Sr vapor species were chemically trapped right after the release from fuel. This trapping effect of Sr by Zry-cladding implies a possibility of preferable Sr retention in the oxide phase of debris.
Imoto, Jumpei; Miwa, Shuhei; Osaka, Masahiko
Proceedings of International Topical Workshop on Fukushima Decommissioning Research (FDR 2019) (Internet), 4 Pages, 2019/05
Boron (B) oxidative vaporization processes from the representative alloys of Fe-B and Zr-B formed in the mixed melt of BWR control material boron carbide, stainless steel and Zircaloy were experimentally investigated toward the construction of B release model under severe accident. The results show that B oxidative vaporization from ZrB would proceed in the formation of ZrO
would proceed in the formation of ZrO and B
and B O
O due to the oxidation of ZrB
due to the oxidation of ZrB , followed by the formation of volatile H-B-O vapor species by the reaction of B
, followed by the formation of volatile H-B-O vapor species by the reaction of B O
O with steam. On the other hand, for Fe
with steam. On the other hand, for Fe B and FeB, the B oxidative vaporization processes would proceed in the same manner. Complex Fe-B-O compounds formation in addition to amorphous B
B and FeB, the B oxidative vaporization processes would proceed in the same manner. Complex Fe-B-O compounds formation in addition to amorphous B O
O were observed by the oxidation of Fe
were observed by the oxidation of Fe B and FeB. Then the B vaporization would occur by the formation of volatile H-B-O compound by the reaction of B
B and FeB. Then the B vaporization would occur by the formation of volatile H-B-O compound by the reaction of B O
O , which were derived from both oxidation of Fe
, which were derived from both oxidation of Fe B and decomposition of Fe-B-O compounds.
B and decomposition of Fe-B-O compounds.
Miyahara, Naoya; Miwa, Shuhei; Horiguchi, Naoki; Sato, Isamu*; Osaka, Masahiko
Journal of Nuclear Science and Technology, 56(2), p.228 - 240, 2019/02
Times Cited Count:7 Percentile:61.94(Nuclear Science & Technology)In order to improve LWR source term under severe accident conditions, the first version of a fission product (FP) chemistry database named "ECUME" was developed. The ECUME is intended to include major chemical reactions and their effective kinetic constants for representative SA sequences. It is expected that the ECUME can serve as a fundamental basis from which FP chemical models in the SA analysis codes can be elaborated. The implemented chemical reactions in the first version were those for representative gas species in Cs-I-B-Mo-O-H system. The chemical reaction kinetic constants were evaluated from either literature data or calculated values using ab-initio calculations. The sample chemical reaction calculation using the presently constructed dataset showed meaningful kinetics effects at 1000 K. Comparison of the chemical equilibrium compositions by using the dataset with those by chemical equilibrium calculations has shown rather good consistency for the representative Cs-I-B-Mo-O-H species. From these results, it was concluded that the present dataset should be useful to evaluate FP chemistry in Cs-I-B-Mo-O-H system under LWA SA conditions.
 -based simulated fuel containing CsI
-based simulated fuel containing CsITakamatsu, Yuki*; Ishii, Hiroto*; Oishi, Yuji*; Muta, Hiroaki*; Yamanaka, Shinsuke*; Suzuki, Eriko; Nakajima, Kunihisa; Miwa, Shuhei; Osaka, Masahiko; Kurosaki, Ken*
Nihon Genshiryoku Gakkai Wabun Rombunshi, 17(3/4), p.106 - 110, 2018/12
In order to establish the synthesis method of simulated fuel contacting Cesium (Cs) which is required for the evaluation of physical/chemical characteristics in fuel and release behavior of Cs, sintering tests of the cerium dioxide (CeO ) based simulated fuels containing Cesium iodide (CsI) are performed by using spark plasma sintering (SPS) method. The sintered CeO
) based simulated fuels containing Cesium iodide (CsI) are performed by using spark plasma sintering (SPS) method. The sintered CeO pellets with homogeneous distribution of several micro meter of CsI spherical precipitates were successfully obtained by optimizing SPS conditions.
pellets with homogeneous distribution of several micro meter of CsI spherical precipitates were successfully obtained by optimizing SPS conditions.
Miwa, Shuhei; Ducros, G.*; Hanus, E.*; Bottomley, P. D. W.*; Van Winckel, S.*; Osaka, Masahiko
Nuclear Engineering and Design, 326, p.143 - 149, 2018/01
Times Cited Count:3 Percentile:30.05(Nuclear Science & Technology)The release and transport behaviors of 13 non-gamma-emitting fission products (FPs) and actinides in steam and hydrogen atmospheres were investigated based on the chemical analysis of their deposits on the components of VERCORS test loops. The new findings were obtained; strontium release was significantly enhanced in hydrogen atmosphere and a part of released strontium was transported towards the lower temperature region, uranium release was enhanced in steam atmosphere but most of released uranium deposited at high temperature region.
Di Lemma, F. G.; Yamashita, Shinichiro; Miwa, Shuhei; Nakajima, Kunihisa; Osaka, Masahiko
Energy Procedia, 127, p.29 - 34, 2017/09
Times Cited Count:4 Percentile:90.81(Energy & Fuels)Chemical effects of molybdenum (Mo) and boron (B), which were considered to form compounds with Cs, on the Cs chemisorption were predicted using a chemical equilibrium calculation. It is seen that Cs MoO
MoO were formed in the chemisorbed compounds. On the other hand, little effects were observed for B. The results suggest that the effects of Mo should be considered for further experimental investigation.
were formed in the chemisorbed compounds. On the other hand, little effects were observed for B. The results suggest that the effects of Mo should be considered for further experimental investigation.
Miyahara, Naoya; Miwa, Shuhei; Nakajima, Kunihisa; Osaka, Masahiko
Proceedings of 2017 Water Reactor Fuel Performance Meeting (WRFPM 2017) (USB Flash Drive), 9 Pages, 2017/09
This paper presents the development of a reproductive experimental setup for FP release and transport and an analysis tool considering chemical reaction kinetics for the construction of the FP chemistry database. The performance test of the reproductive experimental setup TeRRa using CsI compounds show that TeRRa can reproduce well a FP chemistry-related behavior such as aerosol formation, growth and deposition behavior. An analytical tool has been developed based on the commercial ANSYS-FLUENT code. Some additional models was added to evaluate detailed FP chemistry during release and transport in this study. A test analysis simulating the CsI heating test in steam atmosphere was carried out to demonstrate the performance of the improved code. The result shows the appropriateness of the additional models.
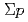 scattering experiment using high intensity pion beams at J-PARC
scattering experiment using high intensity pion beams at J-PARCMiwa, Koji*; Hasegawa, Shoichi; Hayakawa, Shuhei; Hosomi, Kenji; Imai, Kenichi; Kondo, Yasuhiro; Naruki, Megumi; Sako, Hiroyuki; Sato, Susumu; Sugimura, Hitoshi; et al.
JPS Conference Proceedings (Internet), 17, p.041002_1 - 041002_6, 2017/07