Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 O/H
O/H O vapor generator for contrast-variation neutron scattering
O vapor generator for contrast-variation neutron scatteringArima-Osonoi, Hiroshi*; Takata, Shinichi; Kasai, Satoshi*; Ouchi, Keiichi*; Morikawa, Toshiaki*; Miyata, Noboru*; Miyazaki, Tsukasa*; Aoki, Hiroyuki; Iwase, Hiroki*; Hiroi, Kosuke; et al.
Journal of Applied Crystallography, 56(6), p.1802 - 1812, 2023/12
Times Cited Count:0 Percentile:0.02(Chemistry, Multidisciplinary)Shibata, Motoki*; Nakanishi, Yohei*; Abe, Jun*; Arima, Hiroshi*; Iwase, Hiroki*; Shibayama, Mitsuhiro*; Motokawa, Ryuhei; Kumada, Takayuki; Takata, Shinichi; Yamamoto, Katsuhiro*; et al.
Polymer Journal, 55(11), p.1165 - 1170, 2023/11
Times Cited Count:1 Percentile:51.7(Polymer Science)Sanada, Yukihisa; Abe, Tomohisa; Sasaki, Miyuki; Kanno, Marina*; Yamada, Tsutomu*; Nakasone, Takamasa*; Miyazaki, Nobuyuki*; Oshikiri, Keisuke*; Watabe, Hiroshi*
Journal of Nuclear Science and Technology, 10 Pages, 2023/00
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology)The "treated water" from which the main radioactive materials were removed contains tritium, and stakeholders in Japan have been debating how to treat it. The amount of treated water stored in the facility has reached its limit, and the Japanese government has decided that the treatment method will be to discharge the water into the ocean by FY 2023 (FY: Fiscal Year). The present research developed a simple and practical tritium monitor for the Fukushima Daiichi Nuclear Power Station (FDNPS) tritiated water release. A simple and practical tritium monitor was developed for FDNPS tritiated water release based on thin plastic scintillator sheets. The developed devices were calibrated using standard tritium solutions and a method for calculating the minimum detectable activity. Fifteen 0.25 mm-thick scintillators can be placed in the 0.26 L flow cell where the sample water is fed, yielding an active surface area of 3,200 mm . The efficiency of tritiated water with full water is 0.000035 cps Bq
. The efficiency of tritiated water with full water is 0.000035 cps Bq . The minimum detectable activity under simple shielding conditions was 7,800 Bq L
. The minimum detectable activity under simple shielding conditions was 7,800 Bq L (Measurement time was 3,600 s).
(Measurement time was 3,600 s).
Yasuoka, Yumi*; Fujita, Hiroki; Tsuji, Tomoya; Tsujiguchi, Takakiyo*; Sasaki, Michiya*; Miyazaki, Tomoyuki*; Hashima, Shun*; Yasuda, Hiroshi*; Shimada, Kazumasa; Hirota, Seiko*
Hoken Butsuri (Internet), 57(3), p.146 - 155, 2022/12
no abstracts in English
Shimokita, Keisuke*; Yamamoto, Katsuhiro*; Miyata, Noboru*; Arima-Osonoi, Hiroshi*; Nakanishi, Yohei*; Takenaka, Mikihito*; Shibata, Motoki*; Yamada, Norifumi*; Seto, Hideki*; Aoki, Hiroyuki; et al.
Langmuir, 38(41), p.12457 - 12465, 2022/10
Times Cited Count:0 Percentile:0(Chemistry, Multidisciplinary)Miyazaki, Tsukasa*; Miyata, Noboru*; Arima, Hiroshi*; Kira, Hiroshi*; Ouchi, Keiichi*; Kasai, Satoshi*; Tsumura, Yoshihiro*; Aoki, Hiroyuki
Langmuir, 37(32), p.9873 - 9882, 2021/08
Times Cited Count:5 Percentile:38.19(Chemistry, Multidisciplinary)Akutsu, Kazuhiro*; Kira, Hiroshi*; Miyata, Noboru*; Hanashima, Takayasu*; Miyazaki, Tsukasa*; Kasai, Satoshi*; Yamazaki, Dai; Soyama, Kazuhiko; Aoki, Hiroyuki
Polymers (Internet), 12(10), p.2180_1 - 2180_10, 2020/10
Times Cited Count:2 Percentile:7.04(Polymer Science)Arima-Osonoi, Hiroshi*; Miyata, Noboru*; Yoshida, Tessei*; Kasai, Satoshi*; Ouchi, Keiichi*; Zhang, S.*; Miyazaki, Tsukasa*; Aoki, Hiroyuki
Review of Scientific Instruments, 91(10), p.104103_1 - 104103_7, 2020/10
Times Cited Count:9 Percentile:56.16(Instruments & Instrumentation)Miyazaki, Tsukasa*; Miyata, Noboru*; Yoshida, Tessei*; Arima, Hiroshi*; Tsumura, Yoshihiro*; Torikai, Naoya*; Aoki, Hiroyuki; Yamamoto, Katsuhiro*; Kanaya, Toshiji*; Kawaguchi, Daisuke*; et al.
Langmuir, 36(13), p.3415 - 3424, 2020/04
Times Cited Count:14 Percentile:58.64(Chemistry, Multidisciplinary)Kuroki, Ryota; Miyazaki, Hiroshi*; Kato, Takashi
Ketsueki Furonteia, 25(2), p.171 - 180, 2015/02
no abstracts in English
Makihara, Akiko*; Yokose, Tamotsu*; Tsuchiya, Yoshihisa*; Miyazaki, Yoshio*; Abe, Hiroshi; Shindo, Hiroyuki*; Ebihara, Tsukasa*; Maru, Akifumi*; Morikawa, Koichi*; Kuboyama, Satoshi*; et al.
IEEE Transactions on Nuclear Science, 60(1), p.230 - 235, 2013/02
Times Cited Count:6 Percentile:44.02(Engineering, Electrical & Electronic)no abstracts in English
Strasser, P.*; Shimomura, Koichiro*; Koda, Akihiro*; Kawamura, Naritoshi*; Fujimori, Hiroshi*; Makimura, Shunsuke*; Kobayashi, Yasuo*; Nakahara, Kazutaka*; Kato, Mineo*; Takeshita, Soshi*; et al.
Journal of Physics; Conference Series, 225, p.012050_1 - 012050_8, 2010/06
Times Cited Count:12 Percentile:95.21(Physics, Applied) (Si
(Si Ge
Ge )
)
Matsuda, Yasuhiro*; Inami, Toshiya; Owada, Kenji; Murata, Yuto*; Nojiri, Hiroyuki*; Murakami, Yoichi*; Mitsuda, Akihiro*; Wada, Hirofumi*; Miyazaki, Hiroshi*; Harada, Isao*
Journal of the Physical Society of Japan, 77(5), p.054713_1 - 054713_7, 2008/05
Times Cited Count:25 Percentile:74.44(Physics, Multidisciplinary)The magnetic-field-induced valence transition in EuNi (Si
(Si Ge
Ge )
) (x=0.82, 0.85) has been studied by X-ray absorption spectroscopy at low temperatures. The field dependence of the Eu valence is directly observed for the first time. We find a significant decrease in the valences at high magnetic fields above 40 T. The valence of Eu changes from v = 2.75 at a zero field to v = 2.33 at 40 T for x = 0.82. The results are analyzed using a theoretical model. The hybridization parameter V between the Eu
(x=0.82, 0.85) has been studied by X-ray absorption spectroscopy at low temperatures. The field dependence of the Eu valence is directly observed for the first time. We find a significant decrease in the valences at high magnetic fields above 40 T. The valence of Eu changes from v = 2.75 at a zero field to v = 2.33 at 40 T for x = 0.82. The results are analyzed using a theoretical model. The hybridization parameter V between the Eu and Eu
and Eu states and the energy separation
states and the energy separation 
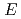 are determined, which are crucial for quantum mechanical mixing.
are determined, which are crucial for quantum mechanical mixing.
 -trifluoromethyl-
-trifluoromethyl- ,
,  -difluorovinyl) terephthalate with excess of various ethers
-difluorovinyl) terephthalate with excess of various ethersEnomoto, Kazuyuki*; Maekawa, Yasunari; Katsumura, Yosuke*; Miyazaki, Toyoaki*; Yoshida, Masaru; Hamana, Hiroshi*; Narita, Tadashi*
Macromolecules, 38(23), p.9584 - 9593, 2005/11
Times Cited Count:9 Percentile:30.73(Polymer Science)Radiation-induced radical polyaddition of bis( -trifluoromethyl-
-trifluoromethyl- ,
, -difluorovinyl) terephthalate [CF
-difluorovinyl) terephthalate [CF =C(CF3)OCOC
=C(CF3)OCOC H
H COOC(CF
COOC(CF )=CF
)=CF ] (BFP) in the presence of an excess of cyclic and acyclic ethers in feed was examined at 0
] (BFP) in the presence of an excess of cyclic and acyclic ethers in feed was examined at 0 C using
C using  -rays irradiation. Irradiation of BFP with an 8-fold excess of 1,4-dioxane (DOX) and diethyl ether (EE) gave monomodal polymers consisting of the
-rays irradiation. Irradiation of BFP with an 8-fold excess of 1,4-dioxane (DOX) and diethyl ether (EE) gave monomodal polymers consisting of the 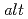 ernating unit of BFP and DOX moieties (poly(BFP-
ernating unit of BFP and DOX moieties (poly(BFP-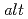 -DOX),
-DOX), 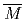
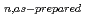 = 5.1
= 5.1 10
10 ) and of BFP and EE (poly(BFP-
) and of BFP and EE (poly(BFP-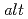 -EE),
-EE), 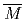
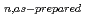 =3.9
=3.9 10
10 ), respectively. The irradiation of BFP with an 8-fold excess of tetrahydrofuran or tetrahydropyran provided only 1:2 addition products in high yields. Mono-functional 2-benzoxypentafluoropropene [CF
), respectively. The irradiation of BFP with an 8-fold excess of tetrahydrofuran or tetrahydropyran provided only 1:2 addition products in high yields. Mono-functional 2-benzoxypentafluoropropene [CF =C(CF
=C(CF )OCOC
)OCOC H
H ] (BPFP) as a model compound showed preferential formation of the 2:1 adduct to the 1:1 adduct with DOX and EE. The kinetic analysis of the model reactions revealed that the rate constants for the formation of the 2:1 adducts (
] (BPFP) as a model compound showed preferential formation of the 2:1 adduct to the 1:1 adduct with DOX and EE. The kinetic analysis of the model reactions revealed that the rate constants for the formation of the 2:1 adducts (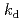 ) of BPFP with DOX and EE were two orders of magnitude higher than those of the 1:1 adducts (
) of BPFP with DOX and EE were two orders of magnitude higher than those of the 1:1 adducts (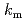 ). Poly(BFP-
). Poly(BFP-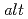 -DOX) showed higher radiation resistance than poly(BFP-
-DOX) showed higher radiation resistance than poly(BFP-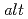 -EE) in solid states as well as in solution. It was concluded that radiation-induced step-growth polymerization under the stoichiometric imbalance of the two compounds requires a high
-EE) in solid states as well as in solution. It was concluded that radiation-induced step-growth polymerization under the stoichiometric imbalance of the two compounds requires a high 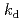 /
/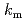 value as well as high radiation resistance for the formation of high molecular weight polymers.
value as well as high radiation resistance for the formation of high molecular weight polymers.
Suzuki, Hiroshi; Miyazaki, Takasato*; Kawamura, Hideki*; Akiniwa, Yoshiaki
no journal, ,
no abstracts in English
Sasada, Yuto*; Miyazaki, Yuji*; Nakano, Motohiro*; Matsuo, Yusuke*; Walker, C.*; Sasamoto, Hiroshi; Mihara, Morihiro
no journal, ,
Cementitous materials are to be used in large quantities in the geological disposal of highly radioactive TRU waste. Thermodynamic data on cement hydrate minerals are important to model the long-term dissolution behavior of cement materials during the reaction of these materials with groundwater. Portlandite (Ca(OH) ) is a major component (20 to 25 wt%) of hydrated Portland cement. Low temperature heat capacity measurements of three types of portlandite (low purity, high purity, and large crystal) showed purity dependence. The large crystal sample followed Debye's T
) is a major component (20 to 25 wt%) of hydrated Portland cement. Low temperature heat capacity measurements of three types of portlandite (low purity, high purity, and large crystal) showed purity dependence. The large crystal sample followed Debye's T rule at low temperatures, while the high and low purity samples showed upturn of heat capacity, possibly due to difference in absorbed water vapor and/or calcite (CaCO
rule at low temperatures, while the high and low purity samples showed upturn of heat capacity, possibly due to difference in absorbed water vapor and/or calcite (CaCO ) contamination. Heat capacities of the cement hydrates (ettringite:Ca
) contamination. Heat capacities of the cement hydrates (ettringite:Ca (Al(OH)
(Al(OH) )
) (SO
(SO )
) (H
(H O)
O) and monosulfate:Ca
and monosulfate:Ca Al
Al (OH)
(OH) (SO
(SO )(H
)(H O)
O) ) exhibited phase transitions associated with the hydration water.
) exhibited phase transitions associated with the hydration water.