Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 , SO
, SO ) precipitates
) precipitatesRai, D.*; Felmy, A. R.*; Moore, D. A.*; Kitamura, Akira; Yoshikawa, Hideki; Doi, Reisuke; Yoshida, Yasushi*
Radiochimica Acta, 102(8), p.711 - 721, 2014/08
Times Cited Count:2 Percentile:15.09(Chemistry, Inorganic & Nuclear)The solubility of Ba(SeO , SO
, SO ) precipitates was determined as a function of the BaSeO
) precipitates was determined as a function of the BaSeO mole fractions, ranging from 0.0015 to 0.3830, and time with an equilibration period extending to as long as 302 days. Equilibrium/steady state conditions in this system are reached in
mole fractions, ranging from 0.0015 to 0.3830, and time with an equilibration period extending to as long as 302 days. Equilibrium/steady state conditions in this system are reached in 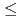 65 days. Pitzer's ion interaction model was used to calculate solid and aqueous phase activity coefficients. Thermodynamic analyses showed that the data do not satisfy Gibbs-Duhem equation, thereby demonstrating that a single-solid solution phase does not control both the selenate and sulfate concentrations. Our extensive data with [Ba], [SeO
65 days. Pitzer's ion interaction model was used to calculate solid and aqueous phase activity coefficients. Thermodynamic analyses showed that the data do not satisfy Gibbs-Duhem equation, thereby demonstrating that a single-solid solution phase does not control both the selenate and sulfate concentrations. Our extensive data with [Ba], [SeO ], and [SO
], and [SO ] can be explained with the formation of an ideal BaSeO
] can be explained with the formation of an ideal BaSeO solid solution phase that controls the selenium concentrations and a slightly disordered/less-crystalline BaSO
solid solution phase that controls the selenium concentrations and a slightly disordered/less-crystalline BaSO (s) that controls the sulfate concentrations. In these experiments the BaSO
(s) that controls the sulfate concentrations. In these experiments the BaSO component of the solid solution phase never reaches thermodynamic equilibrium with the aqueous phase. Thermodynamic interpretations of the data show that both the ideal BaSeO
component of the solid solution phase never reaches thermodynamic equilibrium with the aqueous phase. Thermodynamic interpretations of the data show that both the ideal BaSeO solid solution phase and less-crystalline BaSO
solid solution phase and less-crystalline BaSO (s) phase are in equilibrium with each other in the entire range of BaSeO
(s) phase are in equilibrium with each other in the entire range of BaSeO mole fractions investigated in this study.
mole fractions investigated in this study.
 (cr) in the aqueous Ba
(cr) in the aqueous Ba -SeO
-SeO
 -Na
-Na -H
-H -OH
-OH -H
-H O system; Extending to high selenate concentrations
O system; Extending to high selenate concentrationsRai, D.*; Felmy, A. R.*; Moore, D. A.*; Kitamura, Akira; Yoshikawa, Hideki; Doi, Reisuke; Yoshida, Yasushi*
Radiochimica Acta, 102(9), p.817 - 830, 2014/04
Times Cited Count:2 Percentile:15.09(Chemistry, Inorganic & Nuclear)The aqueous solubility of BaSeO (cr) was studied in Na
(cr) was studied in Na SeO
SeO solutions ranging in concentration from 0.0001 to 4.1 mol.kg
solutions ranging in concentration from 0.0001 to 4.1 mol.kg and maintained in a N
and maintained in a N atmosphere at room temperature (296
atmosphere at room temperature (296  2 K). The studies were conducted from both the undersaturation and oversaturation directions, with equilibration periods ranging from 3 to 596 days. The equilibrium in this system was reached rather rapidly (
2 K). The studies were conducted from both the undersaturation and oversaturation directions, with equilibration periods ranging from 3 to 596 days. The equilibrium in this system was reached rather rapidly (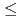 3 days). The SIT and Pitzer's ion-interaction models were used to interpret these data and the predictions based on both of these models agreed closely with the experimental data.
3 days). The SIT and Pitzer's ion-interaction models were used to interpret these data and the predictions based on both of these models agreed closely with the experimental data.
He, R.-H.*; Hashimoto, Makoto*; Karapetyan, H.*; Koralek, J. D.*; Hinton, J. P.*; Testaud, J. P.*; Nathan, V.*; Yoshida, Yoshiyuki*; Yao, H.*; Tanaka, Kiyohisa*; et al.
Science, 331(6024), p.1579 - 1583, 2011/03
Times Cited Count:290 Percentile:98.56(Multidisciplinary Sciences)The nature of the pseudogap phase of cuprate high-temperature superconductors is a major unsolved problem in condensed matter physics. We studied the commencement of the pseudogap state at temperature 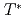 using three different techniques (angle-resolved photoemission spectroscopy, polar Kerr effect, and time-resolved reflectivity) on the same optimally doped Bi2201 crystals. We observed the coincident, abrupt onset at
using three different techniques (angle-resolved photoemission spectroscopy, polar Kerr effect, and time-resolved reflectivity) on the same optimally doped Bi2201 crystals. We observed the coincident, abrupt onset at 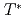 of a particle-hole asymmetric antinodal gap in the electronic spectrum, a Kerr rotation in the reflected light polarization, and a change in the ultrafast relaxational dynamics, consistent with a phase transition. Upon further cooling, spectroscopic signatures of superconductivity begin to grow close to the superconducting transition temperature (
of a particle-hole asymmetric antinodal gap in the electronic spectrum, a Kerr rotation in the reflected light polarization, and a change in the ultrafast relaxational dynamics, consistent with a phase transition. Upon further cooling, spectroscopic signatures of superconductivity begin to grow close to the superconducting transition temperature (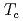 ), entangled in an energy-momentum dependent manner with the preexisting pseudogap features, ushering in a ground state with coexisting orders.
), entangled in an energy-momentum dependent manner with the preexisting pseudogap features, ushering in a ground state with coexisting orders.