Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Inagawa, Jun; Kitatsuji, Yoshihiro; Otobe, Haruyoshi; Nakada, Masami; Takano, Masahide; Akie, Hiroshi; Shimizu, Osamu; Komuro, Michiyasu; Oura, Hirofumi*; Nagai, Isao*; et al.
JAEA-Technology 2021-001, 144 Pages, 2021/08
Plutonium Research Building No.1 (Pu1) was qualified as a facility to decommission, and preparatory operations for decommission were worked by the research groups users and the facility managers of Pu1. The operation of transportation of whole nuclear materials in Pu1 to Back-end Cycle Key Element Research Facility (BECKY) completed at Dec. 2020. In the operation included evaluation of criticality safety for changing permission of the license for use nuclear fuel materials in BECKY, cask of the transportation, the registration request of the cask at the institute, the test transportation, formulation of plan for whole nuclear materials transportation, and the main transportation. This report circumstantially shows all of those process to help prospective decommission.
Kawasaki, Riku*; Sasaki, Yoshihiro*; Nishimura, Tomoki*; Katagiri, Kiyofumi*; Morita, Keiichi*; Sekine, Yurina; Sawada, Shinichi*; Mukai, Sadaatsu*; Akiyoshi, Kazunari*
Advanced Healthcare Materials, 10(9), p.2001988_1 - 2001988_8, 2021/05
Times Cited Count:12 Percentile:58.70(Engineering, Biomedical)Systems for "protein transduction", the intracellular delivery of functional proteins, are needed to address the deliverability challenges of protein therapy, but protein transfer in vivo remains difficult. In this study, we have developed a magnetically induced in vivo protein transfection system using a magnetic nanogel chaperone (MC) composed of iron oxide nanoparticles and polysaccharide nanogels. Experiments using an oral cancer model have shown that this MC system is useful for cancer treatment.
Sasaki, Yuji; Morita, Keisuke; Kitatsuji, Yoshihiro; Ito, Keisuke*; Yoshizuka, Kazuharu*
Solvent Extraction Research and Development, Japan, 28(2), p.121 - 131, 2021/00
Times Cited Count:2 Percentile:9.00(Chemistry, Multidisciplinary)High concentration of Cs is present in high-level radioactive waste. It is well-known that Cs is an alkali element and difficult to extract completely into an organic phase. Crown ether compounds are widely available for Cs extractants; DtBuDB18C6 (di-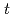 -butyl-dibenzo-18crown6), was used in this study. Organic solvents used for the industrial applications, such as
-butyl-dibenzo-18crown6), was used in this study. Organic solvents used for the industrial applications, such as  -dodecane and 1-octanol, have low solubility concerning the compound; other solvents were employed and tested. In this study, ketone-, ether-, and ester-type solvents showed high solubility for DtBuDB18C6 and DtBuDB18C6, when dissolved in ketones and alcohols, exhibited relatively high Cs distribution ratios (
-dodecane and 1-octanol, have low solubility concerning the compound; other solvents were employed and tested. In this study, ketone-, ether-, and ester-type solvents showed high solubility for DtBuDB18C6 and DtBuDB18C6, when dissolved in ketones and alcohols, exhibited relatively high Cs distribution ratios ( (Cs)), closely to 10.
(Cs)), closely to 10.
Suzuki, Hiroaki*; Morita, Yoshihiro*; Naito, Masanori*; Nemoto, Yoshiyuki; Kaji, Yoshiyuki
Mechanical Engineering Journal (Internet), 7(3), p.19-00450_1 - 19-00450_17, 2020/06
In this study, the SAMPSON code was modified to evaluate severe accidents in a spent fuel pool (SFP). Air oxidation models based on oxidation data obtained on the Zircaroy-4 cladding (ANL model) and the Zircaroy-2 cladding (JAEA model) were included in the modified SAMPSON code. Experiments done by Sandia National Laboratory using simulated fuel assemblies equivalent to those of an actual BWR plant were analyzed by the modified SAMPSON code to confirm the functions for analysis of the severe SFP accidents. The rapid fuel rod temperature rise due to the Zr air oxidation reaction could be reasonably evaluated by the SAMPSON analysis. The SFP accident analyses were conducted with different initial water levels which were no water, water level at bottom of active fuel, and water level at half of active fuel. The present analysis showed that the earliest temperature rise of the fuel rod surface occurred when there was no water in the SFP and natural circulation of air became possible.
Morita, Yoshihiro*; Suzuki, Hiroaki*; Naito, Masanori*; Nemoto, Yoshiyuki; Kaji, Yoshiyuki
Proceedings of 27th International Conference on Nuclear Engineering (ICONE-27) (Internet), 9 Pages, 2019/05
In this study, the SAMPSON code was modified to evaluate severe accidents in a spent fuel pool (SFP). Not only the SFP but also upper spaces of the SFP, walls of the reactor building, and the blowout panel were included. Air oxidation models obtained by the Zircaroy-4 cladding (ANL model) and the Zircaroy-2 cladding (JAEA model) were included in the modified SAMPSON code. Experiments done by Sandia National Laboratory using simulated fuel assemblies equivalent to those of an actual BWR plant were analyzed by the modified SAMPSON code to confirm the functions for analysis of the severe SFP accidents.
Suzuki, Hiroaki*; Morita, Yoshihiro*; Naito, Masanori*; Nemoto, Yoshiyuki; Nagatake, Taku; Kaji, Yoshiyuki
Proceedings of 27th International Conference on Nuclear Engineering (ICONE-27) (Internet), 7 Pages, 2019/05
In this paper, modification of the SAMPSON code was carried out to enable the analysis of spray cooling. The SAMPSON analysis of a spray cooling experiment was performed to confirm reproducibility of spray cooling behavior of fuel claddings. The modified SAMPSON code was applied to a hypothetical loss-of-coolant accident analysis of the SFP. Effectiveness of spray cooling on cladding temperature behavior was investigated. The SAMPSON analysis showed that spraying from the top of the SFP was effective for cooling the fuel assemblies exposed to the gas phase.
Tanaka, Taiki*; Narikiyo, Yoshihiro*; Morita, Kosuke*; Fujita, Kunihiro*; Kaji, Daiya*; Morimoto, Koji*; Yamaki, Sayaka*; Wakabayashi, Yasuo*; Tanaka, Kengo*; Takeyama, Mirei*; et al.
Journal of the Physical Society of Japan, 87(1), p.014201_1 - 014201_9, 2018/01
Times Cited Count:23 Percentile:75.18(Physics, Multidisciplinary)Excitation functions of quasielastic scattering cross sections for the  Ca +
Ca + 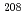 Pb,
Pb, 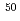 Ti +
Ti + 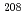 Pb, and
Pb, and  Ca +
Ca +  Cm reactions were successfully measured by using the gas-filled recoil-ion separator GARIS. Fusion barrier distributions were extracted from these data, and compared with the coupled-channels calculations. It was found that the peak energies of the barrier distributions for the
Cm reactions were successfully measured by using the gas-filled recoil-ion separator GARIS. Fusion barrier distributions were extracted from these data, and compared with the coupled-channels calculations. It was found that the peak energies of the barrier distributions for the  Ca +
Ca + 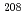 Pb and
Pb and 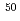 Ti +
Ti + 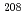 Pb systems coincide with those of the 2n evaporation channel cross sections for the systems, while that of the
Pb systems coincide with those of the 2n evaporation channel cross sections for the systems, while that of the  Ca +
Ca +  Cm is located slightly below the 4n evaporation ones. This results provide us helpful information to predict the optimum beam energy to synthesize superheavy nuclei.
Cm is located slightly below the 4n evaporation ones. This results provide us helpful information to predict the optimum beam energy to synthesize superheavy nuclei.
Sasaki, Yuji; Kitatsuji, Yoshihiro; Sugo, Yumi; Tsubata, Yasuhiro; Suzuki, Tomoya; Kimura, Takaumi; Morita, Yasuji
Proceedings of 20th International Solvent Extraction Conference (ISEC 2014), p.431 - 435, 2014/09
The eight amide extractants including podand-types, which introduce oxygen, nitrogen and sulfur donor atoms in their central frame, are synthesized and their D values for actinide extraction are compared. The central frame affect greatly on their extractability and it is clear that diglycolamide (DGA) has the highest extractability among extractants used here. In addition, DGA having the different length of alkyl chain or phenyl groups attached to amidic N atoms are obtained and the effect of these substituents is studied. Due to the steric hindrance or hydrogen bond, DGA with long and branched alkyl chains, phenyl group, and proton give relatively low D values.
Hasegawa, Noboru; Ochi, Yoshihiro; Kawachi, Tetsuya; Nishikino, Masaharu; Ishino, Masahiko; Imazono, Takashi; Kaihori, Takeshi; Morita, Toshimasa; Sasaki, Akira; Terakawa, Kota*; et al.
X-Ray Lasers 2012; Springer Proceedings in Physics, Vol.147, p.117 - 120, 2014/00
Times Cited Count:0 Percentile:0.00(Engineering, Electrical & Electronic)We have developed the femto-second laser pump and soft X-ray laser probe system in order to observe the dynamical processes of the femto-second laser ablation. By using this system, we succeed to obtain the temporal evolution of the soft X-ray reflectivity from the laser induced Pt surface. The results lead that the rate of decrease in the reflectivity of the probe beam has a non-linear relation with the pump laser fluence.
Sasaki, Yuji; Tsubata, Yasuhiro; Kitatsuji, Yoshihiro; Sugo, Yumi; Shirasu, Noriko; Morita, Yasuji
Proceedings of International Nuclear Fuel Cycle Conference; Nuclear Energy at a Crossroads (GLOBAL 2013) (CD-ROM), p.1079 - 1082, 2013/09
Mutual separation of Am, Cm and lanthanides (Ln) is important to develop the partitioning process of high-level radioactive liquid waste, because the application to their different disposal methods are advantageous. Namely, Am is studied for transmutation due to the reduction of long half-life radionuclides, Cm should be kept in interim storage in order to reduce the calorific value, and Ln should be present in the vitrified radioactive waste toward the geological disposal. However, this mutual separation method is difficult to establish because they have very similar chemical behavior, same oxidation state (III) and similar ionic radii. The development of their mutual separation is termed as the challenging study. In order to obtain the satisfactory results, the property of extractant requires the differentiation of actinide (An) from Ln, high preferability to different ionic radii between Am and Cm, and high extractability to hard acids. Therefore, the extractant have to include both N atom, whose soft donor has high selectivity between An and Ln, and O atoms for the strong extractability to An. The new extractant, NTAamide (N,N,N',N',N'',N''-hexaoctyl-nitrirotriacetamide) is a triamide having N donor at the center of backbone, then NTAamide has hybrid performance of complexation to metals by soft N and three hard amidic O atoms. It is clear that NTAamide can extract trivalent An at diluted HNO with small D(Ln), the separation of An from Ln can be carried out at that condition. The SF of Am/Cm by NTAamide is approximate 1.8, which is not so high to separate each other. The combination of NTAamide of extractant and TEDGA (N,N,N',N'-tetraethyl-diglycolamide) as a masking agent in the aqueous phase shows very high SF(Am/Cm) of maximal 6.5. It is obvious that NTAamide is a promising extractant to achieve the mutual separation among Am/Cm/Ln.
with small D(Ln), the separation of An from Ln can be carried out at that condition. The SF of Am/Cm by NTAamide is approximate 1.8, which is not so high to separate each other. The combination of NTAamide of extractant and TEDGA (N,N,N',N'-tetraethyl-diglycolamide) as a masking agent in the aqueous phase shows very high SF(Am/Cm) of maximal 6.5. It is obvious that NTAamide is a promising extractant to achieve the mutual separation among Am/Cm/Ln.
Sasaki, Yuji; Tsubata, Yasuhiro; Kitatsuji, Yoshihiro; Morita, Yasuji
Chemistry Letters, 42(1), p.91 - 92, 2013/01
Times Cited Count:51 Percentile:77.22(Chemistry, Multidisciplinary)The new N-donor extractant, NTAamide, is synthesized and tested for extraction and separation of Am-Cm-Ln. It was found that the high separation factors for An-Ln can be seen at 0.1-0.2M HNO , which suggest the de-protonation to N donor is important to show the high SF between An and Ln. From the present work, the stable N-donor extractant having high SF of An-Ln from nitric acid to n-dodecane can be obtained. The highest separation factor, 6.5, of Am-Cm for the condition of 0.5M NTAamide (C8) in n-dodecane and 10 mM TEDGA in 0.2M HNO
, which suggest the de-protonation to N donor is important to show the high SF between An and Ln. From the present work, the stable N-donor extractant having high SF of An-Ln from nitric acid to n-dodecane can be obtained. The highest separation factor, 6.5, of Am-Cm for the condition of 0.5M NTAamide (C8) in n-dodecane and 10 mM TEDGA in 0.2M HNO as one of the highest SF values is found. NTAamide is the promising extractants for partitioning process of Am-Cm-Ln.
as one of the highest SF values is found. NTAamide is the promising extractants for partitioning process of Am-Cm-Ln.
Sasaki, Yuji; Tsubata, Yasuhiro; Kitatsuji, Yoshihiro; Sugo, Yumi; Shirasu, Noriko; Morita, Yasuji
Procedia Chemistry, 7, p.380 - 386, 2012/00
Times Cited Count:8 Percentile:91.27(Chemistry, Analytical)Following the nuclear properties, the different disposal methods for Am, Cm and lanthanides in HLW have been investigating, e.g., Am; transmutation, Cm; interim storage and Ln; geological disposal. The mutual separation is an important task. However, these trivalent Ln and An are difficult to separate due to their very similar chemical behavior, same oxidation state and similar ionic radii. We try to use both hydrophilic and lipophilic diamides in an extraction system simultaneously in order to attain the effective mutual separation. In this work, lipophilic DOODA or DGA are used as the extractant and some hydrophilic diamides are employed as the masking agents. The results of mutual separation of Am/Cm/Ln are discussed in this work.
Sasaki, Yuji; Kitatsuji, Yoshihiro; Sugo, Yumi; Tsubata, Yasuhiro; Kimura, Takaumi; Morita, Yasuji
Solvent Extraction Research and Development, Japan, 19, p.51 - 61, 2012/00
Diamides and phosphine oxides having many kinds of the central frames were synthesized and employed in order to see the behavior of actinides(An)(III), (IV), (V) and (VI). Diamides synthesized here have two, three, or four donor atoms of amidic and etheric oxygen (nitrogen or sulfur, instead) atoms in their central frames, namely the extractants can work as bi-, tri-, and tetradentate modes. Di-phosphine di-oxides ((Bis(diphenylphosphoryl)methane (BDPPM) and bis(diphenylphosphoryl)ethylene (BDPPE)), carbamoylmethylphosphine oxide (octylphenyl- ,
, -diisobutyl-carbamoylmethylphosphine oxide (CMPO)) are used as the representative actinide extractants. Metal ions of Eu(III), Th(IV), U(VI), Np(V), Pu(IV) and Am(III) in perchloric acid were extracted into nitrobenzene or n-dodecane. Measurement of
-diisobutyl-carbamoylmethylphosphine oxide (CMPO)) are used as the representative actinide extractants. Metal ions of Eu(III), Th(IV), U(VI), Np(V), Pu(IV) and Am(III) in perchloric acid were extracted into nitrobenzene or n-dodecane. Measurement of  value at the different extractant concentration gives the information on extraction reaction and their extraction ability. From the present work, the best extractant is BDPPM, due to the strong affinity concerning P=O and form of the bidentate mode with six-membered ring chelation. However, this extractant have low solubility in
value at the different extractant concentration gives the information on extraction reaction and their extraction ability. From the present work, the best extractant is BDPPM, due to the strong affinity concerning P=O and form of the bidentate mode with six-membered ring chelation. However, this extractant have low solubility in  -dodecane. Taking the applicatory properties, e.g., high solubility in
-dodecane. Taking the applicatory properties, e.g., high solubility in  -dodecane, easy organic synthesis, gasification by combustion and high actinide extractability, into consideration, diglycolamide (DGA) is a promising extractant for radioactive waste treatment.
-dodecane, easy organic synthesis, gasification by combustion and high actinide extractability, into consideration, diglycolamide (DGA) is a promising extractant for radioactive waste treatment.
 -methyltransferase involved in flower coloration
-methyltransferase involved in flower colorationAkita, Yusuke; Kitamura, Satoshi; Hase, Yoshihiro; Narumi, Issei; Ishizaka, Hiroshi*; Kondo, Emiko*; Kameari, Naoko*; Nakayama, Masayoshi*; Tanikawa, Natsu*; Morita, Yasumasa*; et al.
Planta, 234(6), p.1127 - 1136, 2011/12
Times Cited Count:44 Percentile:75.90(Plant Sciences) -dodecane and HNO
-dodecane and HNO system
systemSasaki, Yuji; Kitatsuji, Yoshihiro; Tsubata, Yasuhiro; Sugo, Yumi; Shirasu, Noriko; Morita, Yasuji
Proceedings of International Conference on Toward and Over the Fukushima Daiichi Accident (GLOBAL 2011) (CD-ROM), 5 Pages, 2011/12
Mutual separation of Am, Cm and lanthanides (Ln) is important to develop the partitioning process of high-level radioactive waste, although this method is difficult to establish due to their very similar chemical behavior, same oxidation state (III) and similar ionic radii. Relative high separation values for these metals can be seen when the multidentate ligands, e.g., diethylene-triamine-N,N,N',N'',N''-pentaacetic acid (DTPA), and softN-donor extractants are employed. We examine to use the combination of two-multidentate ligands having N atoms in their structures in behalf of the high separation of Am, Cm and Ln. Various diamide-type ligands can be synthesized from the initial materials of carboxylic acid and amine, and the aimed materials can be tailored to have high solubility in either HNO orn-dodecane, in case short or long alkyl chains are attached in amidic Natoms. Taking the simultaneous use of these materials into consideration, hydrophilic and lipophilic diamide compounds with different complexing ability for trivalent Ln and actinide (An) are used into both of the aqueous and the organic phases in order to enlarge their separation factors. This technique is applicable in the salt-free system and diamide can be gasificated by combustion, which reduces the secondary-radioactive waste. Two diamide compounds, DGA (diglycolamide) and DOODA (dioxaoctanediamide) as the representative multidentate ligands, have the different features for Ln-complexation, namely DGA has higher D values for middle-Ln than those of light-Ln, and DOODA shows the opposite trend to DGA. TDdDGA (N,N,N',N'-tetradodecyl-diglycolamide) and DOODA(C2) (N,N,N',N'-tetraethyl-dioxaoctanediamide) have high solubility in n-dodecane and HNO
orn-dodecane, in case short or long alkyl chains are attached in amidic Natoms. Taking the simultaneous use of these materials into consideration, hydrophilic and lipophilic diamide compounds with different complexing ability for trivalent Ln and actinide (An) are used into both of the aqueous and the organic phases in order to enlarge their separation factors. This technique is applicable in the salt-free system and diamide can be gasificated by combustion, which reduces the secondary-radioactive waste. Two diamide compounds, DGA (diglycolamide) and DOODA (dioxaoctanediamide) as the representative multidentate ligands, have the different features for Ln-complexation, namely DGA has higher D values for middle-Ln than those of light-Ln, and DOODA shows the opposite trend to DGA. TDdDGA (N,N,N',N'-tetradodecyl-diglycolamide) and DOODA(C2) (N,N,N',N'-tetraethyl-dioxaoctanediamide) have high solubility in n-dodecane and HNO , respectively, and the condition of 0.1 M TDdDGA in n-dodecane and 0.5 MDOODA(C2) in 3 M HNO
, respectively, and the condition of 0.1 M TDdDGA in n-dodecane and 0.5 MDOODA(C2) in 3 M HNO gives the separation factors of La/Gd: 1128 and Am/Cm:3.26.
gives the separation factors of La/Gd: 1128 and Am/Cm:3.26.
 spp., generated by ion-beam irraidation
spp., generated by ion-beam irraidationKondo, Emiko*; Nakayama, Masayoshi*; Kameari, Naoko*; Tanikawa, Natsu*; Morita, Yasumasa*; Akita, Yusuke; Hase, Yoshihiro; Tanaka, Atsushi; Ishizaka, Hiroshi*
JAEA-Review 2010-065, JAEA Takasaki Annual Report 2009, P. 65, 2011/01
no abstracts in English
Sasaki, Yuji; Kitatsuji, Yoshihiro; Tsubata, Yasuhiro; Sugo, Yumi; Morita, Yasuji
Solvent Extraction Research and Development, Japan, 18, p.93 - 101, 2011/00
The mutual separation of Am, Cm and lanthanides (Ln) by the hydrophilic and lipophilic ligands present in both aqueous and organic phases were studied. The hydrophilic ligands used here are DTPA, malonamide, amicacid, TEDGA and DOODA (C2) and four kinds of extractants, TODGA, TDdDGA, DOODA (C8) and DOODA (C12). DOODA can extract light lanthanides with higher D values into the organic phase, on the other hand DGA shows strong complexing ability with middle and heavy lanthanides in the aqueous phase. The high separation factor, over 1000, of La and Gd can be seen in the extraction using extractant, TDdDGA, and DOODA(C2) as the masking agent. The separation factors of Am/Cm by DOODA(C8, C12) and DGA alone extractants are 1.51-1.94, the values with over 3 can be attained by the proposed conditions in this work.
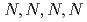 -tetraoctyl-3,6-dioxaoctanediamide and
-tetraoctyl-3,6-dioxaoctanediamide and 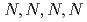 -tetraetyldiglycolamide
-tetraetyldiglycolamideSasaki, Yuji; Morita, Yasuji; Kitatsuji, Yoshihiro; Kimura, Takaumi
Chemistry Letters, 39(8), p.898 - 899, 2010/08
Times Cited Count:26 Percentile:58.39(Chemistry, Multidisciplinary)The separation of Am and Cm from Sm, Eu and Gd was examined by using the combination of new extractant, DOODA (tetraoctyl-3,6-dioxaoctanediamide) and the masking agent, TEDGA (tetraethyl-diglycolamide). These reagents show the neutral donor feature and work in the acidic condition. DOODA can extract light lanthanides with high  values into the organic phase, on the other hand TEDGA shows strong complexing ability with middle and heavy lanthanides in the aqueous phase. Americium and Cm behave as light lanthanides in this extraction system, therefore Am and Cm show high separation from middle lanthanides. The separation factors for
values into the organic phase, on the other hand TEDGA shows strong complexing ability with middle and heavy lanthanides in the aqueous phase. Americium and Cm behave as light lanthanides in this extraction system, therefore Am and Cm show high separation from middle lanthanides. The separation factors for  (Am)/
(Am)/ (Sm),
(Sm),  (Am)/
(Am)/ (Eu), and
(Eu), and  (Am)/
(Am)/ (Gd) are 14.4, 40, and 111, respectively, under the condition of 0.3M DOODA/dodecane and 10 mM TEDGA/ 5M HNO
(Gd) are 14.4, 40, and 111, respectively, under the condition of 0.3M DOODA/dodecane and 10 mM TEDGA/ 5M HNO . The high separation of Am and Cm from these lanthanides, having high neutron cross section, is confirmed in HNO
. The high separation of Am and Cm from these lanthanides, having high neutron cross section, is confirmed in HNO -dodecane system without any pH buffer and metal salts.
-dodecane system without any pH buffer and metal salts.
Morita, Takami*; Niwa, Kentaro*; Fujimoto, Ken*; Kasai, Hiromi*; Yamada, Haruya*; Nishiuchi, Ko*; Sakamoto, Tatsuya*; Godo, Waichiro*; Taino, Seiya*; Hayashi, Yoshihiro*; et al.
Science of the Total Environment, 408(16), p.3443 - 3447, 2010/06
Times Cited Count:17 Percentile:37.12(Environmental Sciences)Iodine-131 ( I) was detected in brown algae collected off the Japanese coast. The maximum measured specific activity of
I) was detected in brown algae collected off the Japanese coast. The maximum measured specific activity of  I in brown algae was 0.37
I in brown algae was 0.37 0.010 Bq/kg-wet. Cesium-137 (
0.010 Bq/kg-wet. Cesium-137 ( Cs) was also detected in all brown algal samples used in this study. There was no correlation between specific activities of
Cs) was also detected in all brown algal samples used in this study. There was no correlation between specific activities of  I and
I and  Cs in these seaweeds. Low specific activity and minimal variability of
Cs in these seaweeds. Low specific activity and minimal variability of  Cs in brown algae indicated that past nuclear weapon tests were the source of
Cs in brown algae indicated that past nuclear weapon tests were the source of  Cs. Although nuclear power facilities are known to be pollution sources of
Cs. Although nuclear power facilities are known to be pollution sources of  I, there was no relationship between the sites where
I, there was no relationship between the sites where  I was detected and the locations of nuclear power facilities. Most of the sites where
I was detected and the locations of nuclear power facilities. Most of the sites where  I was detected were near big cities with large populations. On the basis of the results, we suggest that the likely pollution source of
I was detected were near big cities with large populations. On the basis of the results, we suggest that the likely pollution source of  I, detected in brown seaweeds, is not nuclear power facilities, but nuclear medicine procedures.
I, detected in brown seaweeds, is not nuclear power facilities, but nuclear medicine procedures.
Morita, Yasuji; Sasaki, Yuji; Asakura, Toshihide; Kitatsuji, Yoshihiro; Sugo, Yumi; Kimura, Takaumi
IOP Conference Series; Materials Science and Engineering, 9, p.012057_1 - 012057_11, 2010/05
Times Cited Count:7 Percentile:91.58(Chemistry, Inorganic & Nuclear)The authors have developed a new type of extractant for Am and Cm,  -tetraoctyl-diglycolamide (TODGA), which gives very high distribution ratio at relatively high nitric acid concentration. To apply such a new extractant to the separation process for Am and Cm from HLLW, many criteria should be investigated and satisfied; e.g., separability against fission products (FP), chemical and radiolytic stability, extraction capacity, compatibility with hydrocarbon diluents, and so on. From a viewpoint of extraction capacity, TODGA is modified to
-tetraoctyl-diglycolamide (TODGA), which gives very high distribution ratio at relatively high nitric acid concentration. To apply such a new extractant to the separation process for Am and Cm from HLLW, many criteria should be investigated and satisfied; e.g., separability against fission products (FP), chemical and radiolytic stability, extraction capacity, compatibility with hydrocarbon diluents, and so on. From a viewpoint of extraction capacity, TODGA is modified to  -tetradodecyl-diglycolamide (TDdDGA). Since TDdDGA extract Zr and Pd, effective masking agents for them were examined and selected. With those achievements, a counter-current extraction test with 0.1 M TDdDGA in n-dodecane was carried out using a small-scale mixer-settler and simulated solution of HLLW. As results of the test, a very high recovery of Am, more than 99.96%, was obtained and good separation from FP was observed.
-tetradodecyl-diglycolamide (TDdDGA). Since TDdDGA extract Zr and Pd, effective masking agents for them were examined and selected. With those achievements, a counter-current extraction test with 0.1 M TDdDGA in n-dodecane was carried out using a small-scale mixer-settler and simulated solution of HLLW. As results of the test, a very high recovery of Am, more than 99.96%, was obtained and good separation from FP was observed.