Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
青柳 登; 元川 竜平; 奥村 雅彦; 上田 祐生; 斉藤 拓巳*; 西辻 祥太郎*; 田口 富嗣*; 蓬田 匠; 佐崎 元*; 池田 篤史
Communications Chemistry (Internet), 7, p.128_1 - 128_13, 2024/06
Dissipative structures often appear as an unstable counterpart of ordered structures owing to fluctuations that do not form a homogeneous phase. Even a multiphase mixture may simultaneously undergo one chemical reaction near equilibrium and another one that is far from equilibrium. Here, we observed in real-time crystal seed formation and simultaneous nanocrystal aggregation proceeding from tetravalent cerium complexes to ceria nanoparticles in an acidic aqueous solution and investigated the resultant hierarchical nanoarchitecture. The formed particles exhibited two very different size ranges. The hierarchically assembled structures in solutions were ceria colloids, viz. primary core clusters of crystalline ceria and secondary clusters assembled through surface ions. Such self-assembly is widespread in multi-component complex fluids, paradoxically moderating hierarchical reactions. Stability and instability are not only critical but also complementary for co-optimization around the nearby free energy landscape prior to bifurcation.
熊田 高之; 元川 竜平; 岩瀬 裕希*
Journal of Applied Crystallography, 57(Part 3), p.728 - 733, 2024/06
We found that intensity of small-angle neutron scattering (SANS) from ultrahigh-purity aluminium (5N-Al) was lower than that from aluminium alloys by a factor of 40 in maximum, and thus replaced a 500 mm-diameter aluminium alloy neutron window by the 5N-Al window at SANS-J. We succeeded in decreasing the intensity of background in range of the magnitude of scattering vector Q between 0.02 and 0.2 nm by a factor of 10 in maximum. This result indicates that, the 5N-Al window, which is made thicker to compensate lower tensile strength, generates much lower background than the aluminium alloy windows. The 5N-Al window is suitable for large-diameter neutron window in SANS diffractometers and windows in their environmental apparatus, which cannot be made of single crystalline silicon due to its poor fracture strength, processability, and/or affinity with metallic materials.
by a factor of 10 in maximum. This result indicates that, the 5N-Al window, which is made thicker to compensate lower tensile strength, generates much lower background than the aluminium alloy windows. The 5N-Al window is suitable for large-diameter neutron window in SANS diffractometers and windows in their environmental apparatus, which cannot be made of single crystalline silicon due to its poor fracture strength, processability, and/or affinity with metallic materials.
Guerinoni, E.*; Giusti, F.*; Dourdain, S.*; Dufr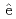 che, J.-F.*; 元川 竜平; 上田 祐生; 青柳 登; Zemb, T.*; Pellet-Rostaing, S.*
che, J.-F.*; 元川 竜平; 上田 祐生; 青柳 登; Zemb, T.*; Pellet-Rostaing, S.*
Journal of Molecular Liquids, 403, p.124820_1 - 124820_11, 2024/06
In uranium production, liquid-liquid extraction using the AMEX (AMine EXtraction) process employs tertiary amines solubilized in an aliphatic diluent. Practical constraints as phase stability problems and co-extraction of competitive elements highlight the need for in-depth investigation and optimization. Modifying gradually the alkyl chain structure of tertiary amines, we investigate here the large variation in extraction performance in terms of Gibbs free energy of transfer using the ienaic decomposition taking into account long range interactions. We show hereby that structuration of the solvent phase can change uranium distribution by 2 orders of magnitude, which is incompatible with standard complexation theory of liquid-liquid extraction. We observe that co-extracted water is required to obtain extraction while extraction is quenched and no pair core can be formed when less than four as effective aggregation number. We conclude that the film term in the ienaic decomposition of the Gibbs energy of transfer, is the one that governs extraction performance. It shows that metal transfer is beyond complexation, and that organization of the solvent phase must be considered to quantitatively interpret the distribution coefficients.
熊田 高之; 中川 洋; 三浦 大輔; 関根 由莉奈; 元川 竜平; 廣井 孝介; 稲村 泰弘; 奥 隆之; 大石 一城*; 森川 利明*; et al.
波紋, 34(2), p.50 - 53, 2024/05
本研究では、スピンコントラスト変調小角散乱法を用いてグルコース濃厚糖溶液を急冷すると氷結晶は平面上になり、その厚みは氷晶生成臨界半径と同程度の数ナノメートルしかないことを明らかにした。本研究は、グルコース分子が氷結晶の特定面に付着してその方向の成長を完全に止めてしまうことを示唆している。
Micheau, C.; 上田 祐生; 元川 竜平; 阿久津 和宏*; 山田 悟史*; 山田 雅子*; Moussaoui, S. A.*; Makombe, E.*; Meyer, D.*; Berthon, L.*; et al.
Journal of Molecular Liquids, 401, p.124372_1 - 124372_12, 2024/05
Supramolecular organization of extractant molecules impacts metal ions separation behavior. Probing bulk and interfacial structures of the relevant systems is expected to provide key insights into the metal ion selectivity and kinetic aspects. The supramolecular features of two solvent extraction systems based on malonamide extractants, N,N,N′,N′-tetrahexylmalonamide (THMA) in toluene and N,N′-dimethyl-N,N′-dibutyl-2-tetradecylmalonamide (DBMA) in n-heptane, were studied using small-angle X-ray scattering for the organic bulk phases, as well as interfacial tension and neutron reflectivity measurements for the interfaces. In the bulk solution, THMA forms dimeric/trimeric associates but no aggregates in toluene, while DBMA forms large aggregates in n-heptane. On the other hand, THMA accumulates in a diffuse layer at the interface at high THMA concentration, whereas DBMA forms a compact but thinner layer. After Pd(II) extraction, the thickness of interfacial layers decreases in the case of THMA, and totally vanishes in the case of DBMA. Based on these new structural information, two mechanisms are proposed for Pd(II) and Nd(III) extraction with malonamides. In toluene, THMA associates slightly accumulate in the vicinity of the interface, then coordinate Pd(II) and diffuse into the organic bulk phase. In n-heptane, DBMA aggregates adsorb at the interface then pick up Nd(III) cations in their polar cores and finally diffuse into the bulk.
熊田 高之; 岩原 大輔*; 西辻 祥太郎*; 阿久津 和宏*; 三浦 大輔; 元川 竜平; 杉田 剛; 鳥飼 直也*; 網野 直也*; 奥 隆之; et al.
Journal of Physical Chemistry C, 128(21), p.8797 - 8802, 2024/05
スピンコントラスト変調中性子反射率法を用いて自動車用ゴムタイヤに用いるブタジエンゴムがシランカップリング剤を通じてシリカ表面とどのように結合しているかを調べた。ブタジエンゴムとシランカップリング剤をシリコン基板にコートした後アニールした試料では、シリコン基板表面にシランカップリング剤とポリブタジエンが7:3の比で絡みあった膜が形成されるのに対し、シランカップリング剤をあらかじめコートした後にポリブタジエンをコートした試料では両者の絡み合いが足りないために容易に剥離してしまうことを突き止めた。
関根 由莉奈; 南川 卓也; 杉田 剛; 永川 栄泰*; 柴山 由樹; 元川 竜平; 深澤 倫子*
Nanoscale, 16(19), p.9400 - 9405, 2024/05
凍結架橋法により強靱なカルボキシメチルセルロースナノファイバー(CMF)/ジルコニウム(Zr)複合ハイドロゲルを開発した。本ハイドロゲルは、Zrを含むHCl溶液を凍結CMFに添加し、それを解凍することによって調製した。作製したハイドロゲルはフッ化物に対して高い吸着性を示した。本研究により、ハイドロゲルと金属の複合体の開発に有用な知見を得た。
 He spin filter and small-angle X-ray scattering
He spin filter and small-angle X-ray scattering領木 研之*; 渡部 史*; 奥平 琢也*; 高橋 慎吾*; 奥 隆之; 廣井 孝介; 元川 竜平; 中村 洋*
Journal of Chemical Physics, 160(11), p.114907_1 - 114907_9, 2024/03
被引用回数:0 パーセンタイル:0.02Small-angle neutron scattering (SANS) and small-angle X-ray scattering (SAXS) measurements were performed for deuterated and non-deuterated poly(ethylene glycol) (d-PEG and h-PEG, respectively) in D O and a D
O and a D O/H
O/H O mixed solvent (Mix) to compere the scattering profiles. To determine the coherent scattering intensity of SANS, a
O mixed solvent (Mix) to compere the scattering profiles. To determine the coherent scattering intensity of SANS, a  He spin filter was utilized. The scattering profiles determined by SANS measurements were analyzed in terms of the wormlike chain model with touched beads along the contour of the chain. However, the SAXS profiles were not explained by the same model with uniform beads but with beads each consisting of a core and a shell having different electron densities. To explore the chain thickness determined form the SANS profile, scattering intensities for different combinations of d-PEG/D
He spin filter was utilized. The scattering profiles determined by SANS measurements were analyzed in terms of the wormlike chain model with touched beads along the contour of the chain. However, the SAXS profiles were not explained by the same model with uniform beads but with beads each consisting of a core and a shell having different electron densities. To explore the chain thickness determined form the SANS profile, scattering intensities for different combinations of d-PEG/D O, d-PEG/Mix, h-PEG/D
O, d-PEG/Mix, h-PEG/D O, and h-PEG/Mix were also examined.
O, and h-PEG/Mix were also examined.
熊田 高之; 元川 竜平; 大場 洋次郎; 中川 洋; 関根 由莉奈; Micheau, C.; 上田 祐生; 杉田 剛; 美留町 厚; 佐々木 未来; et al.
Journal of Applied Crystallography, 56(6), p.1776 - 1783, 2023/12
被引用回数:1 パーセンタイル:56.32(Chemistry, Multidisciplinary)既存のメイン検出器および高位置分解能フォトマル検出器に加えて新規に導入した前面検出器を組み合わせることにより、波数0.002nm から6nm
から6nm にわたる散乱を3つの光学機器配置により切れ目なく測定できるようになった。またグラフィカルユーザーインターフェースの導入と併せて装置のユーザビリティが大幅に向上した。
にわたる散乱を3つの光学機器配置により切れ目なく測定できるようになった。またグラフィカルユーザーインターフェースの導入と併せて装置のユーザビリティが大幅に向上した。
辻 勇人*; 中畑 雅樹*; 菱田 真史*; 瀬戸 秀紀*; 元川 竜平; 井上 大傑*; 江川 泰暢*
Journal of Physical Chemistry Letters (Internet), 14(49), p.11235 - 11241, 2023/12
被引用回数:0 パーセンタイル:0.01(Chemistry, Physical)This work investigates the water-fraction dependence of the aggregation behavior of hydrophobic solutes in water-tetrahydrofuran (THF) and the elucidation of the role of THF using fluorescence microscopy, dynamic light scattering, neutron and X-ray scattering, as well as photoluminescence measurements. Based on the obtained results, the following model is proposed: hydrophobic molecules are molecularly dispersed in the low-water-content region (10-20 vol %), while they form mesoscopic particles upon increasing the water fraction to  30 vol percent. This abrupt change is due to the composition fluctuation of the water-THF binary system to form hydrophobic areas in THF, followed by THF-rich droplets where hydrophobic solutes are incorporated and form loose aggregates. Further increasing the water content prompts the desolvation of THF, which decreases the particle size and generates tight aggregates of solute molecules. This model is consistent with the luminescence behavior of the solutes and will be helpful to control the aggregation state of hydrophobic solutes in various applications.
30 vol percent. This abrupt change is due to the composition fluctuation of the water-THF binary system to form hydrophobic areas in THF, followed by THF-rich droplets where hydrophobic solutes are incorporated and form loose aggregates. Further increasing the water content prompts the desolvation of THF, which decreases the particle size and generates tight aggregates of solute molecules. This model is consistent with the luminescence behavior of the solutes and will be helpful to control the aggregation state of hydrophobic solutes in various applications.
柴田 基樹*; 中西 洋平*; 阿部 淳*; 有馬 寛*; 岩瀬 裕希*; 柴山 充弘*; 元川 竜平; 熊田 高之; 高田 慎一; 山本 勝宏*; et al.
Polymer Journal, 55(11), p.1165 - 1170, 2023/11
被引用回数:1 パーセンタイル:39.87(Polymer Science)Marine ecosystem degradation due to micro plastics is a significant environmental problem, as acknowledged by Sustainable Development Goal 14. Decomposition of plastics using near critical or supercritical water is one of the promising methods to reduce micro plastics. To attain the optimization of the method for improving environmental friendliness, it is necessary to clarify the structural change of materials during the process. We, thus, investigated the decomposition processes of polystyrene particles dispersed in deuterated water (D O) during heating under near critical or supercritical conditions by using in situ small-angle neutron scattering. Under subcritical conditions, the PS particles were swollen by D
O) during heating under near critical or supercritical conditions by using in situ small-angle neutron scattering. Under subcritical conditions, the PS particles were swollen by D O due to increased compatibility with temperature. Near the critical point in subcritical conditions, the cleavage of PS chains in the particles occurred, so that the swollen ratio was much enhanced though the PS particles kept their shapes. In a supercritical condition, the PS particles were degraded into oil including oligomers or monomers and the phase-separated structures with styrene-rich and D
O due to increased compatibility with temperature. Near the critical point in subcritical conditions, the cleavage of PS chains in the particles occurred, so that the swollen ratio was much enhanced though the PS particles kept their shapes. In a supercritical condition, the PS particles were degraded into oil including oligomers or monomers and the phase-separated structures with styrene-rich and D O-rich regions.
O-rich regions.
熊田 高之; 中川 洋; 三浦 大輔; 関根 由莉奈; 元川 竜平; 廣井 孝介; 稲村 泰弘; 奥 隆之; 大石 一城*; 森川 利明*; et al.
Journal of Physical Chemistry Letters (Internet), 14(34), p.7638 - 7643, 2023/08
被引用回数:0 パーセンタイル:0.01(Chemistry, Physical)スピンコントラスト変調中性子小角散乱法を用いて急冷したグルコース濃厚溶液中に生成するナノアイス結晶の構造解析を行ったところ、厚さ1nm程度、半径数十nm以上の平面上の氷晶が生成していることを見出した。本結果はグルコース分子が特定の成長面に吸着することによりその面からの結晶成長を阻害していることを示す。
斉藤 拓巳*; 元川 竜平; 大窪 貴洋*; 三浦 大輔*; 熊田 高之
Environmental Science & Technology, 57(26), p.9802 - 9810, 2023/07
被引用回数:0 パーセンタイル:0.00(Engineering, Environmental)Aggregation of humic acids (HAs) were studied by small-angle neutron and X-ray scattering techniques. The combination of these techniques enables us to examine aggregation structures of heterogeneous HA particles. Two HAs with distinctive compositions were examined; a commercial HA (PAHA) and a HA extracted from deep sedimentary groundwater (HHA). While macroscopic coagulation tests showed that these HAs were stable in solutions except for HHA at pH  6, small-angle neutron scattering (SANS) and X-ray scattering (SAXS) revealed that they formed aggregates with the sizes exceeding sub-micrometer length scale. The SAXS curves of PAHA remarkably varied with pD, whereas the SANS curves did not. With the help of theoretical fittings, it was revealed that PAHA aggregates consisted of two domains: poorly hydrated cores and well-hydrated proton-rich shells. The cores are (dis)aggregated with pD inside the aggregates of the shell. The SANS and SAXS curves of HHA resemble each other, and their intensities at low q increased with a decrease of pD, indicating the formation of homogeneous aggregates. This study revealed that distinctive aggregation behaviors exist in humic substances with heterogeneous structures like PAHA, which is invaluable for their roles in the fate of contaminants or nutrients in aqueous environments.
6, small-angle neutron scattering (SANS) and X-ray scattering (SAXS) revealed that they formed aggregates with the sizes exceeding sub-micrometer length scale. The SAXS curves of PAHA remarkably varied with pD, whereas the SANS curves did not. With the help of theoretical fittings, it was revealed that PAHA aggregates consisted of two domains: poorly hydrated cores and well-hydrated proton-rich shells. The cores are (dis)aggregated with pD inside the aggregates of the shell. The SANS and SAXS curves of HHA resemble each other, and their intensities at low q increased with a decrease of pD, indicating the formation of homogeneous aggregates. This study revealed that distinctive aggregation behaviors exist in humic substances with heterogeneous structures like PAHA, which is invaluable for their roles in the fate of contaminants or nutrients in aqueous environments.
Micheau, C.; 上田 祐生; 元川 竜平; Bauduin, P.*; Girard, L.*; Diat, O.*
Langmuir, 39(31), p.10965 - 10977, 2023/07
被引用回数:0 パーセンタイル:0.00(Chemistry, Multidisciplinary)Understanding clay flotation mechanisms has become a major concern because of the increasing level of environmental contamination of soil and ground water by heavy metals and radionuclides. Clays are often used as sorbents for extracting metals in indirect flotation processes but can function simultaneously as defoamers. However, how foam generation and stability depend on the molecular interactions between the clays and surfactant is still controversial. In the present study, an amine polyethoxylated surfactant was used as a bifunctional surfactant that collected clay particles and acted as a foaming agent in the flotation process. The pH conditions strongly affected the surfactant physicochemical properties, allowing the clay extraction efficiency to be tuned. The interfacial recovery factor of the clays almost reached 100 percent under acidic (pH lower than 6) and neutral (pH 6-10) conditions, whereas it was negative under alkaline conditions (pH higher than 10), contrary to expectations. To elucidate the mechanisms involved in the particle flotation process for each of the pH conditions, the bulk and foam phases were analyzed. The effects of electrostatic interactions between the solutes and multiscale structure on the clay extraction behavior were investigated by electrophoretic measurements, dynamic light scattering, small-angle neutron scattering, and image analysis. Based on these results, three flotation processes were found depending on pH range: surfactant foam fractionation at pH higher than 10; clay particle foam flotation at pH 6-10; and particle froth flotation at pH lower than 6.
大場 洋次郎; 元川 竜平; 金子 耕士; 永井 崇之; 土川 雄介; 篠原 武尚; Parker, J. D.*; 岡本 芳浩
Scientific Reports (Internet), 13, p.10071_1 - 10071_8, 2023/06
被引用回数:1 パーセンタイル:63.33(Multidisciplinary Sciences)The spatial distribution of constituent elements in borosilicate glasses containing simulated high-level radioactive liquid waste (HLLW) was investigated using neutron resonance absorption imaging. The resonance absorption dips of Rh, Pd, Na, Gd, Cs, and Sm were detected in the neutron transmission spectra of the glass samples. The neutron transmission images at the resonance energies of these elements provided the spatial distributions within the glass samples. Rh and Pd led sedimentation, while Gd, Sm, and Cs were uniformly dispersed. These results clearly show the potential of neutron resonance absorption imaging for characterizing borosilicate glasses.
Massey, D.*; Williams, C. D.*; Mu, J.*; Masters, A. J.*; 元川 竜平; 青柳 登; 上田 祐生; Antonio, M. R.*
Journal of Physical Chemistry B, 127(9), p.2052 - 2065, 2023/03
被引用回数:0 パーセンタイル:0.00(Chemistry, Physical)There is an ever-increasing body of evidence that metallic complexes, involving amphiphilic ligands, do not form normal solutions in organic solvents. Instead, they form complex fluids with intricate structures. For example, the metallic complexes may aggregate into clusters and these clusters themselves may aggregate into super-clusters. In order to gain a deeper insight into the mechanisms at play, we have used an improved force field to conduct extensive molecular dynamics simulations of a system composed of zirconium nitrate, water, nitric acid, tri- -butyl phosphate and
-butyl phosphate and  -octane. The important new finding is that a dynamic equilibrium between the cis and trans isomers of the metal complex is likely to play a key role in the aggregation behavior. The isolated cis and trans isomers have similar energies but simulation indicates that the clusters consist predominantly of cis isomers. With increasing metal concentration, we hypothesize that more clustering occurs and the chemical equilibrium shifts towards the cis isomer. It is possible that such isomeric effects play a role in the liquid-liquid extraction of other species and the inclusion of such effects in flowsheet modelling may lead to a better description of the process.
-octane. The important new finding is that a dynamic equilibrium between the cis and trans isomers of the metal complex is likely to play a key role in the aggregation behavior. The isolated cis and trans isomers have similar energies but simulation indicates that the clusters consist predominantly of cis isomers. With increasing metal concentration, we hypothesize that more clustering occurs and the chemical equilibrium shifts towards the cis isomer. It is possible that such isomeric effects play a role in the liquid-liquid extraction of other species and the inclusion of such effects in flowsheet modelling may lead to a better description of the process.
Micheau, C.; 上田 祐生; 阿久津 和宏*; Bourgeois, D.*; 元川 竜平
Solvent Extraction and Ion Exchange, 41(2), p.221 - 240, 2023/02
被引用回数:1 パーセンタイル:39.98(Chemistry, Multidisciplinary)Malonamide derivatives, which are among the most extensively investigated extractants in solvent extraction of lanthanoids, actinides, and platinum group metal ions, were deuterated by using Pd/C and Rh/C catalysts with a D O/2-propanol mixture. This method enables to replace the hydrogen atoms by deuterium atoms in the malonamide chemical structure with a controllable deuteration rate. The maximum rate reaches 75 percent approximately, determined by nuclear magnetic resonance (NMR) and electrospray ionization mass spectrometry. In addition, it has been demonstrated that extraction behavior of the malonamide molecules remains unchanged after the deuteration procedure. Deuterated malonamides should be a powerful tool for fundamental researches on solvent extraction systems, in particular, for structural analysis of the organic phases. The large difference in the neutron scattering cross-section between hydrogen and deuterium leads to a large difference in neutron scattering length density of the malonamide derivatives before and after the deuteration reaction. Therefore, using deuterated malonamides in small-angle neutron scattering and neutron reflectivity studies could give access to the nanoscopic structure of the specific solute species in the bulk organic phase and at the liquid-liquid interface, respectively. This deuteration method could become a general one and be extended to a wide variety of extractant molecules. In this way this work contributes to the development of the fundamental researches in solvent extraction systems.
O/2-propanol mixture. This method enables to replace the hydrogen atoms by deuterium atoms in the malonamide chemical structure with a controllable deuteration rate. The maximum rate reaches 75 percent approximately, determined by nuclear magnetic resonance (NMR) and electrospray ionization mass spectrometry. In addition, it has been demonstrated that extraction behavior of the malonamide molecules remains unchanged after the deuteration procedure. Deuterated malonamides should be a powerful tool for fundamental researches on solvent extraction systems, in particular, for structural analysis of the organic phases. The large difference in the neutron scattering cross-section between hydrogen and deuterium leads to a large difference in neutron scattering length density of the malonamide derivatives before and after the deuteration reaction. Therefore, using deuterated malonamides in small-angle neutron scattering and neutron reflectivity studies could give access to the nanoscopic structure of the specific solute species in the bulk organic phase and at the liquid-liquid interface, respectively. This deuteration method could become a general one and be extended to a wide variety of extractant molecules. In this way this work contributes to the development of the fundamental researches in solvent extraction systems.
小林 大志*; 伏見 朋和*; 水越 寛文*; 元川 竜平; 佐々木 隆之*
Langmuir, 38(48), p.14656 - 14665, 2022/12
被引用回数:2 パーセンタイル:26.09(Chemistry, Multidisciplinary)This study investigates the colloidal behavior of M(IV)(OH) based on its zeta potential and hierarchical structure. The zeta potentials of ZrO
based on its zeta potential and hierarchical structure. The zeta potentials of ZrO and ThO
and ThO in NaCl, NaNO
in NaCl, NaNO , and CaCl
, and CaCl solutions under various pH and ionic strength conditions were measured and elucidated by a surface ionization and complexation model combined with an electric double layer. Based on the assumed similarity of the surface reactions, the model was applied to interpret the zeta potentials of Zr(OH)
solutions under various pH and ionic strength conditions were measured and elucidated by a surface ionization and complexation model combined with an electric double layer. Based on the assumed similarity of the surface reactions, the model was applied to interpret the zeta potentials of Zr(OH) and Th(OH)
and Th(OH) in NaCl, NaNO
in NaCl, NaNO , and CaCl
, and CaCl solutions. The differences in the model parameters were deduced from the different origins of M(IV)(OH)
solutions. The differences in the model parameters were deduced from the different origins of M(IV)(OH) formed by hydrolysis reactions. The size distributions of the selected Zr(OH)
formed by hydrolysis reactions. The size distributions of the selected Zr(OH) suspensions measured by dynamic light scattering techniques suggest that the classical Derjaguin-Landau-Verwey-Overbeek (DLVO) theory governs the colloidal behavior of large aggregates. Additionally, the small angle X-ray scattering measurements elucidated the structure of Zr(OH)
suspensions measured by dynamic light scattering techniques suggest that the classical Derjaguin-Landau-Verwey-Overbeek (DLVO) theory governs the colloidal behavior of large aggregates. Additionally, the small angle X-ray scattering measurements elucidated the structure of Zr(OH) in the nanometer scale. The primary and secondary particle sizes were found to be more dependent on the ionic strength of the electrolyte solutions than on the zeta potentials. The direct interaction of counter ions may affect the formation of primary and secondary particles with comparable size ranges.
in the nanometer scale. The primary and secondary particle sizes were found to be more dependent on the ionic strength of the electrolyte solutions than on the zeta potentials. The direct interaction of counter ions may affect the formation of primary and secondary particles with comparable size ranges.
 microphase separation
microphase separation磯崎 祐嘉*; 東原口 誠也*; 金子 直矢*; 山崎 駿*; 谷口 竜王*; 唐津 孝*; 上田 祐生; 元川 竜平
Chemistry Letters, 51(6), p.625 - 628, 2022/06
被引用回数:2 パーセンタイル:26.09(Chemistry, Multidisciplinary)Polymer photonic crystals (PPCs) that selectively reflect visible light were prepared by triblock polymerization-induced  microphase separation via reversible addition-fragmentation chain transfer polymerization. The addition of homopolymer to the polymerization system swelled a specific phase of the microdomains, enabling the preparation of PPCs with long-wavelength structural colors. Small-angle neutron scattering confirmed the increase in the domain spacing of the lamellar microdomains with the addition of homopolymer, while maintaining their morphology.
microphase separation via reversible addition-fragmentation chain transfer polymerization. The addition of homopolymer to the polymerization system swelled a specific phase of the microdomains, enabling the preparation of PPCs with long-wavelength structural colors. Small-angle neutron scattering confirmed the increase in the domain spacing of the lamellar microdomains with the addition of homopolymer, while maintaining their morphology.
阿久津 和宏*; 山田 悟史*; 上田 祐生; 元川 竜平; 成田 弘一*
Applied Sciences (Internet), 12(3), p.1215_1 - 1215_10, 2022/02
被引用回数:1 パーセンタイル:25.25(Chemistry, Multidisciplinary)液-液系の界面構造を知ることは、二相系の生物・化学反応の理解には必須である。本研究では、界面の層構造を観察できる中性子反射率(NR)測定用の新しいサンプルセルを開発し、典型的な界面活性剤であるドデシル硫酸ナトリウム(SDS)の重トルエン-重水界面への吸着挙動を調べた。開発したセルは、滑らかな界面を作るためにPTFEフレームを底部に配置し、従来のセルに比べて小型化したことが特徴である。得られたNRプロファイルは容易に解析可能であり、重トルエン-重水系と空気-重水系では界面のSDS吸着層構造にわずかな違いがあることがわかった。これは、界面の条件によってSDS分子の吸着挙動が異なることに起因すると考えられる。