Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Fukasawa, Tetsuo*; Suzuki, Akihiro*; Endo, Yoichi*; Inagaki, Yaohiro*; Arima, Tatsumi*; Muroya, Yusa*; Endo, Keita*; Watanabe, Daisuke*; Matsumura, Tatsuro; Ishii, Katsunori; et al.
Journal of Nuclear Science and Technology, 61(3), p.307 - 317, 2024/03
Times Cited Count:2 Percentile:43.92(Nuclear Science & Technology)A flexible waste management system (FWM) is being developed to apply future MA partitioning and transmutation (P&T) technology to current HLLW. This FWM system will store high-level waste (HLLW) in granular form until MA partitioning and transmutation technology is realized. The feasibility of the main process was essentially confirmed by basic experiments and preliminary thermal analysis for granule production by rotary kiln from simulated HLLW and for temporary storage (50 years) of HLW granules at the HLW storage facility, respectively. The granule production experiments revealed that relatively large particles can be produced by the rotary kiln. The results of the thermal analysis showed that the small diameter canisters could be used to safely store the granules at a higher storage density than vitrified HLW. The effectiveness of the FWM system in terms of potential radiotoxicity and repository area was also evaluated, and it was shown that FWM can reduce these factors and has significant advantages in the disposal of HLW generated in current reprocessing plants. Since LWR fuel is stored for a long period of time in Japan and the operation of a reprocessing plant is expected to start soon, the FWM system is considered to be an effective system for reducing the environmental burden of HLW disposal.
Hata, Kuniki; Lin, M.*; Yokoya, Akinari*; Fujii, Kentaro*; Yamashita, Shinichi*; Muroya, Yusa*; Katsumura, Yosuke*
Hoshasen Kagaku (Internet), (103), p.29 - 34, 2017/04
Reactivity of edaravone (3-methyl-1-phenyl-2-pyrazolin-5-one), which is known to show high antioxidative properties, with oxidative species, such as hydroxyl radical (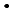 OH) and azide radical (N
OH) and azide radical (N
 ), was investigated by a pulse radiolysis experiment, and generation behavior of edaravone radicals produced through these reactions were observed. It was shown that OH-adducts are produced by the reaction with
), was investigated by a pulse radiolysis experiment, and generation behavior of edaravone radicals produced through these reactions were observed. It was shown that OH-adducts are produced by the reaction with 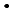 OH in contrast to the other oxidative radicals, which react with edaravone by an electron transfer reaction. Chemical repair properties of edaravone against DNA lesions produced by reactions of DNA with oxidative species were also investigated by a pulse radiolysis experiment with deoxyguanosine monophosphate (dGMP) and a
OH in contrast to the other oxidative radicals, which react with edaravone by an electron transfer reaction. Chemical repair properties of edaravone against DNA lesions produced by reactions of DNA with oxidative species were also investigated by a pulse radiolysis experiment with deoxyguanosine monophosphate (dGMP) and a  -radiolysis experiment with plasmid DNA solutions. It was observed that edaravone reduced dGMP radicals just after produced in a dilute aqueous solution and inhibited some base lesions on plasmid DNA more effectively than single strand breaks. These results show that edaravone may protect living system from oxidative stress, such as radiation, by not only scavenging oxidative species but also reducing precursors of DNA leisons.
-radiolysis experiment with plasmid DNA solutions. It was observed that edaravone reduced dGMP radicals just after produced in a dilute aqueous solution and inhibited some base lesions on plasmid DNA more effectively than single strand breaks. These results show that edaravone may protect living system from oxidative stress, such as radiation, by not only scavenging oxidative species but also reducing precursors of DNA leisons.
Iwamatsu, Kazuhiro*; Muroya, Yusa*; Yamashita, Shinichi*; Kimura, Atsushi; Taguchi, Mitsumasa; Katsumura, Yosuke*
Radiation Physics and Chemistry, 119, p.213 - 217, 2016/02
Times Cited Count:1 Percentile:9.49(Chemistry, Physical)A quick measurement system of a continuous absorption spectrum covering a wide range from 200 to 950 nm was constructed by employing an optical multi-channel detector. Ion beam pulse radiolysis with 12.5 MeV/u He, 18.3 MeV/u C and 17.5 MeV/u Ne ions were performed with the measurement system. Transient absorption spectrum of (SCN)
 was clearly observed in KSCN aqueous solutions within a few minutes in spite of their very small absorbance, demonstrating high sensitivity of 0.001-0.003 in absorbance in the range from 260 to 660 nm as well as short measurement time of a few minutes. Two different absorption peaks attributed to Br
was clearly observed in KSCN aqueous solutions within a few minutes in spite of their very small absorbance, demonstrating high sensitivity of 0.001-0.003 in absorbance in the range from 260 to 660 nm as well as short measurement time of a few minutes. Two different absorption peaks attributed to Br
 and Br
and Br
 were observed simultaneously in NaBr aqueous solutions, showing powerfulness of the measurement system in overviewing chemical kinetics under ion beam irradiation especially in not well investigated chemical systems.
were observed simultaneously in NaBr aqueous solutions, showing powerfulness of the measurement system in overviewing chemical kinetics under ion beam irradiation especially in not well investigated chemical systems.
Yamashita, Shinichi*; Iwamatsu, Kazuhiro; Maehashi, Yuki*; Taguchi, Mitsumasa; Hata, Kuniki; Muroya, Yusa*; Katsumura, Yosuke*
RSC Advances (Internet), 5(33), p.25877 - 25886, 2015/02
Times Cited Count:13 Percentile:37.90(Chemistry, Multidisciplinary)Pulse radiolysis experiments were carried out to observe transient absorptions of reaction intermediates produced in N O
O and Ar-saturated aqueous solutions containing 0.9-900 mM NaBr. The most important species among the reaction intermediates are BrOH
and Ar-saturated aqueous solutions containing 0.9-900 mM NaBr. The most important species among the reaction intermediates are BrOH 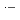 and Br
and Br
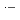 , which commonly have absorption peaks around 360 nm. The experimental results were compared with the results of simulation based on a spur diffusion model. Each of several complicated sequential radiation-induced chemical reactions was carefully considered, optimizing its rate constant within a range of reported values. All the experimental results were able to be universally reproduced by the simulation, assuming a reaction not yet reported, 2BrOH
, which commonly have absorption peaks around 360 nm. The experimental results were compared with the results of simulation based on a spur diffusion model. Each of several complicated sequential radiation-induced chemical reactions was carefully considered, optimizing its rate constant within a range of reported values. All the experimental results were able to be universally reproduced by the simulation, assuming a reaction not yet reported, 2BrOH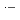
 Br
Br + 2OH
+ 2OH , with a rate constant of 3.8
, with a rate constant of 3.8  10
10 M
M s
s , which is significant only within 10 micro-s for rather high bromide concentrations (
, which is significant only within 10 micro-s for rather high bromide concentrations ( 10 mM). Primary
10 mM). Primary 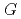 values, which are yields after sufficient diffusion from the spur to the perimeter region during 100 ns, of major water decomposition products, as well as of the reaction intermediates, were calculated for N
values, which are yields after sufficient diffusion from the spur to the perimeter region during 100 ns, of major water decomposition products, as well as of the reaction intermediates, were calculated for N O
O and Ar-saturated conditions as a function of NaBr concentration.
and Ar-saturated conditions as a function of NaBr concentration.
Hata, Kuniki; Urushibara, Ayumi*; Yamashita, Shinichi*; Lin, M.*; Muroya, Yusa*; Shikazono, Naoya; Yokoya, Akinari; Fu, H.*; Katsumura, Yosuke*
Journal of Radiation Research, 56(1), p.59 - 66, 2015/01
Times Cited Count:6 Percentile:27.54(Biology)Lin, M.; Muroya, Yusa*
Genshiryoku, Ryoshi, Kakuyugo Jiten, 4, p.104 - 105, 2014/12
We are invited to write the item "radiation chemistry of aqueous solutions" for the dictionary titled "Nuclear Engineering, Quantum, and nuclear fusion for the beginners" which will be published by Maruzen. Following with an introduction about its importance in various research fields, the history, the reaction mechanism, and the affected factors of water radiolysis are introduced, with easy-to understand explanation about the effects of LET, temperature, etc.
 , CO
, CO
 and Br
and Br in seawater; Model calculation of
in seawater; Model calculation of  radiolysis of seawater
radiolysis of seawaterHata, Kuniki; Hanawa, Satoshi; Kasahara, Shigeki; Muroya, Yusa*; Katsumura, Yosuke*
Proceedings of 2012 Nuclear Plant Chemistry Conference (NPC 2012) (CD-ROM), 6 Pages, 2012/09
Fukushima Daiichi Power Plants experienced seawater injection as emergent measure for a short period after the accident. As a result of seawater injection, structural materials in the reactors are exposed to unexpected corrosion environment due to Cl as well as radiolytic products by
as well as radiolytic products by  -rays from fuels. In the present study, a model calculation on seawater radiolysis was carried out to estimate concentration of radiolytic products under
-rays from fuels. In the present study, a model calculation on seawater radiolysis was carried out to estimate concentration of radiolytic products under  -irradiation. Radiolytic products, H
-irradiation. Radiolytic products, H , O
, O and H
and H O
O , in seawater monotonically increased with dose. H
, in seawater monotonically increased with dose. H production was especially distinguished, and its yield was 4.4
production was especially distinguished, and its yield was 4.4  10
10 mol J
mol J . As similar result was obtained from the calculation of NaBr solution, the main species which increased these products in seawater was thought to be Br
. As similar result was obtained from the calculation of NaBr solution, the main species which increased these products in seawater was thought to be Br . On the other hand, Cl
. On the other hand, Cl and HCO
and HCO
 in seawater hardly affected the concentrations of these radiolytic products.
in seawater hardly affected the concentrations of these radiolytic products.
Yamashita, Shinichi; Baldacchino, G.*; Maeyama, Takuya*; Taguchi, Mitsumasa; Muroya, Yusa*; Lin, M.*; Kimura, Atsushi; Murakami, Takeshi*; Katsumura, Yosuke
Free Radical Research, 46(7), p.861 - 871, 2012/07
Times Cited Count:26 Percentile:54.24(Biochemistry & Molecular Biology)Radiation-induced reactions in aqueous solutions of a water-soluble coumarin derivative, coumarin-3-carboxyl acid (C3CA), have been investigated by pulse radiolysis with 35-MeV electron beam, final product analysis after  Co
Co 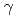 -irradiations, and deterministic model simulations. It was found that C3CA reacts with the hydroxyl radical (
-irradiations, and deterministic model simulations. It was found that C3CA reacts with the hydroxyl radical ( OH) as well as the hydrated electron at nearly diffusion-controlled rate constants: 6.8
OH) as well as the hydrated electron at nearly diffusion-controlled rate constants: 6.8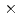 10
10 and 2.1
and 2.1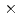 10
10 M
M s
s , respectively. Reactivity of C3CA toward O
, respectively. Reactivity of C3CA toward O

 was not confirmed. Production of a fluorescent molecule 7-hydroxy-coumarin-3-carboxylic acid (7OH-C3CA) was detected by a fluorescence spectrometer coupled with high performance liquid chromatography. Production yields of 7OH-C3CA were in a range from 0.025 to 0.18 (100 eV)
was not confirmed. Production of a fluorescent molecule 7-hydroxy-coumarin-3-carboxylic acid (7OH-C3CA) was detected by a fluorescence spectrometer coupled with high performance liquid chromatography. Production yields of 7OH-C3CA were in a range from 0.025 to 0.18 (100 eV) , depending on irradiation conditions. A variety of the yield with saturating gas, additive, and C3CA concentration implied that there are at least two pathways from scavenging reaction of C3CA toward
, depending on irradiation conditions. A variety of the yield with saturating gas, additive, and C3CA concentration implied that there are at least two pathways from scavenging reaction of C3CA toward  OH to 7OH-C3CA: peroxidation reaction followed by elimination of perhydroxyl radical and disproportionation reaction. A reaction mechanism involving the two pathways was proposed and incorporated into the simulations, showing good explanation of experimentally measured 7OH-C3CA yields with a constant conversion factor from
OH to 7OH-C3CA: peroxidation reaction followed by elimination of perhydroxyl radical and disproportionation reaction. A reaction mechanism involving the two pathways was proposed and incorporated into the simulations, showing good explanation of experimentally measured 7OH-C3CA yields with a constant conversion factor from  OH scavenging to 7OH-C3CA production, 4.7%, unless
OH scavenging to 7OH-C3CA production, 4.7%, unless 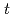 -BuOH is not added.
-BuOH is not added.
Wan, L. K.*; Peng, J.*; Lin, M.; Muroya, Yusa*; Katsumura, Yosuke*; Fu, H. Y.*
Radiation Physics and Chemistry, 81(5), p.524 - 530, 2012/05
Times Cited Count:20 Percentile:77.60(Chemistry, Physical)Rate constants of common crown ethers ((C H
H O)
O) , n=4, 5, 6) and their model compound, 1,4-dioxane(6C2) with some important oxidative radicals,
, n=4, 5, 6) and their model compound, 1,4-dioxane(6C2) with some important oxidative radicals,  OH, SO
OH, SO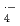 , and NO
, and NO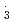 were determined in various aqueous solutions using pulse radiolysis and laser photolysis techniques. The rate constants for 6C2 and crown ethers with
were determined in various aqueous solutions using pulse radiolysis and laser photolysis techniques. The rate constants for 6C2 and crown ethers with  OH and SO
OH and SO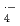 increase with the number of H-atom in the ethers, indicating that H-abstraction is dominant reaction between crown ethers and these two radicals. The presence of cations in solution has negligible effect on the rate constants for
increase with the number of H-atom in the ethers, indicating that H-abstraction is dominant reaction between crown ethers and these two radicals. The presence of cations in solution has negligible effect on the rate constants for  OH and SO
OH and SO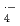 . However, for NO
. However, for NO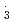 , the rate constants is not in proportional to H-atom number in crown ethers, and the 12-crown-4 is most reactive compared with the others. For the studied crown ethers, the rate constants of these oxidative radicals have the order:
, the rate constants is not in proportional to H-atom number in crown ethers, and the 12-crown-4 is most reactive compared with the others. For the studied crown ethers, the rate constants of these oxidative radicals have the order:  OH
OH  SO
SO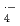
 NO
NO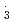 . This is the first report on the kinetic behavior of crown ethers with NO
. This is the first report on the kinetic behavior of crown ethers with NO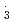 , and it would be helpful for the understanding of stability of crown ethers in the nuclear fuel reprocessing.
, and it would be helpful for the understanding of stability of crown ethers in the nuclear fuel reprocessing.
Lin, M.; Katsumura, Yosuke*; Muroya, Yusa*
Hoshasen Kagaku (Internet), (93), p.3 - 13, 2012/03
The state-of-the-art in the field of water radiolysis at elevated temperatures and supercritical water are reviewed, with the emphasis on the temperature and density effects on the radiolytic yields of water decomposition products, the reaction rate constants and the spectral properties of hydrated electron. The latest studies on supercritical water by picosecond pulse radiolysis as well as some main results about pulse radiolysis of alcohols at elevated temperatures are also introduced.
Fu, H. Y.*; Lin, M.; Muroya, Yusa*; Katsumura, Yosuke*
Research on Chemical Intermediates, 38(1), p.135 - 145, 2012/01
Times Cited Count:2 Percentile:2.65(Chemistry, Multidisciplinary)In the present work, pulse radiolysis was used to demonstrate the oxidizing nature of various intermediates of  -AcMetNH
-AcMetNH and
and  -AcMetOMe via the oxidation of tryptophan (Trp) in a free aqueous solution. The reaction was examined at different pH levels. Electron transfer processes involving Trp and the sulfur-containing three-electron-bonded intermediates were investigated. The results revealed that Trp-Met pairs can form very favorable redox couples in biological systems, which is consistent with earlier reports of more efficient and rapid intramolecular charge transfers in synthetic as well as natural peptides containing methionine (Met).
-AcMetOMe via the oxidation of tryptophan (Trp) in a free aqueous solution. The reaction was examined at different pH levels. Electron transfer processes involving Trp and the sulfur-containing three-electron-bonded intermediates were investigated. The results revealed that Trp-Met pairs can form very favorable redox couples in biological systems, which is consistent with earlier reports of more efficient and rapid intramolecular charge transfers in synthetic as well as natural peptides containing methionine (Met).
Oka, Toshitaka; Yamashita, Shinichi; Midorikawa, Masamichi*; Saiki, Seiichi; Muroya, Yusa*; Kamibayashi, Masato*; Yamashita, Masayuki*; Anzai, Kazunori*; Katsumura, Yosuke*
Analytical Chemistry, 83(24), p.9600 - 9604, 2011/10
Times Cited Count:42 Percentile:78.78(Chemistry, Analytical)Chemical reactions of a novel gauche-type spin trap, G-CYPMPO (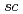 -5-(5,5-dimethyl-2-oxo-1,3,2-dioxaphosphinan-2-yl)-5-methy-1-pyrroline 1-oxide, torsion angle: O1-P1-C6-N1 of 52.8
-5-(5,5-dimethyl-2-oxo-1,3,2-dioxaphosphinan-2-yl)-5-methy-1-pyrroline 1-oxide, torsion angle: O1-P1-C6-N1 of 52.8 ), with reactive oxygen species were examined by pulse radiolysis technique with 35 MeV electron beam and electron spin resonance spectroscopy after
), with reactive oxygen species were examined by pulse radiolysis technique with 35 MeV electron beam and electron spin resonance spectroscopy after  Co
Co  -ray irradiation. The spin trapping reaction rate constants of G-CYPMPO toward hydroxyl radical and hydrated electron were estimated to be (4.2
-ray irradiation. The spin trapping reaction rate constants of G-CYPMPO toward hydroxyl radical and hydrated electron were estimated to be (4.2 0.1)
0.1) 10
10 and (11.8
and (11.8 0.2)
0.2) 10
10 M
M s
s , respectively. Half-lives of spin adducts, hydroxyl radical and perhydroxyl radical adducted G-CYPMPO were estimated to be
, respectively. Half-lives of spin adducts, hydroxyl radical and perhydroxyl radical adducted G-CYPMPO were estimated to be  35 and
35 and  90 min, respectively. A comparison of the results with earlier reports using different radical sources suggests that the purity of the solution and/or the radical generation technique may influence the stability of spin adducts.
90 min, respectively. A comparison of the results with earlier reports using different radical sources suggests that the purity of the solution and/or the radical generation technique may influence the stability of spin adducts.
Fu, H. Y.*; Lin, M.; Katsumura, Yosuke*; Muroya, Yusa*
International Journal of Chemical Kinetics, 43(10), p.590 - 597, 2011/10
Times Cited Count:3 Percentile:5.87(Chemistry, Physical)Free radical scavenging and antioxidant activities of four natural flavonoids, namely silybin, naringenin, naringin and hesperetin, have been studied using nanosecond pulse radiolysis techniques. The kinetics and mechanisms of the reactions of silybin and analogues with various oxidizing radicals have been investigated. Further, the transient species has been assigned and radical scavenging rate constants have also been measured. Moreover, the structure-activity relationships (SAR) between chemical structures of the flavonoids and their radical scavenging activities are further analyzed by theoretical calculation. Combined our previous observation of the fast reparation of DNA damage and efficient DNA protection against radiation damage in vitro, it can be confirmed that test flavonoids are promising molecules to be used for their potential antioxidant properties.
 radical studied by picosecond pulse radiolysis
radical studied by picosecond pulse radiolysisEl Omar, A. K.*; Schmidhammer, U.*; Jeunesse, P.*; Labre, J. P.*; Lin, M.; Muroya, Yusa*; Katsumura, Yosuke*; Pernot, P.*; Mostafavi, M.*
Journal of Physical Chemistry A, 115(44), p.12212 - 12216, 2011/10
Times Cited Count:45 Percentile:82.46(Chemistry, Physical)Picosecond pulse radiolysis measurements using a pulse-probe method are performed to measure directly the time-dependent radiolytic yield of the OH radical in pure water. The time-dependent absorbance of OH
radical in pure water. The time-dependent absorbance of OH radical at 263 nm is deduced from the observed signal by substracting the contribution of the hydated electron and that of the irradiated empty fused silica cell which presents also a transient absoption. The time-dependent radiolytic yield of OH
radical at 263 nm is deduced from the observed signal by substracting the contribution of the hydated electron and that of the irradiated empty fused silica cell which presents also a transient absoption. The time-dependent radiolytic yield of OH is obtained by assuming the yield of the hydrated electron at 20 ps equal to 4.2
is obtained by assuming the yield of the hydrated electron at 20 ps equal to 4.2 10
10 mol/J. The value of the yield of OH
mol/J. The value of the yield of OH radical at 20 ps is found to be (4.80
radical at 20 ps is found to be (4.80 0.12)
0.12) 10
10 mol/J.
mol/J.
Shi, W.-Q.*; Fu, H.-Y.*; Bounds, P. L.*; Muroya, Yusa*; Lin, M.; Katsumura, Yosuke*; Zhao, Y.-L.*; Chai, Z.-F.*
Radiation Research, 176(1), p.128 - 133, 2011/07
Times Cited Count:3 Percentile:16.51(Biology)3-Nitrotyrosine (3-NT) has been reported as an important biomarker of oxidative stress and potential source of reactive oxygen species (ROSs). In this work, the UV-visible absorption spectra of the transients formed by hydrated electron (e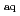
 ) reacting with 3-NT and its derivatives were investigated, the spectra showed many characteristics of aromatic nitro anion radical. The reaction rate constants of e
) reacting with 3-NT and its derivatives were investigated, the spectra showed many characteristics of aromatic nitro anion radical. The reaction rate constants of e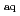
 reacting with 3-NT, N-acetyl-3-nitrotyrosine ethyl ester (NANTE) and nitrotyrosine-containing peptide Gly-nitroTyr-Gly at neutral pH were determined, respectively, which were almost two orders of magnitude higher than that of tyrosine and tyrosine-containing peptides. The pH-dependence of e
reacting with 3-NT, N-acetyl-3-nitrotyrosine ethyl ester (NANTE) and nitrotyrosine-containing peptide Gly-nitroTyr-Gly at neutral pH were determined, respectively, which were almost two orders of magnitude higher than that of tyrosine and tyrosine-containing peptides. The pH-dependence of e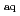
 decay rate constants in presence of 3-NT was also studied.
decay rate constants in presence of 3-NT was also studied.
 C; A Monte-Carlo simulation study
C; A Monte-Carlo simulation studySanguanmith, S.*; Muroya, Yusa*; Tippayamontri, T.*; Meesungnoen, J.*; Lin, M.; Katsumura, Yosuke*; Jay-Gerin, J.-P.*
Physical Chemistry Chemical Physics, 13(22), p.10690 - 10698, 2011/06
Times Cited Count:22 Percentile:59.55(Chemistry, Physical)Monte-Carlo simulations of the radiolysis of the ferrous sulfate (Fricke) dosimeter with low-linear energy transfer (LET) radiation have been performed as a function of temperature from 25 to 350 C. The predicted yields of Fe
C. The predicted yields of Fe oxidation are found to increase with increasing temperature up to 100-150
oxidation are found to increase with increasing temperature up to 100-150 C, and then tend to remain essentially constant at higher temperatures, in very good agreement with experiment. Moreover, the observed dependence of G(Fe
C, and then tend to remain essentially constant at higher temperatures, in very good agreement with experiment. Moreover, the observed dependence of G(Fe ) on temperature largely reflects the influence of temperature upon the primary free-radical product yields of the radiolysis, especially the yield of H atoms. Above 200-250
) on temperature largely reflects the influence of temperature upon the primary free-radical product yields of the radiolysis, especially the yield of H atoms. Above 200-250 C, the more and more pronounced intervention of the reaction of H atoms with water also contributes to the variation of G(Fe
C, the more and more pronounced intervention of the reaction of H atoms with water also contributes to the variation of G(Fe ), which may decrease or increase slightly, depending on the choice made for the rate constant of this reaction.
), which may decrease or increase slightly, depending on the choice made for the rate constant of this reaction.
 C; Monte-Carlo simulations
C; Monte-Carlo simulationsSanguanmith, S.*; Muroya, Yusa*; Meesungnoen, J.*; Lin, M.; Katsumura, Yosuke*; Mirsaleh Kohan, L.*; Guzonas, D. A.*; Stuart, C. R.*; Jay-Gerin, J.-P.*
Chemical Physics Letters, 508(4-6), p.224 - 230, 2011/05
Times Cited Count:42 Percentile:80.73(Chemistry, Physical)Our Monte-Carlo modeling of the high-temperature radiolysis of water by low-LET radiation was re-examined in an attempt to reconcile our computed 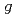 -values of the various radiolytic products with recently re-assessed experimental data up to 350
-values of the various radiolytic products with recently re-assessed experimental data up to 350 C. The inclusion in our simulations of the abrupt drop in the rate constant for the self-reaction of hydrated electron above 150
C. The inclusion in our simulations of the abrupt drop in the rate constant for the self-reaction of hydrated electron above 150 C led us to re-evaluate the temperature dependence of certain parameters intervening in the physicochemical stage of the radiolysis. A very good agreement was found between model and experiment. The importance of the reaction of H atoms with water in the unexplained yield of H
C led us to re-evaluate the temperature dependence of certain parameters intervening in the physicochemical stage of the radiolysis. A very good agreement was found between model and experiment. The importance of the reaction of H atoms with water in the unexplained yield of H above 200
above 200 C was also briefly discussed.
C was also briefly discussed.
 , Br
, Br
 , and Br
, and Br
 in aqueous solutions
in aqueous solutionsLin, M.; Archirel, P.*; Van-Oanh, N. T.*; Muroya, Yusa*; Fu, H.*; Yan, Y.*; Nagaishi, Ryuji; Kumagai, Yuta; Katsumura, Yosuke*; Mostafavi, M.*
Journal of Physical Chemistry A, 115(17), p.4241 - 4247, 2011/04
Times Cited Count:16 Percentile:47.65(Chemistry, Physical)The absorption spectra of Br
 and Br
and Br
 in aqueous solutions are investigated by pulse radiolysis techniques from room temperature to 380 and 350
in aqueous solutions are investigated by pulse radiolysis techniques from room temperature to 380 and 350 C, respectively. The weak temperature effect on the absorption spectra of Br
C, respectively. The weak temperature effect on the absorption spectra of Br
 and Br
and Br
 is because in these two systems, the transition occurs between two valence states. We performed classical dynamics of hydrated Br
is because in these two systems, the transition occurs between two valence states. We performed classical dynamics of hydrated Br system at 20 and 300
system at 20 and 300 C under pressure of 25 MPa. This shows that the first water shell is strongly bound to the anion whatever the temperature. The first two water shells form a cavity of a roughly spherical shape around the anion. By TDDFT method, we calculated the absorption spectra of hydrated Br
C under pressure of 25 MPa. This shows that the first water shell is strongly bound to the anion whatever the temperature. The first two water shells form a cavity of a roughly spherical shape around the anion. By TDDFT method, we calculated the absorption spectra of hydrated Br at two temperatures and we compared the results with the experimental data.
at two temperatures and we compared the results with the experimental data.
 C
CSanguanmith, S.*; Muroya, Yusa*; Meesungnoen, J.*; Lin, M.; Katsumura, Yosuke*; Mirsaleh Kohan, L.*; Guzonas, D. A.*; Stuart, C. R.*; Jay-Gerin, J.-P.*
Proceedings of 5th International Symposium on Supercritical Water-Cooled Reactors (ISSCWR-5) (CD-ROM), 13 Pages, 2011/03
A re-examination of our Monte-Carlo modeling of the high-temperature radiolysis of liquid water by low linear energy transfer (LET  0.3 keV/
0.3 keV/ m) radiation was undertaken in an attempt to reconcile our computed g-values of the various radiolytic products with newly measured or recently re-assessed experimental data over the range from 25 up to 350
m) radiation was undertaken in an attempt to reconcile our computed g-values of the various radiolytic products with newly measured or recently re-assessed experimental data over the range from 25 up to 350 C. Using a global-fit procedure, experimental data were found to be best reproduced when a discontinuity in the temperature dependence of certain physicochemical parameters was introduced at
C. Using a global-fit procedure, experimental data were found to be best reproduced when a discontinuity in the temperature dependence of certain physicochemical parameters was introduced at  100-150
100-150 C. The presence of such a discontinuity was hypothesized to be associated with a change in the liquid structure of water around these temperatures. In addition to the physicochemical factors intervening in the radiolysis, the importance of the reaction of H atoms with water in contributing to the unexplained yield of H
C. The presence of such a discontinuity was hypothesized to be associated with a change in the liquid structure of water around these temperatures. In addition to the physicochemical factors intervening in the radiolysis, the importance of the reaction of H atoms with water in contributing to the unexplained yield of H above 200
above 200 C was also investigated.
C was also investigated.
Hata, Kuniki; Lin, M.; Katsumura, Yosuke*; Muroya, Yusa*; Fu, H. Y.*; Yamashita, Shinichi; Nakagawa, Hidehiko*
Journal of Radiation Research, 52(1), p.15 - 23, 2011/01
Times Cited Count:24 Percentile:63.19(Biology)