Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Hata, Kuniki; Lin, M.; Katsumura, Yosuke*; Muroya, Yusa*; Fu, H. Y.*; Yamashita, Shinichi; Nakagawa, Hidehiko*
Journal of Radiation Research, 52(1), p.15 - 23, 2011/01
Times Cited Count:21 Percentile:60.99(Biology)Kitano, Akihiro; Teruyama, Hidehiko; Nishi, Hiroshi; Yamaoka, Mitsuaki*; Moriki, Yasuyuki*; Nakagawa, Masatoshi*
Saikuru Kiko Giho, (15), p.1 - 16, 2002/06
None
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Fu, H.*; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
Edaravone, 3-methyl-1-phenyl-2-pyrazolin-5-one, is a newly developed free radical scavenger that has been approved in Japan as a neuroprotective drug since 2001. In the present work, the transient intermediates and the reactivity of edaravone towards  OH, N
OH, N
 , Br
, Br
 , SO
, SO
 , and CCl
, and CCl O
O
 are investigated by pulse radiolysis techniques. The oxidation of edaravone by N
are investigated by pulse radiolysis techniques. The oxidation of edaravone by N
 , Br
, Br
 , SO
, SO
 , and CCl
, and CCl O
O
 results in an absorption band with
results in an absorption band with 
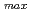 = 345 nm (
= 345 nm (
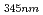 = 2600 M
= 2600 M cm
cm ), which is assigned to the edaravone radical formed by H-abstraction or electron transfer. However, the transient species produced by the reaction of edaravone with
), which is assigned to the edaravone radical formed by H-abstraction or electron transfer. However, the transient species produced by the reaction of edaravone with  OH radical shows an absorption band with
OH radical shows an absorption band with 
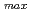 = 320 nm (
= 320 nm (
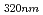 = 4900 M
= 4900 M cm
cm ). Accordingly, the main transient species by the reaction of edaravone with
). Accordingly, the main transient species by the reaction of edaravone with  OH radical in the absence of O
OH radical in the absence of O is attributed to OH-adducts. The rate constants of edaravone reacting with
is attributed to OH-adducts. The rate constants of edaravone reacting with  OH, N
OH, N
 , SO
, SO
 , and CCl
, and CCl O
O
 are estimated to be 8.5
are estimated to be 8.5 10
10 , 5.4
, 5.4 10
10 , 6.0
, 6.0 10
10 , and 5.1
, and 5.1 10
10 M
M s
s , respectively.
, respectively.
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Fu, H.*; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
no abstracts in English
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Fu, H.*; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Fu, H.*; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
The reactivity of edaravone with some oxidative radicals ( OH, N
OH, N
 , Br
, Br
 , SO
, SO
 , CClO
, CClO O
O
 ) has been measured by pulse radiolysis. The reaction product of edaravone with
) has been measured by pulse radiolysis. The reaction product of edaravone with  OH was different from those with other radicals. The rate constants of edaravone derivatives with
OH was different from those with other radicals. The rate constants of edaravone derivatives with  OH have also been determined by both direct measurement and a competition method using CO
OH have also been determined by both direct measurement and a competition method using CO
 and DMPO as competitors.
and DMPO as competitors.
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Yamashita, Shinichi; Fu, H. Y.*; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
In order to investigate the reaction mechanism of edaravone (3-methyl-1-phenyl-2-pyrazolin- 5-one) with OH, the reactivity of edaravone derivatives which have different substituent from edaravone was observed by pulse radiolysis system. From the experiments, OH-adduct radical formed by the addition of OH to phenyl group has been identified as a main intermediate.
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Fu, H.*; Yamashita, Shinichi; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
Edaravone (3-methyl-1-phenyl-2-pyrazolin-5-one), which is clinically used for brain infarct in Japan from 2001, is a potent antioxidative medicine. Edaravone is also expected as a radioprotector for its high reactivity towards oxidizing radicals. Recently, some edaravone derivatives have been being investigated in order to study the Structure Activity Relationship (SAR) and to seek for more efficient radioprotector(s) other than edaravone. It was reported that a derivative with a pyridine ring as a substitute had much higher reactivity with hydroxyl radical than edaravone. However, the early processes of the reactions of edaravone derivatives are not fully understood and the quantitative data about their reaction rate have not been determined. In this study we investigated the reactions between some edaravone derivatives and hydroxyl radical and determined the rate constants of these reactions by pulse radiolysis technique. The rate constants were measured by three different methods. The first was direct measurement from the buildup kinetics of transient radicals. The second was competition method using carbonate ion as a competitor. The third was competitive measurement using DMPO, which is widely used spin-trapping agent for hydroxyl radical or hydrogen atom. The experimental results showed high reactivity of all derivatives investigated in this study. These reactions are near diffusion controlled. Moreover, the rate constants of derivatives with a pyridine ring by competition with DMPO were estimated twice as high as that by direct measurement. A possible interaction between the pyridine ring and DMPO is proposed.
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Fu, H.*; Yamashita, Shinichi; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
Edaravone and its derivatives are good radical scavengers and expected as a potent radioprotector. In this study, their  OH scavenging reactivity was measured by pulse radiolysis technique, and all derivatives showed high reactivity with
OH scavenging reactivity was measured by pulse radiolysis technique, and all derivatives showed high reactivity with  OH. As a result of measuring rate constants by using competition methods by DMPO, it was shown that there was interaction between derivatives including pyridine ring and DMPO.
OH. As a result of measuring rate constants by using competition methods by DMPO, it was shown that there was interaction between derivatives including pyridine ring and DMPO.
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Fu, H.*; Yamashita, Shinichi; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
Edaravone or RadiCut, 3-methyl-1-phenyl-2-pyrazolin-5-one, is a developed free radical scavenger that has been approved in Japan as a neuroprotective drug since 2001. For its antioxidative property and high reactivity with free radicals, edaravone has been expected as a radioprotector. In the last decade, the reactivity of edaravone with peroxyl radicals or some enzymes which induce oxidizing reactions, as well as the antioxidative properties of edaravone, have been extensively investigated. However, there are only a few reports on the measurements of the reactivity of edaravone towrads OH radical, although OH radical is thought to be a main factor of indirect radiation induced damages. In this work, we measured the reactivity of edaravone with OH radical, and elucidated its reaction mechanism. The reactions of edaravone and its model compound (1,3-dimethyl-2-pyrazolin-5-one) with OH radical or other oxidizing radicals were measured by pulse radiolysis techniques. The result shows that the intermediate radical produced by the reaction of edaravone with OH radical was spectroscopically different from that by the reactions with other radicals. On the other hand, for 1,3-dimethyl-2-pyrazolin-5-one, the intermediate radical by the reaction with OH radical is similar to that by the reaction with other oxidizing radical. All these strongly suggest that the phenyl group of edaravone plays an important role on the reaction with OH radical. Thus, in contrast to the previous reports in which the oxidation by OH radical was assumed to be through electron transfer or hydrogen abstraction from pyrazolin-5-one, the formation of OH adducts is crucial for the reaction of edaravone towards OH radical.
Hata, Kuniki; Fu, H. Y.*; Katsumura, Yosuke*; Lin, M.; Muroya, Yusa*; Yamashita, Shinichi; Kudo, Hisaaki*; Nakagawa, Hidehiko*
no journal, ,
no abstracts in English
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Yamashita, Shinichi; Fu, H. Y.*; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
no abstracts in English
Yokobori, Shinichi*; Yang, Y.*; Sugino, Tomohiro*; Kawaguchi, Yuko*; Itahashi, Shiho*; Fujisaki, Kenta*; Fushimi, Hidehiko*; Hasegawa, Sunao*; Hashimoto, Hirofumi*; Hayashi, Nobuhiro*; et al.
no journal, ,
Yokobori, Shinichi*; Yang, Y.*; Sugino, Tomohiro*; Kawaguchi, Yuko*; Fushimi, Hidehiko*; Narumi, Issei; Hashimoto, Hirofumi*; Hayashi, Nobuhiro*; Kawai, Hideyuki*; Kobayashi, Kensei*; et al.
no journal, ,
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Fu, H.*; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
no abstracts in English