Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Miyakawa, Kazuya; Ishii, Eiichi; Imai, Hisashi*; Hirai, Satoru*; Ono, Hirokazu; Nakata, Kotaro*; Hasegawa, Takuma*
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 31(2), p.82 - 95, 2024/12
no abstracts in English
 Ge
Ge
Fujiwara, Hidenori*; Nakatani, Yasuhiro*; Aratani, Hidekazu*; Kanai-Nakata, Yuina*; Yamagami, Kohei*; Hamamoto, Satoru*; Kiss, Takayuki*; Sekiyama, Akira*; Tanaka, Arata*; Ebihara, Takao*; et al.
New Physics; Sae Mulli, 73(12), p.1062 - 1066, 2023/12
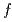 symmetry for anisotropic
symmetry for anisotropic 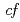 hybridization in the heavy-fermion superconductor CeNi
hybridization in the heavy-fermion superconductor CeNi Ge
Ge
Fujiwara, Hidenori*; Nakatani, Yasuhiro*; Aratani, Hidekazu*; Kanai-Nakata, Yuina*; Yamagami, Kohei*; Hamamoto, Satoru*; Kiss, Takayuki*; Yamasaki, Atsushi*; Higashiya, Atsushi*; Imada, Shin*; et al.
Physical Review B, 108(16), p.165121_1 - 165121_10, 2023/10
Times Cited Count:0 Percentile:0.00(Materials Science, Multidisciplinary)Yamashita, Keishiro*; Komatsu, Kazuki*; Ohara, Takashi; Munakata, Koji*; Irifune, Tetsuo*; Shimmei, Toru*; Sugiyama, Kazumasa*; Kawamata, Toru*; Kagi, Hiroyuki*
High Pressure Research, 42(1), p.121 - 135, 2022/03
Times Cited Count:4 Percentile:46.56(Physics, Multidisciplinary)Kusano, Kanya*; Ichimoto, Kiyoshi*; Ishii, Mamoru*; Miyoshi, Yoshizumi*; Yoden, Shigeo*; Akiyoshi, Hideharu*; Asai, Ayumi*; Ebihara, Yusuke*; Fujiwara, Hitoshi*; Goto, Tadanori*; et al.
Earth, Planets and Space (Internet), 73(1), p.159_1 - 159_29, 2021/12
Times Cited Count:7 Percentile:38.33(Geosciences, Multidisciplinary)The PSTEP is a nationwide research collaboration in Japan and was conducted from April 2015 to March 2020, supported by a Grant-in-Aid for Scientific Research on Innovative Areas from the Ministry of Education, Culture, Sports, Science and Technology of Japan. It has made a significant progress in space weather research and operational forecasts, publishing over 500 refereed journal papers and organizing four international symposiums, various workshops and seminars, and summer school for graduate students at Rikubetsu in 2017. This paper is a summary report of the PSTEP and describes the major research achievements it produced.
 Co
Co Fe
Fe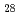 O
O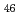
Komabuchi, Mai*; Urushihara, Daisuke*; Asaka, Toru*; Fukuda, Koichiro*; Ohara, Takashi; Munakata, Koji*; Ishikawa, Yoshihisa*
Journal of the Physical Society of Japan, 89(3), p.034601_1 - 034601_5, 2020/03
Times Cited Count:2 Percentile:17.68(Physics, Multidisciplinary)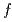 symmetry in CeCu
symmetry in CeCu Ge
Ge ; Soft X-ray absorption and hard X-ray photoemission spectroscopy
; Soft X-ray absorption and hard X-ray photoemission spectroscopyAratani, Hidekazu*; Nakatani, Yasuhiro*; Fujiwara, Hidenori*; Kawada, Moeki*; Kanai-Nakata, Yuina*; Yamagami, Kohei*; Fujioka, Shuhei*; Hamamoto, Satoru*; Kuga, Kentaro*; Kiss, Takayuki*; et al.
Physical Review B, 98(12), p.121113_1 - 121113_6, 2018/09
Times Cited Count:5 Percentile:22.25(Materials Science, Multidisciplinary)Suzuki, Akifumi; Suzuki, Takehiko; Takahashi, Masa; Nakata, Toru; Murayama, Takashi; Tsunoda, Masahiko
JAEA-Technology 2014-049, 19 Pages, 2015/03
Optically Stimulated Luminescence, OSL, dosimeters have been used as individual dosimeters for external radiation in Nuclear Science Research Institute and so on since October, 2014 as successor of the RPL glass dosimeters. Characteristics of the OSL dosimeters such as dose linearity, energy response, angular dependence, fading characteristics and responses at mixed irradiation fields were examined prior to the start of use. As a result, it was found that the OSL dosimeters met the performances that the national standard (JIS Z 4339) determined. The characteristics of OSL dosimeters were comparable with those of the RPL glass dosimeters. In conclusion, it was confirmed the OSL dosimeters had sufficient performances for the practical use on individual monitoring. This report shows the testing methods and the results for the characteristics of OSL dosimeters.
Ito, Kenichi*; Miyahara, Hidetaka*; Ujiie, Toru*; Takeshima, Toshikatsu*; Yokoyama, Shingo*; Nakata, Kotaro*; Nagano, Tetsushi; Sato, Tsutomu*; Hatta, Tamao*; Yamada, Hirohisa*
Nihon Genshiryoku Gakkai Wabun Rombunshi, 11(4), p.255 - 271, 2012/04
no abstracts in English
Takeda, Seiji; Yamaguchi, Tetsuji; Nagasawa, Hirokazu; Watanabe, Masatoshi; Sekioka, Yasushi; Kanzaki, Yutaka; Sasaki, Toshihisa; Ochiai, Toru; Munakata, Masahiro; Tanaka, Tadao; et al.
JAEA-Research 2009-034, 239 Pages, 2009/11
In safety assessment for geological disposal of high level radioactive waste, it is of consequence to estimate the uncertainties due to the long-term frame associated with long-lived radionuclides and the expanded geological environment. The uncertainties result from heterogeneity intrinsic to engineered and natural barrier materials, insufficient understanding of phenomena occurring in the disposal system, erroneous method of measurement, and incomplete construction. It is possible to quantify or to reduce the uncertainties according to scientific and technological progress. We applied a deterministic and a Monte Carlo-based probabilistic method simulation techniques to the uncertainty analysis for performance of hypothetical geological disposal system for high level radioactive waste. This study provides the method to evaluate the effects of the uncertainties with respect to scenarios, models and parameters in engineering barrier system on radiological consequence. The results also help us to specify prioritized models and parameters to be further studied for long-term safety assessment.
Tsuchiya, Kunihiko; Hoshino, Tsuyoshi; Kawamura, Hiroshi; Mishima, Yoshinao*; Yoshida, Naoaki*; Terai, Takayuki*; Tanaka, Satoru*; Munakata, Kenzo*; Kato, Shigeru*; Uchida, Munenori*; et al.
Nuclear Fusion, 47(9), p.1300 - 1306, 2007/09
Times Cited Count:23 Percentile:60.36(Physics, Fluids & Plasmas)no abstracts in English
Mishima, Yoshinao*; Yoshida, Naoaki*; Kawamura, Hiroshi; Ishida, Kiyohito*; Hatano, Yuji*; Shibayama, Tamaki*; Munakata, Kenzo*; Sato, Yoshiyuki*; Uchida, Munenori*; Tsuchiya, Kunihiko; et al.
Journal of Nuclear Materials, 367-370(2), p.1382 - 1386, 2007/08
Times Cited Count:29 Percentile:85.39(Materials Science, Multidisciplinary)no abstracts in English
Tsuchiya, Kunihiko; Hoshino, Tsuyoshi; Kawamura, Hiroshi; Mishima, Yoshinao*; Yoshida, Naoaki*; Terai, Takayuki*; Tanaka, Satoru*; Munakata, Kenzo*; Kato, Shigeru*; Uchida, Munenori*; et al.
Proceedings of 21st IAEA Fusion Energy Conference (FEC 2006) (CD-ROM), 8 Pages, 2007/03
no abstracts in English
Tsuchiya, Kunihiko; Kawamura, Hiroshi; Mishima, Yoshinao*; Yoshida, Naoaki*; Tanaka, Satoru*; Uchida, Munenori*; Ishida, Kiyohito*; Shibayama, Tamaki*; Munakata, Kenzo*; Sato, Yoshiyuki*; et al.
Purazuma, Kaku Yugo Gakkai-Shi, 83(3), p.207 - 214, 2007/03
no abstracts in English
Kawamura, Hiroshi; Tsuchiya, Kunihiko; Mishima, Yoshinao*; Yoshida, Naoaki*; Munakata, Kenzo*; Ishida, Kiyohito*; Hatano, Yuji*; Shibayama, Tamaki*; Sato, Yoshiyuki*; Uchida, Munenori*; et al.
INL/EXT-06-01222, p.1 - 7, 2006/02
no abstracts in English
Kawamura, Hiroshi; Takahashi, Heishichiro*; Yoshida, Naoaki*; Mishima, Yoshinao*; Ishida, Kiyohito*; Iwadachi, Takaharu*; Cardella, A.*; Van der Laan, J. G.*; Uchida, Munenori*; Munakata, Kenzo*; et al.
Journal of Nuclear Materials, 329-333(1), p.112 - 118, 2004/08
Times Cited Count:39 Percentile:89.73(Materials Science, Multidisciplinary)no abstracts in English
Kitamura, Akira; Nakata, Kotaro*; Tanaka, Satoru*; Tomura, Tsutomu*; Kamei, Gento
Saikuru Kiko Giho, (22), p.59 - 66, 2004/03
Redox reactions between neptunium(V) (Np(V)) and magnetite (Fe(II)1Fe(III)2O4) surface were investigated in N gas atmosphere. A batch method was applied to the experiment. High-pure magnetite and a 0.1 M NaCl were mixed in a polypropylene tube, and pH, redox potential and concentration of dissolved neptunium were measured as a function of shaking time (from 1 hour to 7 days), temperature (298 K and 318 K) and liquid/solid ratio (20, 50 and 100 ml.g
gas atmosphere. A batch method was applied to the experiment. High-pure magnetite and a 0.1 M NaCl were mixed in a polypropylene tube, and pH, redox potential and concentration of dissolved neptunium were measured as a function of shaking time (from 1 hour to 7 days), temperature (298 K and 318 K) and liquid/solid ratio (20, 50 and 100 ml.g ). It was observed that the concentration of dissolved neptunium was reduced rapidly within a day, due to the reducing of Np(V) to Np(IV) and the precipitation of Np(IV). This result was shown typically when the magnetite/solution ratio and the temperature were high. The rate constant of the redox reaction and the activation energy for the rate constant were preliminarily obtained. It was suggested that the redox reaction was promoted by not only Fe(II) on magnetite surface but also Fe(II) inside the magnetite.
). It was observed that the concentration of dissolved neptunium was reduced rapidly within a day, due to the reducing of Np(V) to Np(IV) and the precipitation of Np(IV). This result was shown typically when the magnetite/solution ratio and the temperature were high. The rate constant of the redox reaction and the activation energy for the rate constant were preliminarily obtained. It was suggested that the redox reaction was promoted by not only Fe(II) on magnetite surface but also Fe(II) inside the magnetite.
Tanaka, Satoru*; Nagasaki, Shinya*; Nakata, Kotaro*; Oda, Takuji*; Kameda, Jun*; Kamei, Gento; Tachi, Yukio
JNC TY8400 2003-008, 88 Pages, 2003/05
Redox reactions between Cr(VI) and iron(II) chloride (FeCl2) and those between Cr(VI) and magnetite (Fe(II)1Fe(III)2O4) were observed as a preliminary study. According to the experimental results, it was suggested that the redox reactions were promoted more than the amount of Fe(II) on magnetite surface because of electron transfer from internal Fe(II) to magnetite surface. The results were quantitatively supported from quantum chemical calculations. Redox reactions between Np(V) and magnetite and the reduction of Np to tetravalent were observed, while those between Np(V) and FeCl2 were not observed obviously. It was observed that the reactions were promoted rapidly when the magnetite / solution ratio and the temperature were high, and the rate constant of the reactions was obtained. Furthermore, it was found that hydrogen gas and hydrogen ion were generated with crushing the quartz in an inert gas atmosphere.
Katanishi, Shoji; Kunitomi, Kazuhiko; Takei, Masanobu; Nakata, Tetsuo; Watanabe, Takashi*; Izumiya, Toru*
Nihon Genshiryoku Gakkai Wabun Rombunshi, 1(4), p.373 - 383, 2002/12
no abstracts in English
Nakata, Kotaro*; Nagasaki, Shinya*; Tanaka, Satoru*; Sakamoto, Yoshiaki; Tanaka, Tadao; Ogawa, Hiromichi
Radiochimica Acta, 90(9-11), p.665 - 669, 2002/12
Times Cited Count:53 Percentile:93.51(Chemistry, Inorganic & Nuclear)Sorption and desorption experiments of Np on magnetite and hematite under aerobic and anaerobic conditions were carried out to investigate the possibility of reduction of Np(V) to Np(IV) on the surfave of iron oxides including Fe(II). The results indicated that Np sorption mechanism on magnetite under anaerobic condition was completely different from that under aerobic condition. The evidence of the presence of Np(IV) on magnetite surface after sorption was obtained from the extraction experiment with TTA/xylene solution.