Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...

Tamura, Kazuhisa; Oko, Yoshihisa*; Kawamura, Hiroyuki; Yoshikawa, Hideki*; Tatsuma, Tetsu*; Fujishima, Akira*; Mizuki, Junichiro
Electrochimica Acta, 52(24), p.6938 - 6942, 2007/08
Times Cited Count:26 Percentile:48.64(Electrochemistry)The electrochemical behaviour of TiO under X-ray irradiation was studied. Under X-ray irradiation a current, a negative shift in the rest potential, and electrochemical oxidation and decomposition of [Fe
under X-ray irradiation was studied. Under X-ray irradiation a current, a negative shift in the rest potential, and electrochemical oxidation and decomposition of [Fe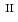 (CN)
(CN) ]
]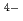 were clearly observed. The incident photon-current conversion efficiency and energy conversion efficiency was 400-2000% and 0.2-2%, respectively, depending on sample conditions. These results show that the photoelectrochemical reactions were promoted by X-rays with a high incident photon-current conversion efficiency. The photocurrent and photopotential were observed above 4.965 keV, which corresponds to the Ti-K edge, indicating that electron-hole pairs are formed during the relaxation process of the excited Ti atoms.
were clearly observed. The incident photon-current conversion efficiency and energy conversion efficiency was 400-2000% and 0.2-2%, respectively, depending on sample conditions. These results show that the photoelectrochemical reactions were promoted by X-rays with a high incident photon-current conversion efficiency. The photocurrent and photopotential were observed above 4.965 keV, which corresponds to the Ti-K edge, indicating that electron-hole pairs are formed during the relaxation process of the excited Ti atoms.

Tamura, Kazuhisa; Oko, Yoshihisa*; Tatsuma, Tetsu*; Mizuki, Junichiro
no journal, ,
We measured a cyclic voltammogram of TiO under intermittent X-ray irradiation and time courses of the rest potential under X-ray irradiation and in the dark. Both the oxidation current and the negative shift of the rest potential were observed only under irradiation, indicating that the electron-hole pair formation and the photocatalytic reaction occur under X-ray irradiation. We also measured action spectra of the photocurrent and photopotential. It was found that both the photocurrent and photopotential with very high quantum efficiency (400%) were observed above Ti-K edge and the fine structure of action spectra had the same structure of XANES spectrum, indicating that electrons in the valence band are excited during the relaxation process of excited inner shell electrons of Ti.
under intermittent X-ray irradiation and time courses of the rest potential under X-ray irradiation and in the dark. Both the oxidation current and the negative shift of the rest potential were observed only under irradiation, indicating that the electron-hole pair formation and the photocatalytic reaction occur under X-ray irradiation. We also measured action spectra of the photocurrent and photopotential. It was found that both the photocurrent and photopotential with very high quantum efficiency (400%) were observed above Ti-K edge and the fine structure of action spectra had the same structure of XANES spectrum, indicating that electrons in the valence band are excited during the relaxation process of excited inner shell electrons of Ti.
Tamura, Kazuhisa; Oko, Yoshihisa*; Matsuzawa, Sadao*; Mizuki, Junichiro
no journal, ,
The electrochemical behavior of TiO under X-ray irradiation was studied. Under X-ray irradiation a current, a negative shift in the rest potential, and electrochemical oxidation and decomposition of [FeII(CN)6]4- were clearly observed. The incident photon-current conversion efficiency was 400-2000 %, depending on sample conditions. These results show that the photoelectrochemical reactions were promoted by X-rays with a high incident photon-current conversion efficiency. The photocurrent and photopotential were observed above 4.965 keV, which corresponds to the Ti-K edge, indicating that electron-hole pairs are formed during the relaxation process of the excited Ti atoms.
under X-ray irradiation was studied. Under X-ray irradiation a current, a negative shift in the rest potential, and electrochemical oxidation and decomposition of [FeII(CN)6]4- were clearly observed. The incident photon-current conversion efficiency was 400-2000 %, depending on sample conditions. These results show that the photoelectrochemical reactions were promoted by X-rays with a high incident photon-current conversion efficiency. The photocurrent and photopotential were observed above 4.965 keV, which corresponds to the Ti-K edge, indicating that electron-hole pairs are formed during the relaxation process of the excited Ti atoms.