Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kaneda-Nakashima, Kazuko*; Shirakami, Yoshifumi*; Kadonaga, Yuichiro*; Watabe, Tadashi*; Oe, Kazuhiro*; Yin, X.*; Haba, Hiromitsu*; Shirasaki, Kenji*; Kikunaga, Hidetoshi*; Tsukada, Kazuaki; et al.
International Journal of Molecular Sciences (Internet), 25(2), p.933_1 - 933_14, 2024/01
Times Cited Count:1 Percentile:0.01(Biochemistry & Molecular Biology) Ni
Ni In
In
Sakai, Hironori; Tokunaga, Yo; Kambe, Shinsaku; Zhu, J.-X.*; Ronning, F.*; Thompson, J. D.*; Kotegawa, Hisashi*; To, Hideki*; Suzuki, Kohei*; Oshima, Yoshiki*; et al.
Physical Review B, 106(23), p.235152_1 - 235152_8, 2022/12
Times Cited Count:0 Percentile:0(Materials Science, Multidisciplinary)We investigate the electronic state of Ni-substituted CeCo Ni
Ni In
In by nuclear quadrupole and magnetic resonance (NQR/NMR) techniques. The heavy fermion superconductivity below
by nuclear quadrupole and magnetic resonance (NQR/NMR) techniques. The heavy fermion superconductivity below 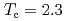 K for
K for 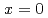 is suppressed by Ni substitutions, and
is suppressed by Ni substitutions, and 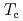 reaches zero for
reaches zero for 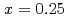 . The
. The 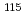 In NQR spectra for
In NQR spectra for 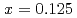 and 0.25 can be explained by simulating the electrical field gradient that is calculated for a virtual supercell with density functional theory. The spin-lattice relaxation rate
and 0.25 can be explained by simulating the electrical field gradient that is calculated for a virtual supercell with density functional theory. The spin-lattice relaxation rate 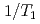 indicates that Ni substitution weakens antiferromagnetic correlations that are not localized near the substituent but instead are uniform in space. The temperature (
indicates that Ni substitution weakens antiferromagnetic correlations that are not localized near the substituent but instead are uniform in space. The temperature (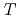 ) dependence of
) dependence of 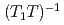 for
for 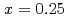 shows a maximum around
shows a maximum around 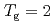 K and
K and 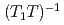 decreases toward almost zero when temperature is further reduced as if a gap might be opening in the magnetic excitation spectrum; however, the magnetic specific heat and the static magnetic susceptibility evolve smoothly through
decreases toward almost zero when temperature is further reduced as if a gap might be opening in the magnetic excitation spectrum; however, the magnetic specific heat and the static magnetic susceptibility evolve smoothly through 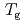 with a
with a 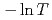 dependence. The peculiar T dependence of
dependence. The peculiar T dependence of 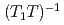 and non-Fermi-liquid specific heat and susceptibility can be interpreted in a unified way by assuming nested antiferromagnetic spin fluctuations in a quasi-two-dimensional electronic system.
and non-Fermi-liquid specific heat and susceptibility can be interpreted in a unified way by assuming nested antiferromagnetic spin fluctuations in a quasi-two-dimensional electronic system.
Yakushev, A.*; Lens, L.*; D llmann, Ch. E.*; Khuyagbaatar, J.*; J
llmann, Ch. E.*; Khuyagbaatar, J.*; J ger, E.*; Krier, J.*; Runke, J.*; Albers, H. M.*; Asai, Masato; Block, M.*; et al.
ger, E.*; Krier, J.*; Runke, J.*; Albers, H. M.*; Asai, Masato; Block, M.*; et al.
Frontiers in Chemistry (Internet), 10, p.976635_1 - 976635_11, 2022/08
Times Cited Count:9 Percentile:79.28(Chemistry, Multidisciplinary)Flerovium (Fl, element 114) is the heaviest element chemically studied so far. The first chemical experiment on Fl suggested that Fl is a noble-gas-like element, while the second studies suggested that Fl has a volatile-metal-like character. To obtain more reliable conclusion, we performed further experimental studies on Fl adsorption behavior on Si oxide and gold surfaces. The present results suggest that Fl is highly volatile and less reactive than the volatile metal, Hg, but has higher reactivity than the noble gas, Rn.
Hirade, Tetsuya; Michishio, Koji*; Kobayashi, Yoshinori*; Oshima, Nagayasu*
Chemical Physics Letters, 795, p.139507_1 - 139507_4, 2022/05
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)We obtained the temperature dependence up to 150 C of the triplet positronium (
C of the triplet positronium (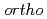 -Ps) lifetime in N,N,N-Trimethyl-N-propylammonium bis(trifluoromethanesulfonyl)imide (TMPA-TFSI) by the vertical slow positron beamline installed at AIST. Positrons penetrate into the liquid surface of TMPA-TFSI with the positron energies of 2 keV and 12 keV to investigate at the near-surface and the balk. The surface structure was visible at 150
-Ps) lifetime in N,N,N-Trimethyl-N-propylammonium bis(trifluoromethanesulfonyl)imide (TMPA-TFSI) by the vertical slow positron beamline installed at AIST. Positrons penetrate into the liquid surface of TMPA-TFSI with the positron energies of 2 keV and 12 keV to investigate at the near-surface and the balk. The surface structure was visible at 150 C, 120
C, 120 C above the melting temperature. The
C above the melting temperature. The 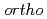 -Ps lifetime became shorter at higher temperatures for both positron energies. Similar temperature dependence had appeared just in water as the result of the reaction of
-Ps lifetime became shorter at higher temperatures for both positron energies. Similar temperature dependence had appeared just in water as the result of the reaction of 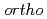 -Ps and radiolysis products such as the OH radicals. The temperature dependence observed for TMPA-TFSI suggested that the chemical reaction of
-Ps and radiolysis products such as the OH radicals. The temperature dependence observed for TMPA-TFSI suggested that the chemical reaction of 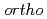 -Ps occurred.
-Ps occurred.
G tz, M.*; Yakushev, A.*; G
tz, M.*; Yakushev, A.*; G tz, S.*; Di Nitto, A.*; D
tz, S.*; Di Nitto, A.*; D llmann, Ch. E.*; Asai, Masato; Kindler, B.*; Krier, J.*; Lommel, B.*; Nagame, Yuichiro*; et al.
llmann, Ch. E.*; Asai, Masato; Kindler, B.*; Krier, J.*; Lommel, B.*; Nagame, Yuichiro*; et al.
Radiochimica Acta, 110(2), p.75 - 86, 2022/02
Times Cited Count:2 Percentile:31.78(Chemistry, Inorganic & Nuclear)The study of volatile superheavy element carbonyl complexes requires more efficient methods because the yield of transactinide elements decreases with increasing atomic number. This is achieved by using a newly developed double chamber system to separate the recoil chamber and the reaction one, thereby avoiding the decomposition of reactive molecules by the projectile ion beam, which hinders the synthesis of carbonyl complexes. The feasibility of this method was verified by synthesizing 5d metal short-lived isotopes as homologous element isotopes of the light transactinide elements Sg, Bh, Hs, and Mt at the Japan Atomic Energy Agency tandem accelerator and conducting model experiments.
Yakushev, A.*; Lens, L.*; D llmann, C. E.*; Block, M.*; Nagame, Yuichiro*; Sato, Tetsuya; Toyoshima, Atsushi*; 42 of others*
llmann, C. E.*; Block, M.*; Nagame, Yuichiro*; Sato, Tetsuya; Toyoshima, Atsushi*; 42 of others*
Frontiers in Chemistry (Internet), 9, p.753738_1 - 753738_9, 2021/11
Times Cited Count:10 Percentile:63.67(Chemistry, Multidisciplinary)Nihonium (Nh, element 113) and flerovium (Fl, element 114) are the first superheavy elements in which the 7p shell is occupied. High volatility and inertness were predicted for Fl due to the strong relativistic stabilization of the closed 7p sub-shell, which originates from a large spin-orbit splitting between the 7p
sub-shell, which originates from a large spin-orbit splitting between the 7p and 7p
and 7p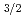 orbitals. One unpaired electron in the outermost 7p
orbitals. One unpaired electron in the outermost 7p sub-shell in Nh is expected to give rise to a higher chemical reactivity. Theoretical predictions of Nh reactivity are discussed, along with results of the first experimental attempts to study Nh chemistry in the gas phase. The experimental observations verify a higher chemical reactivity of Nh atoms compared to its neighbor Fl and call for the development of advanced setups. First tests of a newly developed detection device miniCOMPACT with highly reactive Fr isotopes assure that effective chemical studies of Nh are within reach.
sub-shell in Nh is expected to give rise to a higher chemical reactivity. Theoretical predictions of Nh reactivity are discussed, along with results of the first experimental attempts to study Nh chemistry in the gas phase. The experimental observations verify a higher chemical reactivity of Nh atoms compared to its neighbor Fl and call for the development of advanced setups. First tests of a newly developed detection device miniCOMPACT with highly reactive Fr isotopes assure that effective chemical studies of Nh are within reach.
 ;
; 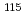 In NQR/NMR and
In NQR/NMR and 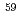 Co NMR study
Co NMR studySakai, Hironori; Tokunaga, Yo; Kambe, Shinsaku; Zhu, J.-X.*; Ronning, F.*; Thompson, J. D.*; Ramakrishna, S. K.*; Reyes, A. P.*; Suzuki, Kohei*; Oshima, Yoshiki*; et al.
Physical Review B, 104(8), p.085106_1 - 085106_12, 2021/08
Times Cited Count:3 Percentile:27.71(Materials Science, Multidisciplinary)Antiferromagnetism in a prototypical quantum critical metal CeCoIn is known to be induced by slight substitutions of non-magnetic Zn atoms for In. In nominally 7% Zn substituted CeCoIn
is known to be induced by slight substitutions of non-magnetic Zn atoms for In. In nominally 7% Zn substituted CeCoIn , an antiferromagnetic (AFM) state coexists with heavy fermion superconductivity. Heterogeneity of the electronic states is investigated in Zn doped CeCoIn
, an antiferromagnetic (AFM) state coexists with heavy fermion superconductivity. Heterogeneity of the electronic states is investigated in Zn doped CeCoIn by means of nuclear quadrupole and magnetic resonances (NQR and NMR). Site-dependent NQR relaxation rates
by means of nuclear quadrupole and magnetic resonances (NQR and NMR). Site-dependent NQR relaxation rates 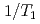 indicate that the AFM state is locally nucleated around Zn substituents in the matrix of a heavy fermion state, and percolates through the bulk at the AFM transition temperature
indicate that the AFM state is locally nucleated around Zn substituents in the matrix of a heavy fermion state, and percolates through the bulk at the AFM transition temperature 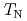 . At lower temperatures, an anisotropic superconducting (SC) gap below the SC transition temperature
. At lower temperatures, an anisotropic superconducting (SC) gap below the SC transition temperature 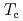 , and the SC state permeates through the AFM regions via a SC proximity effect. Applying an external magnetic field induces a spin-flop transition near 5 T, reducing the volume of the AFM regions. Consequently, a short ranged inhomogeneous AFM state survives and coexists with a paramagnetic Fermi liquid state at high fields.
, and the SC state permeates through the AFM regions via a SC proximity effect. Applying an external magnetic field induces a spin-flop transition near 5 T, reducing the volume of the AFM regions. Consequently, a short ranged inhomogeneous AFM state survives and coexists with a paramagnetic Fermi liquid state at high fields.
Nakasone, Shunya*; Yokoyama, Sumi*; Takahashi, Tomoyuki*; Ota, Masakazu; Kakiuchi, Hideki*; Sugihara, Shinji*; Hirao, Shigekazu*; Momoshima, Noriyuki*; Tamari, Toshiya*; Shima, Nagayoshi*; et al.
Plasma and Fusion Research (Internet), 16, p.2405035_1 - 2405035_5, 2021/02
Removal of impurities such as organic and other types of dissolved matters from environmental water samples is required for precise analysis of tritium with a liquid scintillation counting method. In general, a distillation method is a conventional one for tritium analysis in environmental water samples, but is a time-consuming process that takes 24 hours for removal of impurities. We have proposed a rapid pretreatment method for tritium analysis, that uses ion exchange resins. In this study, we performed batch experiments, to evaluate the effectiveness of the ion exchange resins on the tritium measurement. The results obtained demonstrated that removal of impurities in the sample water by ion exchange resins can be achieved during a short period of time (i.e., in 5 min).
 Bh in the
Bh in the  Cm(
Cm(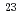 Na,5
Na,5 )
) Bh reaction and its decay properties
Bh reaction and its decay propertiesHaba, Hiromitsu*; Fan, F.*; Kaji, Daiya*; Kasamatsu, Yoshitaka*; Kikunaga, Hidetoshi*; Komori, Yukiko*; Kondo, Narumi*; Kudo, Hisaaki*; Morimoto, Koji*; Morita, Kosuke*; et al.
Physical Review C, 102(2), p.024625_1 - 024625_12, 2020/08
Times Cited Count:6 Percentile:59.56(Physics, Nuclear)Nakasone, Shunya*; Yokoyama, Sumi*; Takahashi, Tomoyuki*; Ota, Masakazu; Kakiuchi, Hideki*; Sugihara, Shinji*; Hirao, Shigekazu*; Momoshima, Noriyuki*; Tamari, Toshiya*; Shima, Nagayoshi*; et al.
Plasma and Fusion Research (Internet), 15, p.2405027_1 - 2405027_3, 2020/05
A quick preprocessing system for tritium analysis of environmental samples is important to judge environmental influence of tritium releases due to accident or tritium-handling facilities. Analysis of tritium in water samples with liquid scintillation counting method requires removal of impurities such as organic matter and ion species from water samples. Generally, a distillation method is adopted as a pretreatment of analysis for tritium; however, the distillation method is a time-consuming process. The aim of this study is to evaluate a rapid pretreatment method for tritium analysis with ion exchange resin. From batch and column experiments that used inland water and ion exchange resin, we confirmed removals of impurities of the water sample and that the removal of impurities was possible for a short time (by 5 minutes).
Hirade, Tetsuya; Michishio, Koji*; Kobayashi, Yoshinori*; Oshima, Nagayasu*
Acta Physica Polonica A, 137(2), p.109 - 112, 2020/02
Times Cited Count:0 Percentile:0(Physics, Multidisciplinary)Recently, it was reported that the positronium (Ps) bubble in Room Temperature Ionic Liquids (RTILs) showed some phenomena that were very different from them observed in other molecular liquids. Now the reason of these interesting phenomena is being understood. The structure caused by the ionic interaction between anions and cations can exist even at the higher temperatures than the melting temperatures. It was the reason why the oscillation of Ps bubble was observed at the temperatures near the melting temperatures in RTILs. The temperature dependence of the Ps bubble size estimated by the triplet Ps (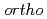 -Ps) pick-off annihilation lifetime indicated that there was a nano-meter structure remained by ionic interactions even at high temperatures. The
-Ps) pick-off annihilation lifetime indicated that there was a nano-meter structure remained by ionic interactions even at high temperatures. The 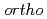 -Ps pick-off annihilation lifetime will be a strong tool to study nano-structure in RTILs.
-Ps pick-off annihilation lifetime will be a strong tool to study nano-structure in RTILs.
Chiera, N. M.; Sato, Tetsuya; Tomitsuka, Tomohiro; Asai, Masato; Ito, Yuta; Shirai, Kaori*; Suzuki, Hayato; Tokoi, Katsuyuki; Toyoshima, Atsushi; Tsukada, Kazuaki; et al.
Journal of Radioanalytical and Nuclear Chemistry, 320(3), p.633 - 642, 2019/06
Times Cited Count:1 Percentile:11.15(Chemistry, Analytical)An isothermal gas-chromatographic (IGC) device has been developed and tested for on-line gas phase studies of volatile oxychlorides of short-lived group-5 transition metals. Radioisotopes of niobium and tantalum, produced in nuclear fusion evaporation reactions, are directly flushed into the IGC setup by an inert gas-jet. Oxychloride compounds are formed by the addition of SOCl and O
and O . Parameters influencing the formation and transport of NbOCl
. Parameters influencing the formation and transport of NbOCl and TaOCl
and TaOCl are investigated. For nuclides with half-lives (
are investigated. For nuclides with half-lives (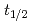 ) of about 30 s, an overall efficiency of 7% is obtained, rendering the IGC setup suitable for the chemical exploration of
) of about 30 s, an overall efficiency of 7% is obtained, rendering the IGC setup suitable for the chemical exploration of  Db(
Db(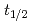 = 34s).
= 34s).
Yokoyama, Sumi*; Takahashi, Tomoyuki*; Ota, Masakazu; Kakiuchi, Hideki*; Sugihara, Shinji*; Hirao, Shigekazu*; Momoshima, Noriyuki*; Tamari, Toshiya*; Shima, Nagayoshi*; Atarashi-Andoh, Mariko; et al.
Plasma and Fusion Research (Internet), 14(Sp.2), p.3405099_1 - 3405099_4, 2019/06
The Large Helical Device of the National Institute for Fusion Science started D-D experiments in 2017. To ensure the safety of the facility, it is important to develop evaluation methods for environmental tritium transfer. Tritiated water (HTO) in atmosphere and soil is transferred to plants, and organically bound tritium (OBT) is formed by photosynthesis. Prediction of OBT formation is important, because OBT accumulates in plants and causes dose through ingestion. The objective of this study is to estimate environmental tritium transfer using a simple compartment model and practical parameters. We proposed a simple compartment model consisting of air-soil-plant components, and tried to validate the model by comparison with a sophisticated model, SOLVEG. In this study, we plan to add wet deposition to the model and obtain parameters from measurements of soil permeability and tritium concentrations in air, soil and plants. We also establish rapid pretreatment methods for OBT analysis.
Chiera, N. M.; Sato, Tetsuya; Tomitsuka, Tomohiro; Asai, Masato; Suzuki, Hayato*; Tokoi, Katsuyuki; Toyoshima, Atsushi; Tsukada, Kazuaki; Nagame, Yuichiro
Inorganica Chimica Acta, 486, p.361 - 366, 2019/02
Times Cited Count:3 Percentile:18.6(Chemistry, Inorganic & Nuclear)The formation of NbOCl and TaOCl
and TaOCl and their adsorption behavior on quartz surfaces was explored by applying an isothermal gas-chromatographic method. Trace amounts of short-lived Nb and Ta isotopes were used. Adsorption enthalpy values (
and their adsorption behavior on quartz surfaces was explored by applying an isothermal gas-chromatographic method. Trace amounts of short-lived Nb and Ta isotopes were used. Adsorption enthalpy values ( ) at zero surface coverage of -
) at zero surface coverage of - (NbOCl
(NbOCl ) = 102
) = 102  4 kJ/mol and -
4 kJ/mol and - (TaOCl
(TaOCl ) = 128
) = 128  5 kJ/mol were determined by analyzing the chromatographic behavior of the Nb andTa complexes with a Monte-Carlo simulation method based on an adsorption-desorption kinetic model.By applying an empirical correlation, the experimental
5 kJ/mol were determined by analyzing the chromatographic behavior of the Nb andTa complexes with a Monte-Carlo simulation method based on an adsorption-desorption kinetic model.By applying an empirical correlation, the experimental  values were successively related to the macroscopic standard sublimation enthalpy,
values were successively related to the macroscopic standard sublimation enthalpy, 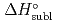 , as a measure of the volatility of each substance. The inferred sublimation enthalpies are in agreement with tabulated thermochemical values. Thus, the linear empirical correlation between
, as a measure of the volatility of each substance. The inferred sublimation enthalpies are in agreement with tabulated thermochemical values. Thus, the linear empirical correlation between  and
and 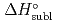 for metal-oxychlorides was updated with the inclusion of the present data. According to the predicted
for metal-oxychlorides was updated with the inclusion of the present data. According to the predicted 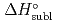 (DbOCl
(DbOCl ), a
), a  (DbOCl
(DbOCl ) value of 135
) value of 135  2 kJ/mol was extrapolated. The future accomplishment of comparative studies with DbOCl
2 kJ/mol was extrapolated. The future accomplishment of comparative studies with DbOCl under the same experimental conditions will provide valuable information on the volatility trend in Group-5 elements, together with an indication on the magnitude of relativistic effects on the electronic structure of dubnium.
under the same experimental conditions will provide valuable information on the volatility trend in Group-5 elements, together with an indication on the magnitude of relativistic effects on the electronic structure of dubnium.
 acidic solutions
acidic solutionsYokoyama, Akihiko*; Kitayama, Yuta*; Fukuda, Yoshiki*; Kikunaga, Hidetoshi*; Murakami, Masashi*; Komori, Yukiko*; Yano, Shinya*; Haba, Hiromitsu*; Tsukada, Kazuaki; Toyoshima, Atsushi*
Radiochimica Acta, 107(1), p.27 - 32, 2019/01
Times Cited Count:1 Percentile:11.15(Chemistry, Inorganic & Nuclear)Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Beerwerth, R.*; Kaneya, Yusuke*; Makii, Hiroyuki; Mitsukai, Akina*; Nagame, Yuichiro; Osa, Akihiko; Toyoshima, Atsushi; et al.
Journal of the American Chemical Society, 140(44), p.14609 - 14613, 2018/11
Times Cited Count:27 Percentile:69.46(Chemistry, Multidisciplinary)The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
 SO
SO and HF/HCl solutions into toluene with Aliquat336; Sulfate and fluoride complex formation of Mo and W towards chemical studies of seaborgium (Sg)
and HF/HCl solutions into toluene with Aliquat336; Sulfate and fluoride complex formation of Mo and W towards chemical studies of seaborgium (Sg)Toyoshima, Atsushi; Mitsukai, Akina; Tsukada, Kazuaki; Oe, Kazuhiro*; Haba, Hiromitsu*; Komori, Yukiko*; Murakami, Masashi; Kaneya, Yusuke*; Sato, Daisuke*; Asai, Masato; et al.
Journal of Radioanalytical and Nuclear Chemistry, 317(1), p.421 - 430, 2018/07
Times Cited Count:2 Percentile:20.93(Chemistry, Analytical)We have studied extraction behavior of group-6 elements Mo and W to search for suitable conditions for an on-line extraction experiment of their heavier homolog, seaborgium (Sg). Batch-wise extraction of carrier-free radiotracers  Mo and
Mo and 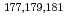 W were carried out from 0.10 - 8.6 M H
W were carried out from 0.10 - 8.6 M H SO
SO and 1.0
and 1.0 10
10 - 5.0 M HF/1.0 M HCl into toluene with a quaternary ammonium compound, Aliquat336. Anionic sulfate complexes of Mo and W with charge - 2 were extracted with Aliquat336 from H
- 5.0 M HF/1.0 M HCl into toluene with a quaternary ammonium compound, Aliquat336. Anionic sulfate complexes of Mo and W with charge - 2 were extracted with Aliquat336 from H SO
SO solutions with concentrations of [H
solutions with concentrations of [H SO
SO ]
]  2 M. In HF/1.0 M HCl, oxyfluoro complexes of Mo and W with charge - 1 were interpreted to be formed and extracted with Aliquat336. From these results, favorable conditions for the extraction of Sg are discussed.
2 M. In HF/1.0 M HCl, oxyfluoro complexes of Mo and W with charge - 1 were interpreted to be formed and extracted with Aliquat336. From these results, favorable conditions for the extraction of Sg are discussed.
Nishio, Katsuhisa; Hirose, Kentaro; Vermeulen, M. J.; Makii, Hiroyuki; Orlandi, R.; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Sato, Tetsuya; Nagame, Yuichiro; et al.
EPJ Web of Conferences, 163, p.00041_1 - 00041_6, 2017/11
Times Cited Count:1 Percentile:61.21(Nuclear Science & Technology)Sugita, Tsuyoshi; Fujiwara, Iori*; Okamura, Hiroyuki; Oshima, Tatsuya*; Baba, Yoshinari*; Naganawa, Hirochika; Shimojo, Kojiro
Solvent Extraction Research and Development, Japan, 24(2), p.61 - 69, 2017/05
We investigated an influence of amide group in diglycolamic acid-type extractants on extraction property of metal ions. The extraction characteristics of  -dodecyldiglycolamic acid (C
-dodecyldiglycolamic acid (C DGAA), with a secondary amide group, for 56 metal ions have been investigated, and compared with those of
DGAA), with a secondary amide group, for 56 metal ions have been investigated, and compared with those of  -dioctyldiglycolamic acid (DODGAA) with a tertiary amide group. Compared with DODGAA, C
-dioctyldiglycolamic acid (DODGAA) with a tertiary amide group. Compared with DODGAA, C DGAA has a poor extraction performance and separation ability for rare-earth metal ions, except for Sc(III). However, C
DGAA has a poor extraction performance and separation ability for rare-earth metal ions, except for Sc(III). However, C DGAA tended to provide better extraction for relatively small-sized metal ions than DODGAA. In addition, it was found that C
DGAA tended to provide better extraction for relatively small-sized metal ions than DODGAA. In addition, it was found that C DGAA enables the selective removal of Hg(II) from aqueous solutions containing various divalent metal ions.
DGAA enables the selective removal of Hg(II) from aqueous solutions containing various divalent metal ions.
Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Stora, T.*; Sato, Nozomi*; Kaneya, Yusuke; Tsukada, Kazuaki; D llmann, C. E.*; Eberhardt, K.*; Eliav, E.*; et al.
llmann, C. E.*; Eberhardt, K.*; Eliav, E.*; et al.
EPJ Web of Conferences, 131, p.05001_1 - 05001_6, 2016/12
Times Cited Count:0 Percentile:0.9(Chemistry, Inorganic & Nuclear)Ionization efficiency in a surface ionization process depends on the first ionization potential of the atom. Based on the dependence, the ionization potential of the atom can be determined. We measured ionization efficiencies of fermium, einsteinium, mendelevium, and lawrencium by using a newly developed gas-jet coupled surface ion-source. The ionization potential of the elements have not been determined so far due to their low production rates and/or their short half-lives. Based on a relationship between the ionization efficiency and the ionization potential obtained via measurements of short-lived lanthanide isotopes, the ionization potentials of these actinide elements have been successfully determined.