Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 O
O -dependent iodide oxidation
-dependent iodide oxidationLi, H.-P.*; Yeager, C. M.*; Brinkmeyer, R.*; Zhang, S.*; Ho, Y.-F.*; Xu, C.*; Jones, W. L.*; Schwehr, K. A.*; Otosaka, Shigeyoshi; Roberts, K. A.*; et al.
Environmental Science & Technology, 46(9), p.4837 - 4844, 2012/03
Times Cited Count:58 Percentile:79.09(Engineering, Environmental)In order to develop an understanding of the role that microorganisms play in the transport of  I in soil-water systems, naturally occurring bacteria isolated from the F-area subsurface of the Savannah River Site (SRS) were assessed for iodide oxidizing activity. Spent liquid medium from a number of SRS bacterial cultures enhanced iodide oxidation 2-10 fold in the presence of hydrogen peroxide (H
I in soil-water systems, naturally occurring bacteria isolated from the F-area subsurface of the Savannah River Site (SRS) were assessed for iodide oxidizing activity. Spent liquid medium from a number of SRS bacterial cultures enhanced iodide oxidation 2-10 fold in the presence of hydrogen peroxide (H O
O ). From a time-series measurements of peroxidase activities and organic acid concentrations, it was hypothesized that microbial organic acid exudate promoted iodide oxidation via following mechanisms; (1) organic acids interact with H
). From a time-series measurements of peroxidase activities and organic acid concentrations, it was hypothesized that microbial organic acid exudate promoted iodide oxidation via following mechanisms; (1) organic acids interact with H O
O to form strong iodide oxidizing agents, peroxy carboxylic acids, and (2) organic acid secretion led to enhanced rates of H
to form strong iodide oxidizing agents, peroxy carboxylic acids, and (2) organic acid secretion led to enhanced rates of H O
O -dependent iodide oxidation by lowering the pH of the culture medium.
-dependent iodide oxidation by lowering the pH of the culture medium.
 I) by soil organic matter and possible consequences of the remedial action at Savannah River Site
I) by soil organic matter and possible consequences of the remedial action at Savannah River SiteXu, C.*; Miller, E. J.*; Zhang, S.*; Li, H.-P.*; Ho, Y.-F.*; Schwehr, K. A.*; Kaplan, D. I.*; Otosaka, Shigeyoshi; Roberts, K. A.*; Brinkmeyer, R.*; et al.
Environmental Science & Technology, 45(23), p.9975 - 9983, 2011/12
Times Cited Count:77 Percentile:84.78(Engineering, Environmental)In order to investigate accumulation process of iodine-129 ( I) in a contaminated F-Area groundwater plume of the US Savannah River Site, soil resuspension experiments simulating surface runoff and erosion events were conducted. Results showed that 72
I) in a contaminated F-Area groundwater plume of the US Savannah River Site, soil resuspension experiments simulating surface runoff and erosion events were conducted. Results showed that 72 77% of the newly-introduced iodide was irreversibly sequestered into the organic-rich soil, while the rest was transformed into colloidal and dissolved organo-iodine by the soil. Laboratory iodination of the soil indicated a preferential incorporation of inorganic iodine into soil organic matter (SOM) at acidic pH (3
77% of the newly-introduced iodide was irreversibly sequestered into the organic-rich soil, while the rest was transformed into colloidal and dissolved organo-iodine by the soil. Laboratory iodination of the soil indicated a preferential incorporation of inorganic iodine into soil organic matter (SOM) at acidic pH (3 4), except for the iodination catalyzed by lactoperoxidase, which favors more alkaline conditions. From this result, we concluded that under very acidic conditions, abiotic iodination of SOM was predominant, while under less acidic conditions (pH
4), except for the iodination catalyzed by lactoperoxidase, which favors more alkaline conditions. From this result, we concluded that under very acidic conditions, abiotic iodination of SOM was predominant, while under less acidic conditions (pH  5), microbial enzymatically-assisted iodination took over.
5), microbial enzymatically-assisted iodination took over.
 I) at the Savannah River Site?
I) at the Savannah River Site?Xu, C.*; Zhang, S.*; Ho, Y.-F.*; Miller, E. J.*; Roberts, K. A.*; Li, H.-P.*; Schwehr, K. A.*; Otosaka, Shigeyoshi; Kaplan, D. I.*; Brinkmeyer, R.*; et al.
Geochimica et Cosmochimica Acta, 75(19), p.5716 - 5735, 2011/10
Times Cited Count:65 Percentile:82.83(Geochemistry & Geophysics)In order to understand the effect of soil organic matter (SOM) on the mobility of iodine in the vicinity of the F-area seepage basin at the U.S. Department Energy's Savannah River Site (SRS), relationships between radioiodine/iodine concentration and properties of SOM (e.g., degree of humification, aromaticity, and molecular weight) were examined. Analyses were carried out for four sequential extracts of SOM (freshwater, alkaline, glycerol, and citric-alkaline solutions). Iodine in SOM was selectively bound to a small-size aromatic subunit (less than 10 kDa), and majority of water soluble  I was associated with a low molecular weight amphiphilic organic carrier (13.5-15 kDa). From these results, it was suggested that (1) SOM behaved as a sink as well as a source for iodine at the SRS, and (2) the function of SOM varies with groundwater chemistry.
I was associated with a low molecular weight amphiphilic organic carrier (13.5-15 kDa). From these results, it was suggested that (1) SOM behaved as a sink as well as a source for iodine at the SRS, and (2) the function of SOM varies with groundwater chemistry.
 I and
I and  I species in an acidic groundwater plume at the Savannah River Site
I species in an acidic groundwater plume at the Savannah River SiteOtosaka, Shigeyoshi; Schwehr, K. A.*; Kaplan, D. I.*; Roberts, K. A.*; Zhang, S.*; Xu, C.*; Li, H.-P.*; Ho, Y.-F.*; Brinkmeyer, R.*; Yeager, C. M.*; et al.
Science of the Total Environment, 409(19), p.3857 - 3865, 2011/09
Times Cited Count:70 Percentile:83.00(Environmental Sciences)Spatial distributions of concentrations and speciation of radioiodine ( I) and stable iodine (
I) and stable iodine ( I) in groundwater in the vicinity of the F-area seepage basin at the U.S. Department Energy of Savannah River Site were investigated.
I) in groundwater in the vicinity of the F-area seepage basin at the U.S. Department Energy of Savannah River Site were investigated.  I concentration in groundwater was 8.6 Bq/L immediately downstream of the seepage basin (well FSB-95DR), and decreased with distance from the infiltration basin.
I concentration in groundwater was 8.6 Bq/L immediately downstream of the seepage basin (well FSB-95DR), and decreased with distance from the infiltration basin.  I concentration decreased similarly to that of
I concentration decreased similarly to that of  I. Although there was no potential
I. Although there was no potential  I source in wastes in the basin,
I source in wastes in the basin,  I also showed a similar gradient to that of
I also showed a similar gradient to that of  I. High concentrations of
I. High concentrations of  I or
I or  I were not detected in groundwater collected from wells located outside of the mixed waste plume of this area. The high iodide concentrations in groundwater near the basin were presumed to be caused by dissolution of iodide from soil due to gradually increasing of pH values in the last decade.
I were not detected in groundwater collected from wells located outside of the mixed waste plume of this area. The high iodide concentrations in groundwater near the basin were presumed to be caused by dissolution of iodide from soil due to gradually increasing of pH values in the last decade.
 He,
He,  B,
B,  Y,
Y, 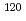 In and
In and 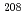 Tl observed in the (
Tl observed in the ( Li,
Li,  Be) charge-exchange reaction
Be) charge-exchange reactionAnnakkage, T.*; J necke, J.*; Winfield, J. S.*; Berg, G. P. A.*; Brown, J. A.*; Gawley, G. A.*; Danczyk, S.*; Fujiwara, Mamoru; Mercer, D. J.*; Pham, K.*; et al.
necke, J.*; Winfield, J. S.*; Berg, G. P. A.*; Brown, J. A.*; Gawley, G. A.*; Danczyk, S.*; Fujiwara, Mamoru; Mercer, D. J.*; Pham, K.*; et al.
Nuclear Physics A, 648(1-2), p.3 - 44, 1999/03
Times Cited Count:22 Percentile:44.59(Physics, Nuclear)no abstracts in English