Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Cho, S. H.*; Cho, S. W.*; Lv, Z.*; Sekine, Yurina; Liu, S.*; Zhou, M.*; Nuxoll, R. F.*; Kanatzidis, E. E.*; Ghaffari, R.*; Kim, D.*; et al.
Lab on a Chip, 9 Pages, 2025/00
Times Cited Count:0 Percentile:0.00(Biochemical Research Methods)Amino acids are essential for protein synthesis and metabolic processes in support of homeostatic balance and healthy body functions. This study quantitatively investigates eccrine sweat as a significant channel for loss of amino acids during exercise, to improve an understanding of amino acid turnover and to provide feedback to users on the need for supplement intake. The measurement platform consists of a soft, skin-interfaced microfluidic system for real-time analysis of amino acid content in eccrine sweat. This system relies on integrated fluorometric assays and smartphone-based imaging techniques for quantitative analysis.
Yang, D. S.*; Wu, Y.*; Kanatzidis, E. E.*; Avila, R.*; Zhou, M.*; Bai, Y.*; Chen, S.*; Sekine, Yurina; Kim, J.*; Deng, Y.*; et al.
Materials Horizons, 10(11), p.4992 - 5003, 2023/09
Times Cited Count:12 Percentile:78.05(Chemistry, Multidisciplinary)This paper presents a set of findings that enhances the performance of these systems through the use of microfluidic networks, integrated valves and microscale optical cuvettes formed by three-dimensional printing in hard/soft hybrid materials systems, for accurate spectroscopic and fluorometric assays. Field studies demonstrate the capability of these microcuvette systems to evaluate the concentrations of copper, chloride, and glucose in sweat, along with the sweat pH, with laboratory grade accuracy and sensitivity.
Liu, S.*; Yang, D. S.*; Wang, S.*; Luan, H.*; Sekine, Yurina; Model, J. B.*; Aranyosi, A. J.*; Ghaffari, R.*; Rogers, J. A.*
EcoMat (Internet), 5(1), p.e12270_1 - e12270_18, 2023/01
Times Cited Count:40 Percentile:92.61(Chemistry, Physical)Advanced capabilities in noninvasive, in situ monitoring of sweat serve as the basis for obtaining real-time insights into human physiological state, health, and performance. Although recently reported microfluidic systems support powerful functions, most are designed as single-use disposables. Here, we introduce materials and molding techniques that bypass these concerns through biodegradable microfluidic systems with a full range of features. The key components fully degrade through the enzymatic action of microorganisms in natural soil environments. Detailed characterization of the device reveal a set of essential performance parameters that are comparable to, or even better than, those of non-degradable counterparts. Human subject studies illustrate the ability of these devices to acquire accurate measurements of sweat loss, sweat rate, pH, and chloride concentration.
Kim, S.*; Lee, B.*; Reeder, J. T.*; Seo, S. H.*; Lee, S.-U.*; Hourlier-Fargette, A.*; Shin, J.*; Sekine, Yurina; Jeong, H.*; Oh, Y. S.*; et al.
Proceedings of the National Academy of Sciences of the United States of America, 117(45), p.27906 - 27915, 2020/11
Times Cited Count:109 Percentile:94.20(Multidisciplinary Sciences)In this study, we present a wireless, battery-free, skin-interfaced microfluidic system that combines lateral flow immunoassay for sweat cortisol assay, fluorometric imaging of glucose and ascorbic acid (vitamin C) assays, and digital tracking of sweat rate using electrodes that measure skin galvanic response. Systematic benchtop testing and on-body field studies on human subjects exercising in a gym environment highlight the key multifunctional features of this platform in tracking the biochemical correlates of physical stress.
Zhang, Y.*; Guo, H.*; Kim, S. B.*; Wu, Y.*; Ostojich, D.*; Park, S. H.*; Wang, X.*; Weng, Z.*; Li, R.*; Bandodkar, A. J.*; et al.
Lab on a Chip, 19(9), p.1545 - 1555, 2019/05
Times Cited Count:189 Percentile:99.70(Biochemical Research Methods)This paper introduces two important advances in recently reported classes of soft, skin-interfaced microfluidic systems for sweat capture and analysis: (1) a simple, broadly applicable means for collection of sweat that bypasses requirements for physical/mental exertion or pharmacological stimulation and (2) a set of enzymatic chemistries and colorimetric readout approaches for determining the concentrations of creatinine and urea in sweat, across physiologically relevant ranges. The results allow for routine, non-pharmacological capture of sweat across patient populations, such as infants and the elderly, that cannot be expected to sweat through exercise, and they create potential opportunities in the use of sweat for kidney disease screening/monitoring.
Bandodkar, A. J.*; Gutruf, P.*; Choi, J.*; Lee, K.-H.*; Sekine, Yurina; Reeder, J. T.*; Jeang, W. J.*; Aranyosi, A. J.*; Lee, S. P.*; Model, J. B.*; et al.
Science Advances (Internet), 5(1), p.eaav3294_1 - eaav3294_15, 2019/01
Times Cited Count:566 Percentile:99.88(Multidisciplinary Sciences)Interest in advanced wearable technologies increasingly extends beyond systems for biophysical measurements to those that enable continuous, non-invasive monitoring of biochemical markers in biofluids. Here, we introduce battery-free, wireless microelectronic platforms that perform sensing via schemes inspired by the operation of biofuel cells. Combining these systems in a magnetically releasable manner with chrono-sampling microfluidic networks that incorporate assays based on colorimetric sensing yields thin, flexible, lightweight, skin-interfaced technologies with broad functionality in sweat analysis. A demonstration device allows simultaneous monitoring of sweat rate/loss, along with quantitative measurements of pH and of lactate, glucose and chloride concentrations using biofuel cell and colorimetric approaches.
Kim, S. B.*; Lee, K.-H.*; Raj, M. S.*; Reeder, J. T.*; Koo, J.*; Hourlier-Fargette, A.*; Bandodkar, A. J.*; Won, S. M.*; Sekine, Yurina; Choi, J.*; et al.
Small, 14(45), p.1802876_1 - 1802876_9, 2018/11
Times Cited Count:95 Percentile:94.03(Chemistry, Multidisciplinary)Excretion of sweat from eccrine glands is a dynamic physiological process that varies with body position, activity level, and health status. Information content embodied in sweat rate and chemistry can be used to assess health status and athletic performance. This paper presents a thin, miniaturized, skin-interfaced microfluidic technology that includes a reusable, battery-free electronics module for measuring sweat conductivity and rate in real-time using wireless power from and data communication with capabilities in near field communications (NFC). Systematic studies of these combined microfluidic/electronic systems, accurate correlations of measurements performed with them to those of laboratory standard instrumentation, and field tests on human subjects establish the key operational features and their utility in sweat analytics.
 quantitative analysis of sweat chemistry
quantitative analysis of sweat chemistrySekine, Yurina; Kim, S. B.*; Zhang, Y.*; Bandodkar, A. J.*; Xu, S.*; Choi, J.*; Irie, Masahiro*; Ray, T. R.*; Kohli, P.*; Kozai, Naofumi; et al.
Lab on a Chip, 18(15), p.2178 - 2186, 2018/08
Times Cited Count:185 Percentile:99.61(Biochemical Research Methods)The rich composition of solutes and metabolites in sweat and its relative ease of collection upon excretion from skin pores make this class of biofluid an attractive candidate for point of care analysis. Here, we present a complementary approach that exploits fluorometric sensing modalities integrated into a soft, skin-interfaced microfluidic system which, when paired with a simple smartphone-based imaging module, allows for in-situ measurement of important biomarkers in sweat. A network array of microchannels and a collection of microreservoirs pre-filled with fluorescent probes that selectively react with target analytes in sweat (e.g. probes), enable quantitative, rapid analysis. Field studies on human subjects demonstrate the ability to measure the concentrations of chloride, sodium and zinc in sweat, with accuracy that matches that of conventional laboratory techniques.
Kim, S. B.*; Zhang, Y.*; Won, S. M.*; Bandodkar, A. J.*; Sekine, Yurina; Xue, Y.*; Koo, J.*; Harshman, S. W.*; Martin, J. A.*; Park, J. M.*; et al.
Small, 14(12), p.1703334_1 - 1703334_11, 2018/03
Times Cited Count:128 Percentile:96.23(Chemistry, Multidisciplinary)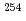 No
NoLopez-Martens, A.*; Henning, G.*; Khoo, T. L.*; Seweryniak, D.*; Alcorta, M.*; Asai, Masato; Back, B. B.*; Bertone, P. F.*; Boilley, D.*; Carpenter, M. P.*; et al.
EPJ Web of Conferences, 131, p.03001_1 - 03001_6, 2016/12
Times Cited Count:1 Percentile:41.81(Chemistry, Inorganic & Nuclear)Fission barrier height and its angular-momentum dependence have been measured for the first time in the nucleus with the atomic number greater than 100. The entry distribution method, which can determine the excitation energy at which fission starts to dominate the decay process, was applied to 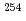 No. The fission barrier of
No. The fission barrier of 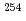 No was found to be 6.6 MeV at zero spin, indicating that the
No was found to be 6.6 MeV at zero spin, indicating that the 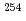 No is strongly stabilized by the nuclear shell effects.
No is strongly stabilized by the nuclear shell effects.
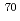 Ni
NiChiara, C. J.*; Weisshaar, D.*; Janssens, R. V. F.*; Tsunoda, Yusuke*; Otsuka, Takaharu*; Harker, J. L.*; Walters, W. B.*; Recchia, F.*; Albers, M.*; Alcorta, M.*; et al.
Physical Review C, 91(4), p.044309_1 - 044309_10, 2015/04
Times Cited Count:40 Percentile:90.62(Physics, Nuclear)The neutron-rich isotope 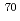 Ni was produced by multi-nucleon transfer reactions of
Ni was produced by multi-nucleon transfer reactions of 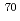 Zn in the Argonne National Laboratory, and an in-beam
Zn in the Argonne National Laboratory, and an in-beam  -ray experiment were performed using the GRETINA array. The
-ray experiment were performed using the GRETINA array. The 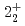 and
and 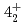 levels of
levels of 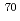 Ni were observed for the first time. Those levels are regarded as large deformed states associated with proton excitation from the
Ni were observed for the first time. Those levels are regarded as large deformed states associated with proton excitation from the 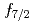 orbit because they cannot be reproduced by a shell-model calculation assuming a small valence space without
orbit because they cannot be reproduced by a shell-model calculation assuming a small valence space without 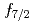 . A theoretical analysis based on the Monte Carlo shell model published in 2014 indicates that those levels corresponds to a prolate deformed band. The present result demonstrates the occurrence of shape coexistence in neutron-rich Ni isotopes other than a known case of
. A theoretical analysis based on the Monte Carlo shell model published in 2014 indicates that those levels corresponds to a prolate deformed band. The present result demonstrates the occurrence of shape coexistence in neutron-rich Ni isotopes other than a known case of 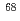 Ni, and confirms the predictive power of the Monte Carlo shell-model calculation.
Ni, and confirms the predictive power of the Monte Carlo shell-model calculation.
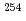 No and
No and 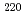 Th
ThHenning, G.*; Khoo, T. L.*; Lopez-Martens, A.*; Seweryniak, D.*; Alcorta, M.*; Asai, Masato; Back, B. B.*; Bertone, P. F.*; Boilley, D.*; Carpenter, M. P.*; et al.
Physical Review Letters, 113(26), p.262505_1 - 262505_6, 2014/12
Times Cited Count:34 Percentile:81.70(Physics, Multidisciplinary)Fission barrier heights of a shell-stabilized superheavy nucleus 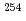 No have been determined as a function of spin up to 19
No have been determined as a function of spin up to 19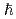 through the measured distribution of entry points of
through the measured distribution of entry points of  deexcitations in the excitation energy vs. spin plane. The fission barrier height of
deexcitations in the excitation energy vs. spin plane. The fission barrier height of 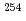 No was determined to be 6.0 MeV at spin 15
No was determined to be 6.0 MeV at spin 15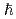 , and 6.6 MeV at spin 0
, and 6.6 MeV at spin 0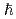 by extrapolation. This demonstrates that the shell effect actually enlarges the fission barrier in such heavy nuclei and keeps the barrier high even at high spin.
by extrapolation. This demonstrates that the shell effect actually enlarges the fission barrier in such heavy nuclei and keeps the barrier high even at high spin.
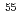 Ni hole-states from (p,d) transfer reactions
Ni hole-states from (p,d) transfer reactionsSanetullaev, A.*; Tsang, M. B.*; Lynch, W. G.*; Lee, J.*; Bazin, D.*; Chan, K. P.*; Coupland, D.*; Hanzl, V.*; Hanzlova, D.*; Kilburn, M.*; et al.
Physics Letters B, 736, p.137 - 141, 2014/09
Times Cited Count:17 Percentile:69.17(Astronomy & Astrophysics)no abstracts in English
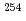 No
NoHenning, G.*; Lopez-Martens, A.*; Khoo, T. L.*; Seweryniak, D.*; Alcorta, M.*; Asai, Masato; Back, B. B.*; Bertone, P. F.*; Boilley, D.*; Carpenter, M. P.*; et al.
EPJ Web of Conferences, 66, p.02046_1 - 02046_8, 2014/03
Times Cited Count:3 Percentile:68.16(Physics, Nuclear)Fission barrier heights of 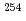 No have been determined through the entry distribution method. The entry distribution is the initial distribution of excitation energy and spin from which the
No have been determined through the entry distribution method. The entry distribution is the initial distribution of excitation energy and spin from which the  deexcitation starts in the fusion-evaporation reaction. The initial distribution is extracted from measured
deexcitation starts in the fusion-evaporation reaction. The initial distribution is extracted from measured  -ray multiplicity and total
-ray multiplicity and total  -ray energy. This paper describes the details of the entry distribution method, and reports the first determination of the fission barrier heights of
-ray energy. This paper describes the details of the entry distribution method, and reports the first determination of the fission barrier heights of 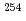 No, which is the heaviest nucleus whose fission barrier has been measured.
No, which is the heaviest nucleus whose fission barrier has been measured.