Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Bh in the
Bh in the  Cm(
Cm(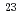 Na,5
Na,5 )
) Bh reaction and its decay properties
Bh reaction and its decay propertiesHaba, Hiromitsu*; Fan, F.*; Kaji, Daiya*; Kasamatsu, Yoshitaka*; Kikunaga, Hidetoshi*; Komori, Yukiko*; Kondo, Narumi*; Kudo, Hisaaki*; Morimoto, Koji*; Morita, Kosuke*; et al.
Physical Review C, 102(2), p.024625_1 - 024625_12, 2020/08
Times Cited Count:7 Percentile:55.09(Physics, Nuclear)Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Beerwerth, R.*; Kaneya, Yusuke*; Makii, Hiroyuki; Mitsukai, Akina*; Nagame, Yuichiro; Osa, Akihiko; Toyoshima, Atsushi; et al.
Journal of the American Chemical Society, 140(44), p.14609 - 14613, 2018/11
Times Cited Count:32 Percentile:69.40(Chemistry, Multidisciplinary)The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
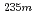 U oxide and fluoride
U oxide and fluorideShigekawa, Yudai*; Kasamatsu, Yoshitaka*; Yasuda, Yuki*; Kaneko, Masashi; Watanabe, Masayuki; Shinohara, Atsushi*
Physical Review C, 98(1), p.014306_1 - 014306_5, 2018/07
Times Cited Count:4 Percentile:32.05(Physics, Nuclear)The nuclear half-life of 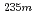 U has been reported to vary depending on the chemical environment. In this study, both the half-life and the internal-conversion (IC) electron energy spectrum were measured for
U has been reported to vary depending on the chemical environment. In this study, both the half-life and the internal-conversion (IC) electron energy spectrum were measured for 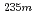 U with identical chemical environments for the first time.
U with identical chemical environments for the first time. 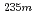 U oxide and fluoride samples were subjected to these measurements, and clear differences in the half-life and the energy spectrum between these samples were observed. The peaks in the energy spectra were identified with the relativistic density functional theory calculation, and the molecular orbital states of the
U oxide and fluoride samples were subjected to these measurements, and clear differences in the half-life and the energy spectrum between these samples were observed. The peaks in the energy spectra were identified with the relativistic density functional theory calculation, and the molecular orbital states of the 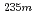 U oxide and fluoride estimated from the energy spectra and the calculation qualitatively explained the difference in the half-lives between the samples.
U oxide and fluoride estimated from the energy spectra and the calculation qualitatively explained the difference in the half-lives between the samples.
Oe, Kazuhiro*; Attallah, M. F.*; Asai, Masato; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke*; Kasamatsu, Yoshitaka*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1317 - 1320, 2015/02
Times Cited Count:10 Percentile:60.46(Chemistry, Analytical)A new technique for continuous dissolution of nuclear reaction products transported by a gas-jet system was developed for superheavy element (SHE) chemistry. In this technique, a hydrophobic membrane is utilized to separate an aqueous phase from the gas phase. With this technique, the dissolution efficiencies of short-lived radionuclides of 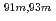 Mo and
Mo and 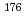 W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
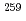 Lr
LrAsai, Masato; Kamada, Hiroki*; Shigekawa, Yudai*; Tsukada, Kazuaki; Sato, Tetsuya; Toyoshima, Atsushi; Mitsukai, Akina; Nagame, Yuichiro; Nishio, Katsuhisa; Hirose, Kentaro; et al.
no journal, ,
To clarify the fission mechanism of mass-symmetric spontaneous fissions observed around the neutron-rich Fm region, we have measured kinetic energies and mass distribution of spontaneous-fission fragments in this nuclear region precisely. The experiment was performed at the JAEA tandem accelerator facility using an on-line isotope separator (ISOL). The mass-separated 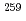 Lr nuclei were implanted into thin foil, and two fragments of their spontaneous fission were coincidently measured with two Si detectors. The spontaneous fission of
Lr nuclei were implanted into thin foil, and two fragments of their spontaneous fission were coincidently measured with two Si detectors. The spontaneous fission of 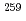 Lr was found to show a narrow symmetric mass distribution. On the other hand, their total kinetic energy TKE was found to be low, which is apparently different from the high-TKE symmetric fissions observed in the Fm isotopes. This indicates that the fission mechanisms of these two symmetric fissions are different.
Lr was found to show a narrow symmetric mass distribution. On the other hand, their total kinetic energy TKE was found to be low, which is apparently different from the high-TKE symmetric fissions observed in the Fm isotopes. This indicates that the fission mechanisms of these two symmetric fissions are different.
Sato, Tetsuya; Kaneya, Yusuke*; Asai, Masato; Tsukada, Kazuaki; Toyoshima, Atsushi; Mitsukai, Akina*; Osa, Akihiko; Makii, Hiroyuki; Hirose, Kentaro; Nagame, Yuichiro; et al.
no journal, ,
Our experimental results on the first ionization potential measurement of lawrencium (Lr, element 103) have strongly suggested that the Lr atom has a [Rn]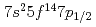 configuration as a result of the influence of strong relativistic effects. The configuration is different from that expected from the lanthanide homologue, lutetium (Lu). According to a semi-empirical consideration, it is expected that the change of the electronic configuration leads higher volatility of Lr than that of Lu. In this work, adsorption behaviors of Lr and various short-lived rare earth isotopes on a tantalum surface were investigated via observation of their surface ionization efficiencies. It was found that Lr would behave like low volatile rare earth elements such as Lu contrary to the semi-empirical expectation.
configuration as a result of the influence of strong relativistic effects. The configuration is different from that expected from the lanthanide homologue, lutetium (Lu). According to a semi-empirical consideration, it is expected that the change of the electronic configuration leads higher volatility of Lr than that of Lu. In this work, adsorption behaviors of Lr and various short-lived rare earth isotopes on a tantalum surface were investigated via observation of their surface ionization efficiencies. It was found that Lr would behave like low volatile rare earth elements such as Lu contrary to the semi-empirical expectation.
Sato, Tetsuya; Asai, Masato; Kaneya, Yusuke*; Tsukada, Kazuaki; Toyoshima, Atsushi; Mitsukai, Akina*; Takeda, Shinsaku*; Vascon, A.*; Sakama, Minoru*; Sato, Daisuke*; et al.
no journal, ,
The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
Kaneya, Yusuke*; Tomitsuka, Tomohiro; Sato, Tetsuya; Asai, Masato; Tsukada, Kazuaki; Toyoshima, Atsushi; Mitsukai, Akina; Makii, Hiroyuki; Hirose, Kentaro; Osa, Akihiko; et al.
no journal, ,
Toyoshima, Atsushi; Miyashita, Sunao*; Oe, Kazuhiro*; Kitayama, Yuta*; Lerum, H. V.*; Goto, Naoya*; Kaneya, Yusuke; Komori, Yukiko*; Mitsukai, Akina*; Vascon, A.; et al.
no journal, ,
no abstracts in English
Nagame, Yuichiro; Sato, Tetsuya; Asai, Masato; Kaneya, Yusuke*; Makii, Hiroyuki; Mitsukai, Akina; Osa, Akihiko; Sch del, M.*; Toyoshima, Atsushi; Tsukada, Kazuaki; et al.
del, M.*; Toyoshima, Atsushi; Tsukada, Kazuaki; et al.
no journal, ,
Kaneya, Yusuke*; Asai, Masato; Sato, Tetsuya; Tomitsuka, Tomohiro; Tsukada, Kazuaki; Toyoshima, Atsushi; Mitsukai, Akina; Makii, Hiroyuki; Hirose, Kentaro; Osa, Akihiko; et al.
no journal, ,
To study the influence of the valence 7p electronic orbital on chemical properties of lawrencium, a measurement of the adsorption enthalpy of lawrencium was carried out. A new method using a surface ionization technique coupled to an on-line isotope separator was developed, which enabled one to measure temperature dependence of lawrencium surface adsorption on a metallic tantalum surface at high temperature up to 2800 K. The temperature dependences of adsorption of lawrencium as well as various lanthanide elements were investigated with this method, and the adsorption enthalpy of lawrencium was successfully extracted.
electronic orbital on chemical properties of lawrencium, a measurement of the adsorption enthalpy of lawrencium was carried out. A new method using a surface ionization technique coupled to an on-line isotope separator was developed, which enabled one to measure temperature dependence of lawrencium surface adsorption on a metallic tantalum surface at high temperature up to 2800 K. The temperature dependences of adsorption of lawrencium as well as various lanthanide elements were investigated with this method, and the adsorption enthalpy of lawrencium was successfully extracted.
Sato, Tetsuya; Kaneya, Yusuke*; Asai, Masato; Tsukada, Kazuaki; Toyoshima, Atsushi; Mitsukai, Akina*; Osa, Akihiko; Makii, Hiroyuki; Hirose, Kentaro; Nagame, Yuichiro; et al.
no journal, ,
Our experimental results on the first ionization potential measurement of lawrencium (Lr, element 103) have strongly suggested that the Lr atom has a [Rn]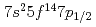 configuration as a result of the influence of strong relativistic effects. The configuration is different from that expected from the lanthanide homologue, lutetium (Lu). According to a semi-empirical consideration, it is expected that the change of the electronic configuration leads higher volatility of Lr than that of Lu. In this work, adsorption behaviors of Lr and various short-lived rare earth isotopes on a tantalum surface were investigated via observation of their surface ionization efficiencies. It was found that Lr would behave like low volatile rare earth elements such as Lu contrary to the semi-empirical expectation.
configuration as a result of the influence of strong relativistic effects. The configuration is different from that expected from the lanthanide homologue, lutetium (Lu). According to a semi-empirical consideration, it is expected that the change of the electronic configuration leads higher volatility of Lr than that of Lu. In this work, adsorption behaviors of Lr and various short-lived rare earth isotopes on a tantalum surface were investigated via observation of their surface ionization efficiencies. It was found that Lr would behave like low volatile rare earth elements such as Lu contrary to the semi-empirical expectation.
Toyoshima, Atsushi; Asai, Masato; Attallah, M. F.*; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke; Kasamatsu, Yoshitaka*; et al.
no journal, ,
Towards electrolytic reduction of Sg, batch-wise electrolytic reduction of carrier-free  Mo and
Mo and  W radiotracers was studied using a flow electrolytic column (FEC). The electrolyzed samples from a FEC were chemically analyzed by solvent extraction with TOA and HDEHP to separate and identify reduced species from the stable Mo(VI) and W(VI) ones based on their different extraction behavior.
W radiotracers was studied using a flow electrolytic column (FEC). The electrolyzed samples from a FEC were chemically analyzed by solvent extraction with TOA and HDEHP to separate and identify reduced species from the stable Mo(VI) and W(VI) ones based on their different extraction behavior.  Mo and
Mo and  W were applied as radiotracers. We also performed cyclic voltammetry and UV/Vis absorption spectrometry of macro amounts of Mo and W in acidic solutions to obtain information on redox reactions of these elements under given conditions. In the conference, the present status of the preparatory reduction experiments with Mo and W will be presented.
W were applied as radiotracers. We also performed cyclic voltammetry and UV/Vis absorption spectrometry of macro amounts of Mo and W in acidic solutions to obtain information on redox reactions of these elements under given conditions. In the conference, the present status of the preparatory reduction experiments with Mo and W will be presented.
Toyoshima, Atsushi; Oe, Kazuhiro*; Asai, Masato; Attallah, M. F.*; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Kaneko, Masashi*; Kaneya, Yusuke; Kasamatsu, Yoshitaka*; et al.
no journal, ,
Due to short half-lives less than 10 s and extremely low production rates, transactinide elements heavier than seaborgium (Sg) are produced on an atom per hour scale. Therefore, a continuous rapid chemistry assembly is required to study aqueous-phase chemistry of these heaviest elements. In the present study, we started developments of a continuous chemistry assembly. Our first attempt was made in on-line experiments with Mo and W, lighter homologs of Sg, to optimize a chemistry assembly consisting of a newly developed membrane degasser as an interface between gas-jet and aqueous phase, a flow electrolytic column apparatus utilized to control oxidation states of Mo and W ions, and the continuous liquid-liquid extraction apparatus of SISAK for separation. In the conference, present status of the developments will be presented.
Sato, Tetsuya; Kaneya, Yusuke*; Asai, Masato; Tsukada, Kazuaki; Toyoshima, Atsushi; Mitsukai, Akina*; Osa, Akihiko; Makii, Hiroyuki; Nishio, Katsuhisa; Hirose, Kentaro; et al.
no journal, ,
The ground state electronic configuration of lawrencium (Lr, Z =103) is predicted to be [Rn]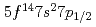 , which is different from that of the lanthanide homolog Lu [Xe]
, which is different from that of the lanthanide homolog Lu [Xe]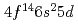 due to strong relativistic effects. According to semi-empirical considerations, volatility of Lr is expected to be higher than that of Lu. We have investigated adsorption behavior of
due to strong relativistic effects. According to semi-empirical considerations, volatility of Lr is expected to be higher than that of Lu. We have investigated adsorption behavior of  Lr, which was produced in the reaction of
Lr, which was produced in the reaction of  Cf(
Cf(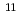 B, 4n), on a tantalum (Ta) metal surface using a surface ion-source installed into the isotope separator on-line (ISOL) at the JAEA tandem accelerator facility. The observed adsorption behavior of
B, 4n), on a tantalum (Ta) metal surface using a surface ion-source installed into the isotope separator on-line (ISOL) at the JAEA tandem accelerator facility. The observed adsorption behavior of  Lr was similar to those of Tb and Lu which have relatively higher adsorption enthalpy on Ta surface. It implies that Lr would have low volatility like such as Lu and Tb.
Lr was similar to those of Tb and Lu which have relatively higher adsorption enthalpy on Ta surface. It implies that Lr would have low volatility like such as Lu and Tb.
 solution
solutionKato, Mizuho*; Adachi, Sadia*; Toyoshima, Atsushi*; Tsukada, Kazuaki; Asai, Masato; Haba, Hiromitsu*; Yokokita, Takuya*; Komori, Yukiko*; Shigekawa, Yudai*; Sueki, Keisuke*
no journal, ,
A few studies have been so far performed on aqueous chemistry of Db, the group-5 superheavy element, by anion-exchange experiments in HF and HF/HNO mixture solutions. Although the distribution coefficient (Kd) of Db was shown to trend to Ta
mixture solutions. Although the distribution coefficient (Kd) of Db was shown to trend to Ta  Nb
Nb  Db
Db  Pa in the group-5 homologs and pseudo-homolog elements, its chemical species has not been clarified. In this work, for identification of fluoride species of Db, we performed anion-exchange experiments of Nb, Ta, and Db in HF/1.0 M HNO
Pa in the group-5 homologs and pseudo-homolog elements, its chemical species has not been clarified. In this work, for identification of fluoride species of Db, we performed anion-exchange experiments of Nb, Ta, and Db in HF/1.0 M HNO mixture solutions by using an automated rapid chemistry apparatus, ARCA.
mixture solutions by using an automated rapid chemistry apparatus, ARCA.
Sato, Tetsuya; Asai, Masato; Kaneya, Yusuke*; Tsukada, Kazuaki; Toyoshima, Atsushi; Mitsukai, Akina*; Takeda, Shinsaku*; Vascon, A.*; Sakama, Minoru*; Sato, Daisuke*; et al.
no journal, ,
The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.