Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 -bis(thiocyanato)aurate(I) complexes in ionic liquids
-bis(thiocyanato)aurate(I) complexes in ionic liquidsAoyagi, Noboru; Shinha, Yusuke*; Ikeda, Atsushi*; Haga, Yoshinori; Shimojo, Kojiro; Brooks, N. R.*; Izuoka, Akira*; Naganawa, Hirochika; Kimura, Takaumi; Binnemans, K.*
Crystal Growth & Design, 15(3), p.1422 - 1429, 2015/03
Times Cited Count:11 Percentile:64.34(Chemistry, Multidisciplinary)The photochemistry of a gold(I) thiocyanate complex has been investigated to determine the coordination structure in both the solid and liquid states. The coordination geometries of the supramolecular complex and the concomitant exciplex have mainly been analyzed by crystallographic analysis and X-ray absorption spectroscopy. The Au-S bond distance and Au-Au separation of the compound in the ground state and in the phosphorescent excited state were compared. Upon irradiation with UV light, the excimeric interactions were enhanced, resulting in a contraction of the Au-Au aurophilic distance. A broad luminescence spectrum was observed for the one-dimensional chain suprastructure. The time-resolved luminescence spectra indicated the entity of several oligomeric species in the crude liquid without neutral solvent molecules. In addition, EXAFS spectroscopy exhibited a slight change in the nearest Au-S distance due to the photo-switched transformation. The deformation of the (Au-Au)* exciplexes was not apparently promoted in the liquid state with the asymmetrical imidazoium cations having a non-local charge distribution in the present observation. This is plausibly due to a flexible molecular structure and a property in the liquid state. In conclusion, the photo-excited nature of the pseudo-one dimensional complexes is emerged in controlling bond distances among the supramolecular networks.
Shinha, Yusuke; Aoyagi, Noboru; Shimojo, Kojiro; Naganawa, Hirochika; Izuoka, Akira*
no journal, ,
no abstracts in English
Shinha, Yusuke; Aoyagi, Noboru; Shimojo, Kojiro; Naganawa, Hirochika; Izuoka, Akira*
no journal, ,
no abstracts in English
Nagaishi, Ryuji; Aoyagi, Noboru; Shinha, Yusuke; Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*
no journal, ,
no abstracts in English
Nagaishi, Ryuji; Aoyagi, Noboru; Shinha, Yusuke; Taguchi, Mitsumasa; Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*
no journal, ,
Radiolytic reactions in ionic liquids of thiocyanate salts were studied by means of the nano-second pulse radiolysis technique. One of the significant ionization sites of liquids was found out to be thiocyanate ion by observing direct formation of its dimer radical anion, not via the diffusion reaction between thiocyanate radical and ion. The formation and reaction behaviors of solvated electron and dimer were examined by changing the composition of liquids and by adding electron scavengers to the liquid.
Aoyagi, Noboru; Shinha, Yusuke; Shimojo, Kojiro; Naganawa, Hirochika; Izuoka, Akira*
no journal, ,
no abstracts in English
Shinha, Yusuke; Aoyagi, Noboru; Shimojo, Kojiro; Naganawa, Hirochika; Izuoka, Akira*
no journal, ,
no abstracts in English
Nagaishi, Ryuji; Aoyagi, Noboru; Shinha, Yusuke; Taguchi, Mitsumasa; Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*
no journal, ,
Radiolytic reactions in ionic liquids of thiocyanate salts were studied by means of the nano-second pulse radiolysis technique. One of the significant ionization sites of liquids was found out to be thiocyanate ion by observing direct formation of its dimer radical ion, not 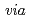 the diffusive encounter between thiocyanate radical and ion. The formation and reaction behaviors of solvated electron and dimer were examined by changing the composition of liquids and by adding electron scavengers to the liquid.
the diffusive encounter between thiocyanate radical and ion. The formation and reaction behaviors of solvated electron and dimer were examined by changing the composition of liquids and by adding electron scavengers to the liquid.
Nagaishi, Ryuji; Aoyagi, Noboru; Shinha, Yusuke; Taguchi, Mitsumasa; Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*
no journal, ,
Radiolytic behavior of ionic liquids of thiocyanate salts was studied by using the ns-pulse radiolysis technique. The transient absorption of thiocyante dimer radical ion was observed in the liquids, and the rise was within the width of electron pulse. The maximum absorbance of dimer in the liquids was equivalent to that in aqueous solutions, where the dimer is formed from the reaction of thiocyanate ion with OH, and increased linearly with increasing the thiocyanate concentration in the liquids. These indicate that one of the ionization sites of liquids should be thiocyanate ion.
Shinha, Yusuke; Aoyagi, Noboru; Shimojo, Kojiro; Izuoka, Akira*; Naganawa, Hirochika
no journal, ,
no abstracts in English
Aoyagi, Noboru; Shinha, Yusuke; Ikeda, Atsushi; Shimojo, Kojiro; Naganawa, Hirochika; Izuoka, Akira*
no journal, ,
no abstracts in English
Aoyagi, Noboru; Shinha, Yusuke*; Ikeda, Atsushi*; Haga, Yoshinori; Shimojo, Kojiro; Watanabe, Masayuki; Brooks, N. R.*; Izuoka, Akira*; Naganawa, Hirochika; Binnemans, K.*; et al.
no journal, ,
no abstracts in English