Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kitazato, Kohei*; Milliken, R. E.*; Iwata, Takahiro*; Abe, Masanao*; Otake, Makiko*; Matsuura, Shuji*; Takagi, Yasuhiko*; Nakamura, Tomoki*; Hiroi, Takahiro*; Matsuoka, Moe*; et al.
Nature Astronomy (Internet), 5(3), p.246 - 250, 2021/03
Times Cited Count:43 Percentile:96.93(Astronomy & Astrophysics)Here we report observations of Ryugu's subsurface material by the Near-Infrared Spectrometer (NIRS3) on the Hayabusa2 spacecraft. Reflectance spectra of excavated material exhibit a hydroxyl (OH) absorption feature that is slightly stronger and peak-shifted compared with that observed for the surface, indicating that space weathering and/or radiative heating have caused subtle spectral changes in the uppermost surface. However, the strength and shape of the OH feature still suggests that the subsurface material experienced heating above 300  C, similar to the surface. In contrast, thermophysical modeling indicates that radiative heating does not increase the temperature above 200
C, similar to the surface. In contrast, thermophysical modeling indicates that radiative heating does not increase the temperature above 200  C at the estimated excavation depth of 1 m, even if the semimajor axis is reduced to 0.344 au. This supports the hypothesis that primary thermal alteration occurred due to radiogenic and/or impact heating on Ryugu's parent body.
C at the estimated excavation depth of 1 m, even if the semimajor axis is reduced to 0.344 au. This supports the hypothesis that primary thermal alteration occurred due to radiogenic and/or impact heating on Ryugu's parent body.
Yamasaki, Shinya; Shirai, Osamu*; Kano, Kenji*; Kozai, Naofumi; Sakamoto, Fuminori; Onuki, Toshihiko
Chemistry Letters, 42(8), p.819 - 821, 2013/08
Times Cited Count:5 Percentile:24.26(Chemistry, Multidisciplinary)The adsorption behavior of lanthanide ions (except for Pm) on liposomes composed of phosphatidylcholine and cholesterol was examined to understand the interaction between lanthanide ions and the phosphoryl moiety of phospholipids. The adsorption of lanthanide ions increased with an increase in pH under the weakly acidic conditions. Selective adsorption with the local maximum at the Er ion and local minimum at the Er
ion and local minimum at the Er ion was observed, similar to the selective adsorption of the bacterial cell surface but different from that of orthophosphates. These results indicate that the adsorption of lanthanide on the phospholipid does not result from simple adsorption on orthophosphate functional groups but by the composition and molecular structure of the phospholipid. Our results strongly suggest that liposomes can be used as a simple biomembrane model without any biological activity for the study of adsorption of lanthanide ions.
ion was observed, similar to the selective adsorption of the bacterial cell surface but different from that of orthophosphates. These results indicate that the adsorption of lanthanide on the phospholipid does not result from simple adsorption on orthophosphate functional groups but by the composition and molecular structure of the phospholipid. Our results strongly suggest that liposomes can be used as a simple biomembrane model without any biological activity for the study of adsorption of lanthanide ions.
Shirai, Osamu*; Onuki, Toshihiko; Kitazumi, Yuki*; Kano, Kenji*
Electroanalysis, 25(8), p.1823 - 1826, 2013/08
Times Cited Count:5 Percentile:18.12(Chemistry, Analytical)The ion transport current was clearly observed across a bilayer lipid membrane (BLM) between two aqueous phases (W1, W2) containing 0.1 M CsCl. In addition, the current density at a given potential between W1 and W2 increased using I instead of Cl as the counter ion. Our results strongly suggest that the cell membrane transport is partially responsible for the internal exposure of 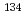 Cs and
Cs and  Cs.
Cs.
Shirai, Osamu*; Ozaki, Shunsuke*; Onishi, Jun*; Kozai, Naofumi; Onuki, Toshihiko; Kano, Kenji*
Chemistry Letters, 38(11), p.1038 - 1039, 2009/11
Times Cited Count:6 Percentile:29.42(Chemistry, Multidisciplinary)Potential-controlled electrolysis was performed between two aqueous phases containing 0.1 M NaCl across a bilayer lipid membrane in the presence of dipicrylaminate (DPA ). By use of radioisotope (
). By use of radioisotope (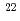 Na), it has been confirmed that Na
Na), it has been confirmed that Na is transported mainly and that DPA
is transported mainly and that DPA serves as a carrier of Na
serves as a carrier of Na transport.
transport.
 /U
/U and U
and U /U couples in molten LiCl-RbCl eutectic
/U couples in molten LiCl-RbCl eutecticNagai, Takayuki; Uehara, Akihiro*; Fujii, Toshiyuki*; Shirai, Osamu*; Sato, Nobuaki*; Yamana, Hajimu*
Denki Kagaku Oyobi Kogyo Butsuri Kagaku, 77(8), p.614 - 616, 2009/08
Times Cited Count:14 Percentile:31.08(Electrochemistry)Formal redox potentials of the U /U
/U and U
and U /U couples in molten LiCl-RbCl eutectic were determined by cyclic voltammetry. These redox potentials were more negative than those in the LiCl-KCl eutectic but positive comparing to those in the NaCl-CsCl eutectic. This relation would be correlated with the averaged alkali cation radius.
/U couples in molten LiCl-RbCl eutectic were determined by cyclic voltammetry. These redox potentials were more negative than those in the LiCl-KCl eutectic but positive comparing to those in the NaCl-CsCl eutectic. This relation would be correlated with the averaged alkali cation radius.
 /Pu
/Pu and PuO
and PuO
 /Pu
/Pu couples in molten NaCl-CsCl eutectic as measured by absorption spectrophotometry
couples in molten NaCl-CsCl eutectic as measured by absorption spectrophotometryNagai, Takayuki; Uehara, Akihiro*; Fujii, Toshiyuki*; Shirai, Osamu*; Myochin, Munetaka; Yamana, Hajimu*
Radiochimica Acta, 97(4-5), p.209 - 212, 2009/05
Times Cited Count:3 Percentile:24.52(Chemistry, Inorganic & Nuclear)Redox behavior of uranium and plutonium in molten salts is the essential information for establishing an effective reprocessing process with spent MOX fuel. In an oxide electro-winning reprocessing process, the spent nuclear fuel is dissolved into the molten NaCl-CsCl eutectic, and the dissolved U(VI) and Pu(VI) species are reduced to be dioxides by electrolysis. In the present study, formal redox potentials of the Pu /Pu
/Pu and PuO
and PuO
 /Pu
/Pu couples in the molten NaCl-CsCl eutectic at 923 K were determined. The fractions of Pu
couples in the molten NaCl-CsCl eutectic at 923 K were determined. The fractions of Pu , Pu
, Pu , and PuO
, and PuO
 were determined by UV/Vis/NIR absorption spectrophotometry. PuO
were determined by UV/Vis/NIR absorption spectrophotometry. PuO was dissolved into the molten NaCl-CsCl eutectic by Cl
was dissolved into the molten NaCl-CsCl eutectic by Cl
 gas purging. Valences of plutonium (Pu
gas purging. Valences of plutonium (Pu , Pu
, Pu , Pu
, Pu , and PuO
, and PuO
 ) were controlled by changing the flow rates of Cl
) were controlled by changing the flow rates of Cl , O
, O , and Ar gases.
, and Ar gases.
 /U
/U and U
and U /U couples in molten LiCl-RbCl eutectic
/U couples in molten LiCl-RbCl eutecticNagai, Takayuki; Uehara, Akihiro*; Fujii, Toshiyuki*; Shirai, Osamu*; Sato, Nobuaki*; Yamana, Hajimu*
Proceedings of 2008 Joint Symposium on Molten Salts (USB Flash Drive), p.927 - 932, 2008/10
Formal redox potentials of the U /U
/U and U
and U /U couples in molten LiCl-RbCl eutectic were determined by cyclic voltammetry. These redox potentials were more negative than those in the LiCl-KCl eutectic but approximately similar to those in the NaCl-CsCl eutectic. These redox potentials in molten alkali chlorides would be correlated with the averaged alkali cationic radius of the melt.
/U couples in molten LiCl-RbCl eutectic were determined by cyclic voltammetry. These redox potentials were more negative than those in the LiCl-KCl eutectic but approximately similar to those in the NaCl-CsCl eutectic. These redox potentials in molten alkali chlorides would be correlated with the averaged alkali cationic radius of the melt.
 organic solution interface in chelate extraction
organic solution interface in chelate extractionUehara, Akihiro*; Kasuno, Megumi*; Okugaki, Tomohiko*; Kitatsuji, Yoshihiro; Shirai, Osamu*; Yoshida, Zenko; Kihara, Sorin*
Journal of Electroanalytical Chemistry, 604(2), p.115 - 124, 2007/06
The distribution ratio of a metal ion between an aqueous solution and an organic solution in solvent extraction with a chelating agent was evaluated by using physicochemical constants determined electrochemically such as standard Gibbs energies for transfers of the ions, the overall complex formation constants and the acid dissociation constants of the chelating agent. The distribution ratio thus evaluated agreed well with those determined by the distribution experiment.
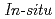 measurement of UO
measurement of UO
 concentration in molten NaCl-2CsCl by differential pulse voltammetry
concentration in molten NaCl-2CsCl by differential pulse voltammetryNagai, Takayuki; Uehara, Akihiro*; Fujii, Toshiyuki*; Shirai, Osamu*; Yamana, Hajimu*
Journal of Nuclear Science and Technology, 43(12), p.1511 - 1516, 2006/12
Times Cited Count:13 Percentile:65.83(Nuclear Science & Technology)In order to assess the applicability of Differential Pulse Voltammetry technique to the  measurement of UO
measurement of UO
 concentration in the oxide electro-winning process, DPV measurements for UO
concentration in the oxide electro-winning process, DPV measurements for UO Cl
Cl in molten NaCl-2CsCl were studied. DPV measurement of UO
in molten NaCl-2CsCl were studied. DPV measurement of UO
 in NaCl-2CsCl at 923 K, with a set of optimized parameters (potential sweep rate -0.1 V/s, pulse cycle 0.1 s, pulse width 10 ms, and pulse potential height 50 mV), showed a clear current peak at -0.9 V vs. Cl
in NaCl-2CsCl at 923 K, with a set of optimized parameters (potential sweep rate -0.1 V/s, pulse cycle 0.1 s, pulse width 10 ms, and pulse potential height 50 mV), showed a clear current peak at -0.9 V vs. Cl /Cl
/Cl , and it was attributed to the reduction of UO
, and it was attributed to the reduction of UO
 to UO
to UO
 . The relation between the current peak height and the analytical concentration of the UO
. The relation between the current peak height and the analytical concentration of the UO
 showed good proportionality in the concentration region up to 0.06 mol/
showed good proportionality in the concentration region up to 0.06 mol/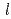 , and the applicability of UO
, and the applicability of UO
 concentration measurement by DPV was confirmed up to 0.4 mol/
concentration measurement by DPV was confirmed up to 0.4 mol/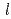 . In order to assess the interference by the coexisting fission product elements to the measurement of UO
. In order to assess the interference by the coexisting fission product elements to the measurement of UO
 concentration, DPV measurements of UO
concentration, DPV measurements of UO
 concentration in molten NaCl-2CsCl containing PdCl
concentration in molten NaCl-2CsCl containing PdCl , NdCl
, NdCl , SmCl
, SmCl , and CeCl
, and CeCl were performed also. Even before removing Pd, the current peak at -0.9 V vs. Cl
were performed also. Even before removing Pd, the current peak at -0.9 V vs. Cl /Cl
/Cl by the reduction of UO
by the reduction of UO
 to UO
to UO
 was found to be distinguishable from the reduction currents of Pd
was found to be distinguishable from the reduction currents of Pd to Pd at -0.7 V vs. Cl
to Pd at -0.7 V vs. Cl /Cl
/Cl . As a result, the applicability of DPV measurement technique to the
. As a result, the applicability of DPV measurement technique to the  monitoring of UO
monitoring of UO
 concentration in oxide electro-winning method is proposed.
concentration in oxide electro-winning method is proposed.
Shirai, Osamu*; Yamana, Hajimu*; Arai, Yasuo
Journal of Alloys and Compounds, 408-412, p.1267 - 1273, 2006/02
Times Cited Count:39 Percentile:84.47(Chemistry, Physical)no abstracts in English
Yoshida, Yumi*; Maeda, Koji*; Shirai, Osamu*; Onuki, Toshihiko
Chemistry Letters, 35(1), p.132 - 133, 2006/01
Times Cited Count:2 Percentile:13.65(Chemistry, Multidisciplinary)no abstracts in English
 /U
/U in molten NaCl-2CsCl by UV-Vis spectrophotometery and cyclic voltammetry
in molten NaCl-2CsCl by UV-Vis spectrophotometery and cyclic voltammetryNagai, Takayuki; Uehara, Akihiro*; Fujii, Toshiyuki*; Shirai, Osamu*; Sato, Nobuaki*; Yamana, Hajimu*
Journal of Nuclear Science and Technology, 42(12), p.1025 - 1031, 2005/12
Times Cited Count:53 Percentile:94.93(Nuclear Science & Technology)Focusing on the cover layer materials (as the Radon Barrier Materials), which could have the effect to restrain the radon from scattering into the air and the effect of the radiation shielding, we produced the radon barrier materials with crude bentonite on an experimental basis, using the rotary type comprehensive unit for grinding and mixing, through which we carried out the evaluation of the characteristics thereof.
Shirai, Osamu*; Uehara, Akihiro*; Yamana, Hajimu*; Onuki, Toshihiko; Yoshida, Yumi*; Kihara, Sorin*
Journal of Nuclear and Radiochemical Sciences, 6(1), p.55 - 60, 2005/07
The ion transport from one aqueous phase (W1) to another (W2) across a bilayer lipid membrane (BLM) in a cell system in the presence of a hydrophobic ion or an ionophore was investigated by voltammetry. The hydrophobic ion was distributed into the BLM with the counter ion to hold the electroneutrality within the BLM. It was pointed out that the counter ion could transfer between W1 and W2 across the BLM since concentrations of the counter ion in W1, BLM and W2 were so high as to cause the ion transfer current while concentrations of the hydrophobic ion were very low. The facilitated transports of alkali ions across a BLM containing valinomycin (Val) used as an ionophore were also investigated by considering the hydrophobicity of both the objective cation and the counter anion and the formation of the alkali metal ion-Val complex..
Shirai, Osamu; Kato, Tetsuya*; Iwai, Takashi; Arai, Yasuo; Yamashita, Toshiyuki
Journal of Physics and Chemistry of Solids, 66(2-4), p.456 - 460, 2005/02
Times Cited Count:8 Percentile:37.11(Chemistry, Multidisciplinary)Electrochemical behaviors of PuN and (U, Pu)N in the LiCl-KCl eutectic melt containing UCl and PuCl
and PuCl at 773 K were investigated by cyclic voltammetry. The electrochemical dissolution of PuN and (U, Pu)N began nearly at -1.0 V vs. the Ag/AgCl reference electrode. The rest potentials of PuN and (U, Pu)N were observed at about 0.15 V more negative potential than that of UN since the equilibrium potential of UN is about 0.15 V more positive than that of PuN. In the cyclic voltammogram measured by using (U, Pu)N as the working electrode, a steep rise of the positive current was observed at more positive potential than -0.4 V in analogy with the cyclic voltammogram measured by using UN as the working electrode. In addition, there were two anodic current waves in the voltammogram with (U, Pu)N, though the wave form was not clear. This indicates that UN and PuN would be dissolved independently irrespective of formation of the solid solution, (U, Pu)N.
at 773 K were investigated by cyclic voltammetry. The electrochemical dissolution of PuN and (U, Pu)N began nearly at -1.0 V vs. the Ag/AgCl reference electrode. The rest potentials of PuN and (U, Pu)N were observed at about 0.15 V more negative potential than that of UN since the equilibrium potential of UN is about 0.15 V more positive than that of PuN. In the cyclic voltammogram measured by using (U, Pu)N as the working electrode, a steep rise of the positive current was observed at more positive potential than -0.4 V in analogy with the cyclic voltammogram measured by using UN as the working electrode. In addition, there were two anodic current waves in the voltammogram with (U, Pu)N, though the wave form was not clear. This indicates that UN and PuN would be dissolved independently irrespective of formation of the solid solution, (U, Pu)N.
Shirai, Osamu*; Yamada, Hajimu*; Onuki, Toshihiko; Yoshida, Yumi*; Kihara, Sorin*
Journal of Electroanalytical Chemistry, 570(2), p.219 - 226, 2004/09
Times Cited Count:24 Percentile:56.4(Chemistry, Analytical)no abstracts in English
 /Np couple at liquid Cd and Bi electrodes in LiCl-KCl eutectic melts
/Np couple at liquid Cd and Bi electrodes in LiCl-KCl eutectic meltsShirai, Osamu; Uozumi, Koichi*; Iwai, Takashi; Arai, Yasuo
Journal of Applied Electrochemistry, 34(3), p.323 - 330, 2004/03
Times Cited Count:28 Percentile:52.41(Electrochemistry)The electrode reactions of the Np /Np couple at liquid Cd and Bi electrodes were investigated by cyclic voltammetry at 723, 773 and 823 K in LiCl-KCl eutectic melt. It was found that the diffusion of Np
/Np couple at liquid Cd and Bi electrodes were investigated by cyclic voltammetry at 723, 773 and 823 K in LiCl-KCl eutectic melt. It was found that the diffusion of Np in the salt phase was a rate-determining step in the cathodic reaction when the concentration of NpCl
in the salt phase was a rate-determining step in the cathodic reaction when the concentration of NpCl was less than about 1 wt.% and the liquid Cd or Bi phase was not saturated with Np. The redox potentials of the Np
was less than about 1 wt.% and the liquid Cd or Bi phase was not saturated with Np. The redox potentials of the Np /Np couple at liquid Cd electrode at 723, 773 and 823 K were observed more positively than those at Mo electrode by 0.158, 0.140 and 0.126 V, respectively. The potential shift would result from a lowering of activity of Np in Cd phase according to the alloy formation of NpCd
/Np couple at liquid Cd electrode at 723, 773 and 823 K were observed more positively than those at Mo electrode by 0.158, 0.140 and 0.126 V, respectively. The potential shift would result from a lowering of activity of Np in Cd phase according to the alloy formation of NpCd at 723 K and NpCd
at 723 K and NpCd at 773 and 823 K. The redox potentials of the Np
at 773 and 823 K. The redox potentials of the Np /Np couple at liquid Bi electrode at 723, 773 and 823 K were more positive than those at Mo electrode by 0.427, 0.419 and 0.410 V, respectively, which would be attributable to a lowering of activity of Np in Bi phase according to the formation of NpBi
/Np couple at liquid Bi electrode at 723, 773 and 823 K were more positive than those at Mo electrode by 0.427, 0.419 and 0.410 V, respectively, which would be attributable to a lowering of activity of Np in Bi phase according to the formation of NpBi .
.
Uozumi, Koichi*; Iizuka, Masatoshi*; Kato, Tetsuya*; Inoue, Tadashi*; Shirai, Osamu*; Iwai, Takashi; Arai, Yasuo
Journal of Nuclear Materials, 325(1), p.34 - 43, 2004/02
Times Cited Count:109 Percentile:98.55(Materials Science, Multidisciplinary)Experiments were conducted on simultaneous recovering of uranium and plutonium electrochemically into laboratory scale liquid cadomium cathodes(LCCs) at different U/Pu ratios in the salt phase. The influence of the salt composition on the recovered amount of uranium and plutonium, the morphology of uranium and plutonium in the LCCs, and the behavior of americium were examined. It was shouwn that there is a threshold in the U/Pu ratio in the salt phase for the successful simultaneous recovery of uranium and plutonium up to 10wt% in high current efficiencies.
Kato, Tetsuya*; Uozumi, Koichi*; Inoue, Tadashi*; Shirai, Osamu*; Iwai, Takashi; Arai, Yasuo
Proceedings of GLOBAL2003 Atoms for Prosperity; Updating Eisenhower's Global Vision for Nuclear Energy (CD-ROM), p.1591 - 1595, 2003/11
Electrolysis experiments were carried to recover plutonium and uranium into liquid cadmium cathodes from molten salt at high cathode current densities. In the electrolysis at 101mA/cm , 10.4wt.% of heavy metals in the cathode was recovered at almost 100% of current efficiency. In the electrolysis at 156mA/cm
, 10.4wt.% of heavy metals in the cathode was recovered at almost 100% of current efficiency. In the electrolysis at 156mA/cm , the cathode potential ascended after approximately 8wt.% of heavy metals was recovered and some deposit was observed outside of the crucible.
, the cathode potential ascended after approximately 8wt.% of heavy metals was recovered and some deposit was observed outside of the crucible.
Mikkelsen, D. R.*; Shirai, Hiroshi; Urano, Hajime*; Takizuka, Tomonori; Kamada, Yutaka; Hatae, Takaki; Koide, Yoshihiko; Asakura, Nobuyuki; Fujita, Takaaki; Fukuda, Takeshi*; et al.
Nuclear Fusion, 43(1), p.30 - 39, 2003/01
Times Cited Count:28 Percentile:64.08(Physics, Fluids & Plasmas)The stiffness of thermal transport in ELMy H-modes is explained in a series of carefully chosen JT-60U plasmas, and measured temperature are compared with the predictions of several transport models. A heating power scan with constant Tped, a scan of pedestal temperature Tped with constant heating power, and an on/off-axis heating comparison are presented. Predictions of the RLWB and IFS/PPPL models generally agree with the measured temperature outside 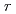
 0.3
0.3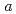 , but the Multimode model uniformly predicts temperatures that are too high except in the central region.
, but the Multimode model uniformly predicts temperatures that are too high except in the central region.
Shirai, Osamu*; Yamana, Hajimu*; Iwai, Takashi; Arai, Yasuo
Proceedings of Nuclear Fuel Cycle Technologies Closing the Fuel Cycle (CD-ROM), 7 Pages, 2003/00
no abstracts in English