Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ishii, Yasuo; Toyoshima, Atsushi; Tsukada, Kazuaki; Asai, Masato; Li, Z.*; Nagame, Yuichiro; Miyashita, Sunao*; Mori, Tomotaka*; Suganuma, Hideo*; Haba, Hiromitsu*; et al.
Bulletin of the Chemical Society of Japan, 84(9), p.903 - 911, 2011/09
Times Cited Count:17 Percentile:49.33(Chemistry, Multidisciplinary)The cation-exchange behavior of element 104, rutherfordium (Rf), was investigated together with its lighter group-4 homologues Zr and Hf, and the tetravalent pseudo-homologue Th in HF/HNO mixed solution. The results demonstrate that distribution coefficients (
mixed solution. The results demonstrate that distribution coefficients (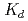 ) of Rf in HF/0.10 M HNO
) of Rf in HF/0.10 M HNO decrease with increasing concentration of the fluoride ion [F
decrease with increasing concentration of the fluoride ion [F ], indicating the consecutive formation of fluorido complexes of Rf. We also measured the
], indicating the consecutive formation of fluorido complexes of Rf. We also measured the 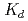 values of Rf and the homologues as a function of the hydrogen ion concentration [H
values of Rf and the homologues as a function of the hydrogen ion concentration [H ]. The log
]. The log 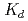 values decrease linearly with an increase of log [H
values decrease linearly with an increase of log [H ] with slopes between -2.1 and -2.5. This indicates that these elements are likely to form the same chemical compounds: mixture of [MF]
] with slopes between -2.1 and -2.5. This indicates that these elements are likely to form the same chemical compounds: mixture of [MF] and [MF
and [MF ]
] (M = Rf, Zr, Hf and Th) in the studied solution. It is also ascertained that sequence in the fluoride complex formation is Zr
(M = Rf, Zr, Hf and Th) in the studied solution. It is also ascertained that sequence in the fluoride complex formation is Zr  Hf
Hf  Rf
Rf  Th.
Th.
Ishii, Yasuo; Toyoshima, Atsushi; Tsukada, Kazuaki; Asai, Masato; Tome, Hayato; Nishinaka, Ichiro; Nagame, Yuichiro; Miyashita, Sunao*; Mori, Tomotaka*; Suganuma, Hideo*; et al.
Chemistry Letters, 37(3), p.288 - 289, 2008/03
Times Cited Count:20 Percentile:55.03(Chemistry, Multidisciplinary)We have investigated cation-exchange behavior of Rf together with the lighter homologues of the group-4 elements Zr and Hf, and the tetravalent pseudo-homologue Th, in HF/HNO solution using Automated Ion exchange separation apparatus coupled with the Detection system for Alpha spectroscopy (AIDA). The
solution using Automated Ion exchange separation apparatus coupled with the Detection system for Alpha spectroscopy (AIDA). The 
 values of Zr, Hf, Th and Rf in HF/0.1 M HNO
values of Zr, Hf, Th and Rf in HF/0.1 M HNO were decreased with increasing the concentration of the fluoride ion [F
were decreased with increasing the concentration of the fluoride ion [F ], indicating the formation of the fluoride complexes. The sequence of the fluoride complexation strength is Zr
], indicating the formation of the fluoride complexes. The sequence of the fluoride complexation strength is Zr  Hf
Hf  Rf
Rf  Th.
Th.
 O/alcohol mixed media by time-resolved laser-induced fluorescence spectroscopy
O/alcohol mixed media by time-resolved laser-induced fluorescence spectroscopyArisaka, Makoto*; Kimura, Takaumi; Suganuma, Hideo*; Yoshida, Zenko
Journal of Radioanalytical and Nuclear Chemistry, 255(2), p.385 - 389, 2003/02
Times Cited Count:5 Percentile:36.81(Chemistry, Analytical)no abstracts in English
Fukushima, Masahiro; Suganuma, Hideo*; Chiba, Satoshi
Progress of Theoretical Physics, 107(6), p.1147 - 1161, 2002/06
Times Cited Count:1 Percentile:13.39(Physics, Multidisciplinary)Quantum Chromodynamics (QCD) is established as the standard theory of the strong interaction. Based on the lattice gauge theory, we investigate a deformation of stabilized instanton solutions in a nontrivial topological sector and further discuss appearance of monopole trajectories around the instanton.
Arisaka, Makoto*; Kimura, Takaumi; Suganuma, Hideo*; Yoshida, Zenko
Radiochimica Acta, 90(4), p.193 - 197, 2002/05
Times Cited Count:14 Percentile:65.6(Chemistry, Inorganic & Nuclear)no abstracts in English
Arisaka, Makoto*; Kimura, Takaumi; Suganuma, Hideo*; Yoshida, Zenko
Radiochimica Acta, 89(8), p.593 - 598, 2001/08
Times Cited Count:11 Percentile:61.99(Chemistry, Inorganic & Nuclear)no abstracts in English
Seki, Masami; Ikeda, Yoshitaka; *; Imai, Tsuyoshi; ; Yokokura, Kenji; Sawahata, Masayuki; Suganuma, Kazuaki; Sato, Minoru; *; et al.
Fusion Technology 1990, p.1060 - 1064, 1991/00
no abstracts in English
 /HF mixed solution, 2
/HF mixed solution, 2Ishii, Yasuo; Tome, Hayato; Toyoshima, Atsushi; Asai, Masato; Nishinaka, Ichiro; Tsukada, Kazuaki; Nagame, Yuichiro; Miyashita, Sunao*; Mori, Tomotaka*; Suganuma, Hideo*; et al.
no journal, ,
Chemical properties of rutherfordium (Rf, the element 104) in HNO /HF mixed solutions have been investigated using cation-exchange chromatography. Distribution coefficients of Rf on the cation-exchange resin as a function of hydrofluoric-acid and nitric-acid concentrations have been measured precisely, and it was found that the strength of the fluoride complex formation of Rf is much weaker than those of lighter group 4 homologs Zr and Hf. The same result was obtained through the anion-exchange chromatography. These results enabled us to quantitatively account for the fluoride complexation of Rf.
/HF mixed solutions have been investigated using cation-exchange chromatography. Distribution coefficients of Rf on the cation-exchange resin as a function of hydrofluoric-acid and nitric-acid concentrations have been measured precisely, and it was found that the strength of the fluoride complex formation of Rf is much weaker than those of lighter group 4 homologs Zr and Hf. The same result was obtained through the anion-exchange chromatography. These results enabled us to quantitatively account for the fluoride complexation of Rf.
 /HF solution
/HF solutionIshii, Yasuo; Tome, Hayato; Toyoshima, Atsushi; Asai, Masato; Nishinaka, Ichiro; Tsukada, Kazuaki; Nagame, Yuichiro; Miyashita, Sunao*; Mori, Tomotaka*; Suganuma, Hideo*; et al.
no journal, ,
The cation-exchange behavior of rutherfordium(Rf) in HNO /HF solution has been studied based on an atom-at-a-time scale. In the symposium, we will discuss the fluoride complexation of rutherfordium in comparison with that of Zr, Hf and Th.
/HF solution has been studied based on an atom-at-a-time scale. In the symposium, we will discuss the fluoride complexation of rutherfordium in comparison with that of Zr, Hf and Th.