Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Re-MAG3-conjugated bisphosphonate (
Re-MAG3-conjugated bisphosphonate ( Re-MAG3-HBP), an agent for treatment of painful bone metastases
Re-MAG3-HBP), an agent for treatment of painful bone metastasesOgawa, Kazuma*; Mukai, Takahiro*; Kawai, Keiichi*; Takamura, Norito*; Hanaoka, Hirofumi*; Hashimoto, Kazuyuki; Shiba, Kazuhiro*; Mori, Hirofumi*; Saji, Hideo*
European Journal of Nuclear Medicine and Molecular Imaging, 36(1), p.115 - 121, 2009/01
Times Cited Count:23 Percentile:57.81(Radiology, Nuclear Medicine & Medical Imaging)We have developed a  Re-MAG3 complex-conjugated bisphosphonate (
Re-MAG3 complex-conjugated bisphosphonate ( Re-MAG3-HBP) for the treatment of painful bone metastases. We assumed competitive inhibitors of protein binding to be useful for procuring a favorable biodistribution of
Re-MAG3-HBP) for the treatment of painful bone metastases. We assumed competitive inhibitors of protein binding to be useful for procuring a favorable biodistribution of  Re-MAG3-HBP because it has been reported that the concurrent administration of
Re-MAG3-HBP because it has been reported that the concurrent administration of  Tc-MAG3 and drugs with high affinity for serum protein enhanced total clearance and tissue distribution. The protein binding of
Tc-MAG3 and drugs with high affinity for serum protein enhanced total clearance and tissue distribution. The protein binding of  Re-MAG3-HBP in a human serum albumin solution was significantly decreased by the addition of ceftriaxone. In the biodistribution experiments, pretreatment with ceftriaxone enhanced the clearance of the radioactivity of
Re-MAG3-HBP in a human serum albumin solution was significantly decreased by the addition of ceftriaxone. In the biodistribution experiments, pretreatment with ceftriaxone enhanced the clearance of the radioactivity of  Re-MAG3-HBP in blood and nontarget tissues but had no effect on accumulation in bone. The findings suggested that the use of protein-binding competitive inhibitors would be effective in improving the pharmacokinetics of radiopharmaceuticals with high affinity for serum protein.
Re-MAG3-HBP in blood and nontarget tissues but had no effect on accumulation in bone. The findings suggested that the use of protein-binding competitive inhibitors would be effective in improving the pharmacokinetics of radiopharmaceuticals with high affinity for serum protein.
Ogawa, Kazuma*; Mukai, Takahiro*; Kawai, Keiichi*; Takamura, Norito*; Hanaoka, Hirofumi*; Hashimoto, Kazuyuki; Saji, Hideo*
no journal, ,
Previously, based on the concept of bifunctional radiopharmaceuticals, we developed a highly stable Re-MAG3 complex conjugated bisphosphonate (
Re-MAG3 complex conjugated bisphosphonate ( Re-MAG3-HBP) for treatment of the painful bone metastases. We assumed that competitive inhibitors of protein binding might be useful for
Re-MAG3-HBP) for treatment of the painful bone metastases. We assumed that competitive inhibitors of protein binding might be useful for Re-MAG3-HBP because it has been reported that the concurrent administration between
Re-MAG3-HBP because it has been reported that the concurrent administration between 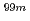 Tc-MAG3 and drugs with high protein binding affinity produced competitive displacement at the specific binding sites and enhanced total clearance and tissue distribution. In this study, the displacement effect between
Tc-MAG3 and drugs with high protein binding affinity produced competitive displacement at the specific binding sites and enhanced total clearance and tissue distribution. In this study, the displacement effect between  Re-MAG3-HBP and several protein binding inhibitors were investigated. Effects of each competitive protein binding inhibitor were examined in in vitro human serum. Bucolome, ceftriaxione and cefazolin as displacers of human serum albumin (HSA) binding site I, and ibuprofen as a displacer of HSA binding site II were added to each serum before addition of
Re-MAG3-HBP and several protein binding inhibitors were investigated. Effects of each competitive protein binding inhibitor were examined in in vitro human serum. Bucolome, ceftriaxione and cefazolin as displacers of human serum albumin (HSA) binding site I, and ibuprofen as a displacer of HSA binding site II were added to each serum before addition of  Re-MAG3-HBP. As a result of in vitro study, the free fraction rate of
Re-MAG3-HBP. As a result of in vitro study, the free fraction rate of  Re-MAG3-HBP was significantly increased by the addition of ceftriaxone. In biodistribution studies, the loading ceftriaxone enhanced the clearance of radioactivity of
Re-MAG3-HBP was significantly increased by the addition of ceftriaxone. In biodistribution studies, the loading ceftriaxone enhanced the clearance of radioactivity of  Re-MAG3-HBP in blood and non-target tissues without the reduction of the bone accumulation. These findings suggested that the use of a protein binding competitive inhibitor would be effective for lowering the side effects of
Re-MAG3-HBP in blood and non-target tissues without the reduction of the bone accumulation. These findings suggested that the use of a protein binding competitive inhibitor would be effective for lowering the side effects of  Re-MAG3-HBP.
Re-MAG3-HBP.