Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Nishimura, Shoichiro*; Torii, Hiroyuki*; Fukao, Yoshinori*; Ito, Takashi; Iwasaki, Masahiko*; Kanda, Sotaro*; Kawagoe, Kiyotomo*; Kawall, D.*; Kawamura, Naritoshi*; Kurosawa, Noriyuki*; et al.
Physical Review A, 104(2), p.L020801_1 - L020801_6, 2021/08
Times Cited Count:13 Percentile:83.13(Optics)Strasser, P.*; Abe, Mitsushi*; Aoki, Masaharu*; Choi, S.*; Fukao, Yoshinori*; Higashi, Yoshitaka*; Higuchi, Takashi*; Iinuma, Hiromi*; Ikedo, Yutaka*; Ishida, Katsuhiko*; et al.
EPJ Web of Conferences, 198, p.00003_1 - 00003_8, 2019/01
Times Cited Count:13 Percentile:99.06(Quantum Science & Technology)Strasser, P.*; Aoki, Masaharu*; Fukao, Yoshinori*; Higashi, Yoshitaka*; Higuchi, Takashi*; Iinuma, Hiromi*; Ikedo, Yutaka*; Ishida, Katsuhiko*; Ito, Takashi; Iwasaki, Masahiko*; et al.
Hyperfine Interactions, 237(1), p.124_1 - 124_9, 2016/12
Times Cited Count:7 Percentile:90.97(Physics, Atomic, Molecular & Chemical)Hidaka, Koshi*; Kimura, Toru*; Abdel-Rahman, H. M.*; Nguyen, J.-T.*; McDaniel, K. F.*; Kohlbrenner, W. E.*; Molla, A.*; Adachi, Motoyasu; Tamada, Taro; Kuroki, Ryota; et al.
Journal of Medicinal Chemistry, 52(23), p.7604 - 7617, 2009/07
Times Cited Count:19 Percentile:44.7(Chemistry, Medicinal)A series of HIV protease inhibitor based on the allophenylnorstatine structure with various P2' moieties were synthesized. Among these analogues, we discovered that a small allyl group would maintain potent enzyme inhibitory activity compared to that of the 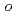 -methylbenzyl moiety in clinical candidate 1 (KNI-764, also known as JE-2147, AG-1776 or SM-319777). Introduction of an anilinic amino group to 2 (KNI-727) improved water-solubility and anti-HIV-1 activity. X-ray crystallographic analysis of 13k (KNI-1689) with a
-methylbenzyl moiety in clinical candidate 1 (KNI-764, also known as JE-2147, AG-1776 or SM-319777). Introduction of an anilinic amino group to 2 (KNI-727) improved water-solubility and anti-HIV-1 activity. X-ray crystallographic analysis of 13k (KNI-1689) with a  -methallyl group at P2' position revealed hydrophobic interactions with Ala28, Ile84, and Ile50' similar to that of 1. The presence of an additional methyl group on the allyl group in compound 13k significantly increased anti-HIV activity over 1, while providing a rational drug design for structural minimization and improving membrane permeability.
-methallyl group at P2' position revealed hydrophobic interactions with Ala28, Ile84, and Ile50' similar to that of 1. The presence of an additional methyl group on the allyl group in compound 13k significantly increased anti-HIV activity over 1, while providing a rational drug design for structural minimization and improving membrane permeability.
Sato, Susumu; Tomisawa, Tetsuo; Hiroki, Fumio; Lee, S.*; Igarashi, Zenei*; Ikegami, Masanori*; Ueno, Akira; Kondo, Yasuhiro; Hasegawa, Kazuo; Toyama, Takeshi*; et al.
Proceedings of 1st Annual Meeting of Particle Accelerator Society of Japan and 29th Linear Accelerator Meeting in Japan, p.467 - 469, 2004/08
As a joint project of KEK and JAERI, a MW class of high intensity proton accelerator (J-PARC), consisting of Linac, 3 GeV-RCS, 50 GeV-MR, is under construction. For this accelerator, it is required to minimize the beam loss (typically, lower than0.1 1 W/m at the linac). To achieve the requirement, beam trajectory needs to be controlled with accuracy of some 100
1 W/m at the linac). To achieve the requirement, beam trajectory needs to be controlled with accuracy of some 100  m. The first stage of the acceleration (up to 181 MeV during the first stage of construction) is done by linac. The beam position monitor (BPM) in the linac utilizes 4 stripline pickups (50 ohm) on the beam transportation chamber. In this paper, systematic calibration of the BPM is described.
m. The first stage of the acceleration (up to 181 MeV during the first stage of construction) is done by linac. The beam position monitor (BPM) in the linac utilizes 4 stripline pickups (50 ohm) on the beam transportation chamber. In this paper, systematic calibration of the BPM is described.
Sato, Susumu; Igarashi, Zenei*; Lee, S.*; Tomisawa, Tetsuo; Hiroki, Fumio; Kishiro, Junichi; Ikegami, Masanori*; Kondo, Yasuhiro; Hasegawa, Kazuo; Ueno, Akira; et al.
Proceedings of 22nd International Linear Accelerator Conference (LINAC 2004), p.429 - 431, 2004/00
As a joint project of KEK and JAERI, a MW class of high intensity proton accelerator (J-PARC), consisting of Linac, 3 GeV-RCS, 50 GeV-MR, is under construction at Tokai, Japan. For this accelerator, it is required to minimize the beam loss (typically, lower than 0.1 1 W/m at the linac). To achieve the requirement, beam trajectory needs to be controlled with accuracy of some 100 micro-meter. The first stage of the acceleration (up to 181 MeV during the first stage of construction) is done by linac. The beam position monitor (BPM) in the linac utilizes 4 strip-line pickups (50 ohm) on the beam transportation chamber. In this paper, systematic calibration of the BPM is described.
1 W/m at the linac). To achieve the requirement, beam trajectory needs to be controlled with accuracy of some 100 micro-meter. The first stage of the acceleration (up to 181 MeV during the first stage of construction) is done by linac. The beam position monitor (BPM) in the linac utilizes 4 strip-line pickups (50 ohm) on the beam transportation chamber. In this paper, systematic calibration of the BPM is described.
 -FeSi
-FeSi fabricated by ion beam sputter deposition method
fabricated by ion beam sputter deposition methodSaito, Takeru; Yamamoto, Hiroyuki; Yamaguchi, Kenji; Nakanoya, Takamitsu; Hojo, Kiichi; Haraguchi, Masaharu*; Imamura, Motoyasu*; Matsubayashi, Nobuyuki*; Tanaka, Tomoaki*; Shimada, Hiromichi*
Nuclear Instruments and Methods in Physics Research B, 206, p.321 - 325, 2003/05
Times Cited Count:7 Percentile:46.83(Instruments & Instrumentation)no abstracts in English
 -FeSi
-FeSi thin films on Si(III) surface by solid phase epitaxy (SPE) analyzed by means of synchrotron radiation XPS (SR-XPS)
thin films on Si(III) surface by solid phase epitaxy (SPE) analyzed by means of synchrotron radiation XPS (SR-XPS)Saito, Takeru; Yamamoto, Hiroyuki; Asaoka, Hidehito; Haraguchi, Masaharu*; Imamura, Motoyasu*; Matsubayashi, Nobuyuki*; Tanaka, Tomoaki*; Shimada, Hiromichi*; Hojo, Kiichi
Analytical Sciences (CD-ROM), 17(Suppl.), p.1073 - 1076, 2002/03
no abstracts in English
Matsubayashi, Nobuyuki*; Tanaka, Tomoaki*; Imamura, Motoyasu*; Shimada, Hiromichi*; Saito, Takeru
Analytical Sciences (CD-ROM), 17(Suppl.), p.119 - 121, 2002/03
no abstracts in English

Bando, Kyoko*; Saito, Takeru; Sato, Koichi*; Tanaka, Tomoaki*; Dumeignil, F.*; Imamura, Motoyasu*; Matsubayashi, Nobuyuki*; Shimada, Hiromichi*
Topics in Catalysis, 18(1-2), p.59 - 65, 2002/01
no abstracts in English
 -FeSi
-FeSi on Si(111) substrate studied by means of SR-XPS
on Si(111) substrate studied by means of SR-XPSSaito, Takeru; Yamamoto, Hiroyuki; Haraguchi, Masaharu*; Imamura, Motoyasu*; Matsubayashi, Nobuyuki*; Tanaka, Tomoaki*; Shimada, Hiromichi*; Hojo, Kiichi
Photon Factory Activity Report 2001, (19), P. 205, 2001/00
no abstracts in English
Adachi, Motoyasu; Arai, Shigeki; Matsumoto, Fumiko; Kuroki, Ryota; Hatanaka, Takaaki*; Ito, Yuji*; Hidaka, Koshi*; Tsuda, Yuko*; Kiso, Yoshiaki*
no journal, ,
HIV protease is known as a drug target protein. To compare interactions between HIVPR and inhibitors, single-chained HIVPR linked by a disulfide bond was designed as N98C mutant. Both WT N98C and A17 N98C mutants were expressed as inclusion body and refolded by dilution method. The yield of the purified enzymes was similar to that of WT. We will report the results of interactions between inhibitors and WT N98C and A17 N98C mutants by physicochemical and crystal structure analyses.
Matsumoto, Fumiko; Hatanaka, Takaaki*; Adachi, Motoyasu; Shimizu, Rumi; Tamada, Taro; Ito, Yuji*; Kuroki, Ryota
no journal, ,
no abstracts in English
Adachi, Motoyasu; Hatanaka, Takaaki*; Ito, Yuji*; Hidaka, Koshi*; Tsuda, Yuko*; Kiso, Yoshiaki*; Kuroki, Ryota
no journal, ,
HIV protease is known as a drug target protein. It is important to clear relationship between structural data and kinetic and physicochemical parameters for drug design. We prepared single-chained enzyme linked by two amino acids and cross-liked enzyme bridged by disulfide bond. The two single-chained enzymes were expressed as inclusion body and refolded by dilution method. The purified enzyme complexed with inhibitor KNI-272 was crystallized, and solved the crystal structures. The designed sc- and cl-HIV-PR will be useful for evaluate the affinity of newly designed inhibitors from kinetic and thermodynamic point of view. Finally, we also report the results of analysis in affinity of inhibitors by surface plasmon resonance.
Hwang, T.; Hasegawa, Akira*; Tomura, Keiko*; Ebisawa, Naoki*; Toyama, Takeshi*; Nagai, Yasuyoshi*; Fukuda, Makoto*; Miyazawa, Takeshi*; Tanaka, Teruya*; Nogami, Shuhei*
no journal, ,
Tanaka, Tatsuya*; Toya, Naruhisa*; Okugi, Sakura*; Hashimoto, Shuya*; Matsui, Hiroya
no journal, ,
no abstracts in English