Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Takahashi, Rieko*; Taniguchi, Naoki
Zairyo To Kankyo, 73(6), p.153 - 163, 2024/06
Carbon steel is one of the candidate materials for overpacks in geological disposal of high-level radioactive waste, and is known to susceptible to stress corrosion cracking(SCC) depending on the condition in carbonate environment. In order to understand the influence of temperature on the SCC susceptibility of carbon steel, slow strain rate test (SSRT) of rolled steel were performed in NaHCO aqueous solution with varying temperature in the range of 303-393K for conditions of 0.1-0.5 mol/dm
aqueous solution with varying temperature in the range of 303-393K for conditions of 0.1-0.5 mol/dm , which is assumed to be the upper limit of carbonate concentration in groundwater in a geological disposal environment. As the results, no obvious influence of temperature on mechanical properties such as fracture strain ratio and reduction area ratio were observed, but SCC susceptibility based on SCC fracture ratio increased at relatively low temperatures of 303K and 323K. It was suggested that the reason for the higher SCC sensitivity at lower temperatures was due to slower repassivation at lower temperatures. Regarding the type of SCC, intergranular SCC was dominant at low temperatures and tended to transition to intergranular SCC at higher temperatures. Transgranular SCC tended to be observed at lower potentials than those at which intergranular SCC was observed.
, which is assumed to be the upper limit of carbonate concentration in groundwater in a geological disposal environment. As the results, no obvious influence of temperature on mechanical properties such as fracture strain ratio and reduction area ratio were observed, but SCC susceptibility based on SCC fracture ratio increased at relatively low temperatures of 303K and 323K. It was suggested that the reason for the higher SCC sensitivity at lower temperatures was due to slower repassivation at lower temperatures. Regarding the type of SCC, intergranular SCC was dominant at low temperatures and tended to transition to intergranular SCC at higher temperatures. Transgranular SCC tended to be observed at lower potentials than those at which intergranular SCC was observed.
Fujimoto, Shinji*; Tsuchiya, Hiroaki*; Ogawa, Soma*; Iida, Yoshihisa; Taniguchi, Naoki
Materials and Corrosion, 72(1-2), p.333 - 338, 2021/01
Times Cited Count:3 Percentile:13.68(Materials Science, Multidisciplinary)The stress corrosion cracking (SCC) of pure copper in bentonite was examined by a slow strain rate test (SSRT). The bentonite was swollen with pure water or aqueous solutions containing NH of 0.05 M and 0.1 M. The thick corrosion films and particulate deposits were formed on copper surface after SSRT. Typical tarnish rupture type SCC occurred on pure copper in swollen bentonite with and without NH
of 0.05 M and 0.1 M. The thick corrosion films and particulate deposits were formed on copper surface after SSRT. Typical tarnish rupture type SCC occurred on pure copper in swollen bentonite with and without NH . Crack propagation rate was enhanced by NH
. Crack propagation rate was enhanced by NH . It is confirmed thick oxide layer is formed on copper during plastic deformation which resulting in the tarnish crack type SCC. The many particulate deposits observed on the surface might be formed as a results of rapid dissolution of Cu
. It is confirmed thick oxide layer is formed on copper during plastic deformation which resulting in the tarnish crack type SCC. The many particulate deposits observed on the surface might be formed as a results of rapid dissolution of Cu to form porous CuO at locallized deformed sites.
to form porous CuO at locallized deformed sites.
Ogawa, Yusuke*; Suzuki, Satoru*; Taniguchi, Naoki; Kawasaki, Manabu*; Suzuki, Hiroyuki*; Takahashi, Rieko*
Materials and Corrosion, 72(1-2), p.52 - 66, 2021/01
Times Cited Count:2 Percentile:8.90(Materials Science, Multidisciplinary)Cast steel is one of the promising alternative to forged steel that is the current reference material for carbon steel overpack. In this study, the full-scale cast steel overpack was produced experimentally and the distribution of casting defects were investigated. The corrosion test regarding corrosion rate and stress corrosion cracking (SCC) susceptibility were also conducted using samples taken from the full-scale cast steel overpack and the corrosion resistance of cast steel was compared with that of forged steel. From above two corrosion tests, it can be said that the corrosion resistance of cast steel is mostly the same as that of forged steel.
Kitayama, Ayami; Taniguchi, Naoki; Mitsui, Seiichiro
Materials and Corrosion, 72(1-2), p.211 - 217, 2021/01
Times Cited Count:0 Percentile:0.00(Materials Science, Multidisciplinary)Sugita, Yutaka; Taniguchi, Naoki; Makino, Hitoshi; Kanamaru, Shinichiro*; Okumura, Taisei*
Nihon Genshiryoku Gakkai Wabun Rombunshi, 19(3), p.121 - 135, 2020/09
A series of structural analysis of disposal containers for direct disposal of spent fuel was carried out to provide preliminary estimates of the required pressure resistance thickness of the disposal container. Disposal containers were designed to contain either 2, 3 or 4 spent fuel assemblies in linear, triangular or square arrangements, respectively. The required pressure resistance thickness was evaluated using separation distance of the housing space for each spent fuel assembly as a key model parameter to obtain the required thickness of the body and then the lid of the disposal container. This work also provides additional analytical technical knowledge, such as the validity of the setting of the stress evaluation line and the effect of the model length on the analysis. These can then be referred to and used again in the future as a basis for conducting similar evaluations under different conditions or proceeding with more detailed evaluations.
Nakayama, Masashi; Saiga, Atsushi; Kimura, Shun; Mochizuki, Akihito; Aoyagi, Kazuhei; Ono, Hirokazu; Miyakawa, Kazuya; Takeda, Masaki; Hayano, Akira; Matsuoka, Toshiyuki; et al.
JAEA-Research 2019-013, 276 Pages, 2020/03
The Horonobe Underground Research Laboratory (URL) Project is being pursued by the Japan Atomic Energy Agency (JAEA) to enhance the reliability of relevant disposal technologies for geological disposal of High-level Radioactive Waste through investigations of the deep geological environment within the host sedimentary rock at Horonobe Town in Hokkaido, north Japan. The investigations will be conducted in three phases, namely "Phase 1: Surface based investigations", "Phase 2: Construction phase" (investigations during construction of the underground facilities) and "Phase 3: Operation phase" (research in the underground facilities). According to the research plan described in the 3rd Mid- and Long- term Plan of JAEA, "Near-field performance study", "Demonstration of repository design option", and "Verification of crustal-movement buffering capacity of sedimentary rocks" are important issues of the Horonobe URL Project, and schedule of future research and backfill plans of the project will be decided by the end of 2019 Fiscal Year. The present report summarizes the research and development activities of these 3 important issues carried out during 3rd Medium to Long-term Research Phase.
Taniguchi, Naoki; Nakayama, Masashi
Zairyo To Kankyo, 67(12), p.487 - 494, 2018/12
This article describes the current status of corrosion monitoring methods and examples of the measurement under deep underground environments for carbon steel overpacks for high-level radioactive waste disposal. Based on the studies on corrosion monitoring using AC Impedance technique, some of the typical measurement systems such as the electrodes arrangement are introduced. In-situ corrosion monitoring in engineering scale test is also being attempted using a deep underground research facility, and the measurement method and results are presented in this article.
 O in Fe
O in Fe O
O film on Fe formed in an NaOH solution containing oxidants
film on Fe formed in an NaOH solution containing oxidantsHaruna, Takumi*; Miyataki, Yuki*; Hirohata, Yohei*; Shibata, Toshio*; Taniguchi, Naoki; Tachikawa, Hirokazu*
Zairyo To Kankyo, 67(9), p.375 - 380, 2018/09
This research aimed to confirm the formation of Fe O
O film on Fe immersed in aqueous 45 mass% NaOH solution containing some oxidants at the boiling temperature, to recognize the optimum immersion time for the formation of thick and protective film, and to reveal the absorption behavior of D
film on Fe immersed in aqueous 45 mass% NaOH solution containing some oxidants at the boiling temperature, to recognize the optimum immersion time for the formation of thick and protective film, and to reveal the absorption behavior of D O in the Fe
O in the Fe O
O film at room temperature. The results were obtained as follows. It was confirmed that Fe
film at room temperature. The results were obtained as follows. It was confirmed that Fe O
O film was formed on Fe immersed in the NaOH solution for a time more than 0.6 ks, and the film thickness increased parabolically with an increase in the immersion time. D
film was formed on Fe immersed in the NaOH solution for a time more than 0.6 ks, and the film thickness increased parabolically with an increase in the immersion time. D O absorption test was carried out to the films formed in the NaOH solution for immersion times of 1.2 and 3.6 ks. An amount of D
O absorption test was carried out to the films formed in the NaOH solution for immersion times of 1.2 and 3.6 ks. An amount of D O absorbed into the film increased with an increase in an absorption time up to 1000 ks, and an absorption time more than 1000 ks made an amount of D
O absorbed into the film increased with an increase in an absorption time up to 1000 ks, and an absorption time more than 1000 ks made an amount of D O constant. The constant amount of D
O constant. The constant amount of D O was larger for the film formed on Fe immersed in the NaOH solution for 3.6 ks than that for 1.2 ks. The transient of the amount of D
O was larger for the film formed on Fe immersed in the NaOH solution for 3.6 ks than that for 1.2 ks. The transient of the amount of D O absorbed into the film was analyzed on the basis of Fick's law for diffusion, and diffusion coefficients of D
O absorbed into the film was analyzed on the basis of Fick's law for diffusion, and diffusion coefficients of D O were obtained to be 5.1
O were obtained to be 5.1 10
10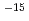 cm
cm s
s and 9.9
and 9.9 10
10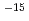 cm
cm s
s for the films formed for 1.2 and 3.6 ks, respectively. Therefore it was estimated that the diffusion coefficient of the Fe
for the films formed for 1.2 and 3.6 ks, respectively. Therefore it was estimated that the diffusion coefficient of the Fe O
O film was in the region from 5.1
film was in the region from 5.1 10
10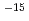 cm
cm s
s to 9.9
to 9.9 10
10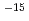 cm
cm s
s .
.
Taniguchi, Naoki
Zairyo To Kankyo, 65(9), p.363 - 364, 2016/09
no abstracts in English
 irradiation
irradiationYukawa, Takuji*; Inoue, Hiroyuki*; Kojima, Takao*; Iwase, Akihiro*; Taniguchi, Naoki; Tachikawa, Hirokazu*
Zairyo To Kankyo 2016 Koenshu (CD-ROM), p.359 - 362, 2016/05
The immersion tests of pure titanium were carried out in aqueous solution containing carbonate/bicarbonate with 50 mM-chloride ion under gamma irradiation. The effect of pH on general corrosion rate of titanium were studied. The experimental results showed that the concentration of hydrogen preoxide was increased with pH, and the corrosion rate increased with the hydrogen preoxide concentration. The corrosion rate in pH12 and 13 were 5 to10 times larger than those under unirradiated conditions.
 O in the film on Fe oxidized at high temperature in air
O in the film on Fe oxidized at high temperature in airHaruna, Takumi*; Yamamoto, Tatsuya*; Miyairi, Yoji*; Shibata, Toshio*; Taniguchi, Naoki; Sakamaki, Keiko; Tachikawa, Hirokazu*
Zairyo To Kankyo, 64(5), p.201 - 206, 2015/05
Diffusion coefficients of D O in the films was determined in order to estimate corrosion rate of carbon steel for the overpack in ground water. Fe plates were heated to form oxide films. The films were characterized with XRD and SEM. After that, the specimen was contacted with D
O in the films was determined in order to estimate corrosion rate of carbon steel for the overpack in ground water. Fe plates were heated to form oxide films. The films were characterized with XRD and SEM. After that, the specimen was contacted with D O for 5184 ks, followed by subjected to TDS to obtain an amount of D
O for 5184 ks, followed by subjected to TDS to obtain an amount of D O absorbing into the film. As a result, single-layered film of Fe
O absorbing into the film. As a result, single-layered film of Fe O
O was formed at 573 and 723 K, and double-layered film of Fe
was formed at 573 and 723 K, and double-layered film of Fe O
O and Fe
and Fe O
O was formed at 873 K. It was found that an amount of D
was formed at 873 K. It was found that an amount of D O in the film correlated linearly with a square root of the absorption period, and that the amount was steady for a long period. From the results and Fick's second law, diffusion coefficients of D
O in the film correlated linearly with a square root of the absorption period, and that the amount was steady for a long period. From the results and Fick's second law, diffusion coefficients of D O was determined as 9.7
O was determined as 9.7 10
10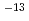 cm
cm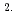 s
s for the Fe
for the Fe O
O film, and 5.5
film, and 5.5 10
10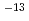 cm
cm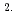 s
s to 2.2
to 2.2 10
10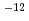 cm
cm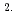 s
s for Fe
for Fe O
O film.
film.
Shibata, Toshio*; Watanabe, Masatoshi; Taniguchi, Naoki; Shimizu, Akihiko*
Zairyo To Kankyo, 62(2), p.70 - 77, 2013/02
In the oxygen depleted underground environment, carbon steel reacts with H O, producing H
O, producing H gas and forming corrosion film on the steel surface. Corrosion rate is controlled by diffusion of reaction species through corrosion film. Diffusion constants of some species working in the corrosion process were obtained from literatures. However, no data were found on the diffusion constant of H
gas and forming corrosion film on the steel surface. Corrosion rate is controlled by diffusion of reaction species through corrosion film. Diffusion constants of some species working in the corrosion process were obtained from literatures. However, no data were found on the diffusion constant of H O in iron oxides based on an appropriate assumption. Mass transfer model for the corrosion rate was used to simulate the corrosion rate of carbon steel. Liquid phase diffusion model of Fe
O in iron oxides based on an appropriate assumption. Mass transfer model for the corrosion rate was used to simulate the corrosion rate of carbon steel. Liquid phase diffusion model of Fe or H
or H O through pores in the corrosion film and solid phase diffusion model of H
O through pores in the corrosion film and solid phase diffusion model of H O through corrosion film itself were examined by Excel simulation. Change in corrosion current density and corrosion loss with time and pH and temperature dependence of corrosion current density were examined. By comparing the results, it is suggested that the solid phase diffusion of H
O through corrosion film itself were examined by Excel simulation. Change in corrosion current density and corrosion loss with time and pH and temperature dependence of corrosion current density were examined. By comparing the results, it is suggested that the solid phase diffusion of H O in the corrosion film controls the corrosion rate of carbon steel in oxygen depleted environment.
O in the corrosion film controls the corrosion rate of carbon steel in oxygen depleted environment.
Shibata, Masahiro; Sawada, Atsushi; Tachi, Yukio; Makino, Hitoshi; Hayano, Akira; Mitsui, Seiichiro; Taniguchi, Naoki; Oda, Chie; Kitamura, Akira; Osawa, Hideaki; et al.
JAEA-Research 2012-032, 298 Pages, 2012/09
JAEA and NUMO have conducted a collaborative research work which is designed to enhance the methodology of repository design and performance assessment in preliminary investigation phase. The topics and the conducted research are follows; (1) Study on selection of host rock: in terms of hydraulic properties, items for assessing rock property, and assessment methodology of groundwater travel time has been organized with interaction from site investigation. (2) Study on development of scenario: the existing approach has been embodied, in addition, the phenomenological understanding regarding dissolution of and nuclide release from vitrified waste, corrosion of the overpack, long-term performance of the buffer are summarized. (3) Study on setting nuclide migration parameters: the approach for parameter setting has been improved for sorption and diffusion coefficient of buffer/rock, and applied and tested for parameter setting of key radionuclides. (4) Study on ensuring quality of knowledge: framework for ensuring quality of knowledge has been studied and examined aimed at the likely disposal facility condition.
Kobayashi, Masato*; Yokoyama, Yutaka*; Takahashi, Rieko*; Asano, Hidekazu*; Taniguchi, Naoki; Naito, Morimasa
Corrosion Engineering, Science and Technology, 46(2), p.212 - 216, 2011/04
Times Cited Count:4 Percentile:27.82(Materials Science, Multidisciplinary)The corrosion behaviour of a carbon steel weld joint under anaerobic conditions was investigated to estimate the long-term integrity of the carbon steel overpack. The weld specimens in this study were produced using three welding methods: GTAW, GMAW and EBW. General corrosion was observed for each immersion specimen and the weld joint corrosion rate was the same as or less than that of the base metal. The hydrogen concentration absorbed during immersion testing was less than 2.48 10
10 mol kg[Fe]
mol kg[Fe] (0.05 ppm) after three years, a value regarded as having little influence on hydrogen embrittlement. The susceptibility to hydrogen embrittlement was highest in the base metal, suggesting that there was little adverse effect on the weld joint from welding. The welded carbon steel overpack is assumed to maintain its resistance to corrosion as a disposal container for the expected lifetime under anaerobic underground conditions.
(0.05 ppm) after three years, a value regarded as having little influence on hydrogen embrittlement. The susceptibility to hydrogen embrittlement was highest in the base metal, suggesting that there was little adverse effect on the weld joint from welding. The welded carbon steel overpack is assumed to maintain its resistance to corrosion as a disposal container for the expected lifetime under anaerobic underground conditions.
Taniguchi, Naoki; Suzuki, Hiroyuki; Kawasaki, Manabu; Naito, Morimasa; Kobayashi, Masato*; Takahashi, Rieko*; Asano, Hidekazu*
Corrosion Engineering, Science and Technology, 46(2), p.117 - 123, 2011/04
Times Cited Count:10 Percentile:47.91(Materials Science, Multidisciplinary)Carbon steel has been selected as one of the candidate materials for overpack for geological disposal of high-level radioactive waste in Japan. Corrosion of carbon steel is divided into two types; general corrosion and localized corrosion. In this study, propagation behaviors of general and localized corrosions (pitting corrosion and crevice corrosion) were investigated by immersion tests of carbon steel under aerobic condition. The results of the immersion tests showed that the growth rate of corrosion was strongly dependent on the environmental condition and steel type, but the upper limit of pitting factor (the ratio of the maximum corrosion depth and the average corrosion depth) was approximately determined by only average corrosion depth. Based on these experimental data and literature data, an empirical model that predicts the maximum corrosion depth of an overpack from average corrosion depth was developed by applying the extreme value statistical analysis using the Gumbel distribution function.
Fujita, Tomoo; Taniguchi, Naoki; Matsui, Hiroya; Tanai, Kenji; Maekawa, Keisuke; Sawada, Atsushi; Makino, Hitoshi; Sasamoto, Hiroshi; Yoshikawa, Hideki; Shibata, Masahiro; et al.
JAEA-Research 2011-001, 193 Pages, 2011/03
This report summarizes the progress of research and development on geological disposal during the surface-based investigation phase (2001-2005) in the Horonobe Underground Research Laboratory project, of which aims are to apply the design methods of geological disposal and mass transport analysis to actual geological conditions obtained from the surface-based investigations in the Horonobe Underground Research Laboratory project as an example of actual geological environment.
Taniguchi, Naoki; Kawasaki, Manabu; Naito, Morimasa
Zairyo To Kankyo, 59(11), p.418 - 429, 2010/11
Immersion tests of carbon steel were performed in compacted bentonite for 10 years duration under anaerobic condition. The ferrous carbonates were identified as the corrosion product by XRD and XPS analysis in almost test cases. The amount of corrosion in high carbonate concentration was smaller than those of other test conditions throughout the test periods. Although the corrosion rate at 50 C was initially smaller than that at 80
C was initially smaller than that at 80 C, it resulted in larger value after several years. Such the effects of carbonate content and temperature on the long-term corrosion rate seemed to be correlated to the behavior of dissolution/precipitation of iron carbonate. Additionally, the correlation between initial corrosion amount and the protectiveness of corrosion product film was examined. Except for high carbonate condition, as the corrosion amount at early stage of immersion was larger, the corrosion product film tended to become more protective. The long-term corrosion depth was estimated by the extrapolation of the laboratory test results. The range of the estimated value was well agreed with that of archaeological analogue data.
C, it resulted in larger value after several years. Such the effects of carbonate content and temperature on the long-term corrosion rate seemed to be correlated to the behavior of dissolution/precipitation of iron carbonate. Additionally, the correlation between initial corrosion amount and the protectiveness of corrosion product film was examined. Except for high carbonate condition, as the corrosion amount at early stage of immersion was larger, the corrosion product film tended to become more protective. The long-term corrosion depth was estimated by the extrapolation of the laboratory test results. The range of the estimated value was well agreed with that of archaeological analogue data.
Taniguchi, Naoki; Suzuki, Hiroyuki*; Naito, Morimasa
JAEA-Research 2009-068, 31 Pages, 2010/03
Corrosion of metal is an interaction between the material and the environment, so that the corrosion behavior of carbon steel overpack might be affected by not only the environmental factors but also the material factors. In this study, the effect of general impurities in carbon steel such as C, Si, Mn, P and S on the electrochemical behavior and the corrosion rate were studied using carbonate aqueous solution and synthetic seawater. The experimental results were summarized as follows; (1) The effect of the impurities on the critical passivation current density, 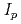 and the passive current density,
and the passive current density, 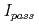 were small in 0.01M carbonate aqueous solution at pH10. (2) Breakdown of passive film and increase in anodic dissolution were observed in the tests for high Si condition of 0.73% and 0.97%. (3) In buffer material saturated with 0.01M carbonate aqueous solution, no passivation was observed and the effect of impurities on the anodic polarization behavior was small. (4) The corrosion rate of carbon steel in seawater was increased with the concentration of impurities. Among the impurity elements, the effects of P and Mn were relatively large. (5) It was inferred that the increase in corrosion rates in synthetic seawater by the addition of Si, Mn and P was promoted by the activation of hydrogen evolution reaction as a cathodic reaction.
were small in 0.01M carbonate aqueous solution at pH10. (2) Breakdown of passive film and increase in anodic dissolution were observed in the tests for high Si condition of 0.73% and 0.97%. (3) In buffer material saturated with 0.01M carbonate aqueous solution, no passivation was observed and the effect of impurities on the anodic polarization behavior was small. (4) The corrosion rate of carbon steel in seawater was increased with the concentration of impurities. Among the impurity elements, the effects of P and Mn were relatively large. (5) It was inferred that the increase in corrosion rates in synthetic seawater by the addition of Si, Mn and P was promoted by the activation of hydrogen evolution reaction as a cathodic reaction.
Taniguchi, Naoki; Kawasaki, Manabu; Naito, Morimasa
JAEA-Research 2009-067, 29 Pages, 2010/03
Since the propagation rate of stress corrosion cracking (SCC) is generally larger than that of other corrosion mode without cracking, it is difficult to avoid the penetration due to SCC by designing the corrosion allowance. Therefore, it is important to clarify the possibility of SCC initiation or conditions where SCC is possible to be occurred. It has been known that copper and copper alloys are susceptible to SCC in ammonia environment depending on the conditions. In this study, the SCC susceptibility of oxygen free copper was investigated in ammonia solution and groundwater containing ammonium ion under oxidizing condition by slow strain rate technique. As the results, no SCC was observed both in 0.05M and 0.1M NH OH solution. In Horonobe groundwater containing ammonium ion, brittle fracture surface and cracks were observed at -144mV vs. SCE. The morphologies of the SCC were not only intergranular type but also transgranular type and transgranular cracks branched from intergranular crack. In these test conditions, corrosion products were strongly adhered to the specimen surface and inside of the cracks. This indicates that the SCC was caused by tarnish rupture mechanism. In buffer material saturated with Horonobe groundwater, mechanical properties such as maximum stress and fracture strain were comparable with those in silicon oil, and no distinct cracks due to SCC were detected on the specimens.
OH solution. In Horonobe groundwater containing ammonium ion, brittle fracture surface and cracks were observed at -144mV vs. SCE. The morphologies of the SCC were not only intergranular type but also transgranular type and transgranular cracks branched from intergranular crack. In these test conditions, corrosion products were strongly adhered to the specimen surface and inside of the cracks. This indicates that the SCC was caused by tarnish rupture mechanism. In buffer material saturated with Horonobe groundwater, mechanical properties such as maximum stress and fracture strain were comparable with those in silicon oil, and no distinct cracks due to SCC were detected on the specimens.
Taniguchi, Naoki; Suzuki, Hiroyuki*; Naito, Morimasa
JAEA-Research 2009-066, 18 Pages, 2010/03
It has been known that carbon steel can be passivated in high pH environment and sometimes be attacked by localized corrosion such as pitting corrosion, crevice corrosion. If carbon steel overpack is attacked by localized corrosion, it is possible that the overpack may be penetrated in a short term, since the propagation rate of localized corrosion is larger than that of general corrosion in general. It has been known that the pitting corrosion and crevice corrosion are initiated in the presence of aggressive species for passive film represented by chloride ion. In repository environment, it is possible that the pH in groundwater containing chloride ion such as seawater type groundwater is raised by a contact with the cement material in concrete structures constructed around the engineered barrier system and then pitting corrosion or crevice corrosion is caused on the carbon steel overpacks. In this study, the propagation behavior of pitting corrosion and crevice corrosion was examined by immersion tests under air equilibrium condition using artificial groundwater at Horonobe as an example of seawater-type groundwater. As the results, the pitting factor (ratio of the maximum corrosion depth and average corrosion depth) were within the literature data obtained in neutral and alkaline water and in various natural water environments. The maximum corrosion depth of carbon steel overpack was predicted by extreme value statistical analysis of the experimental data, and it was confirmed that the predicted corrosion depths were not over the values calculated from current empirical models for propagation of pitting corrosion and crevice corrosion in every cases.