Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kimata, Tetsuya*; Kakitani, Kenta*; Yamamoto, Shunya*; Shimoyama, Iwao; Matsumura, Daiju; Iwase, Akihiro*; Mao, W.*; Kobayashi, Tomohiro*; Yamaki, Tetsuya*; Terai, Takayuki*
Physical Review Materials (Internet), 6(3), p.035801_1 - 035801_7, 2022/03
Times Cited Count:7 Percentile:44.71(Materials Science, Multidisciplinary)Tanaka, Nobuyuki; Yamaki, Tetsuya*; Asano, Masaharu*; Terai, Takayuki*
Journal of Membrane Science, 587, p.117171_1 - 117171_9, 2019/10
Times Cited Count:10 Percentile:33.86(Engineering, Chemical)Electro-electrodialysis (EED) has been applied to thermochemical water-splitting iodine-sulfur process for hydrogen production in order to enhance the HI component in the HI-I -H
-H O mixture (HIx solution). In this paper, HIx solution concentration dependence of the conductivity (
O mixture (HIx solution). In this paper, HIx solution concentration dependence of the conductivity ( ) and transport number (
) and transport number (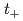 ) as the membrane-performance indexes of Nafion 212 and a styrene-grafted poly(ethylene-co-tetrafluoroethylene) (ETFE) membrane was experimentally investigated. From the theoretical analysis for the measurements based on the new EED model, the effects of the H
) as the membrane-performance indexes of Nafion 212 and a styrene-grafted poly(ethylene-co-tetrafluoroethylene) (ETFE) membrane was experimentally investigated. From the theoretical analysis for the measurements based on the new EED model, the effects of the H and I
and I content and the diffusion coefficients of H
content and the diffusion coefficients of H and I
and I in the evaluated membranes on
in the evaluated membranes on  and
and 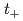 were as follows: HIx solution concentration dependence of ion contents exhibited a significantly small or different variation in comparison with that of
were as follows: HIx solution concentration dependence of ion contents exhibited a significantly small or different variation in comparison with that of  and t
and t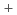 , which indicates that the dominant factors for
, which indicates that the dominant factors for  and
and 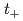 would not be ion contents; thereby, the effect of ion diffusion coefficients should not be ruled out. For
would not be ion contents; thereby, the effect of ion diffusion coefficients should not be ruled out. For  of Nafion 212, H
of Nafion 212, H diffusion is the dominant factor because the existence of I
diffusion is the dominant factor because the existence of I was negligible, whereas for the ETFE-St membrane, the effect of I
was negligible, whereas for the ETFE-St membrane, the effect of I diffusion should be considered. I
diffusion should be considered. I diffusion could more strongly affect
diffusion could more strongly affect 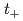 for the evaluated membranes rather than H
for the evaluated membranes rather than H diffusion, which indicates that H
diffusion, which indicates that H selectivity of the membranes could be relatively determined by the variation of the I
selectivity of the membranes could be relatively determined by the variation of the I behavior in the membranes.
behavior in the membranes.
Kakitani, Kenta*; Kimata, Tetsuya*; Yamaki, Tetsuya*; Yamamoto, Shunya*; Matsumura, Daiju; Taguchi, Tomitsugu*; Terai, Takayuki*
Radiation Physics and Chemistry, 153, p.152 - 155, 2018/12
Times Cited Count:4 Percentile:32.93(Chemistry, Physical)Wakai, Eiichi; Watanabe, Kazuyoshi*; Ito, Yuzuru*; Suzuki, Akihiro*; Terai, Takayuki*; Yagi, Juro*; Kondo, Hiroo; Kanemura, Takuji; Furukawa, Tomohiro; Hirakawa, Yasushi; et al.
Plasma and Fusion Research (Internet), 11, p.2405112_1 - 2405112_4, 2016/11
Kimata, Tetsuya*; Kato, Sho*; Yamaki, Tetsuya; Yamamoto, Shunya; Kobayashi, Tomohiro*; Terai, Takayuki*
Surface & Coatings Technology, 306(Part A), p.123 - 126, 2016/11
Times Cited Count:13 Percentile:48.74(Materials Science, Coatings & Films)Platinum (Pt) nanoparticle catalysts with oxygen reduction reaction (ORR) activity are required for practical applications of polymer electrolyte fuel cells. We prepared Pt nanoparticles on an Ar -irradiated glassy carbon (GC) surface by a radio-frequency magnetron sputtering method to investigate the influence of the ion-induced lattice defects in GC on the ORR activity of the deposited Pt nanoparticles. Interestingly, the Pt nanoparticles on the irradiated surface exhibited ca. 2.5 times higher specific activity than those on the non-irradiated one. X-ray photoelectron spectroscopy suggested the interfacial Pt-C interaction occurring between the irradiated GC and Pt nanoparticles, which should be a reason for improvement of the ORR activity.
-irradiated glassy carbon (GC) surface by a radio-frequency magnetron sputtering method to investigate the influence of the ion-induced lattice defects in GC on the ORR activity of the deposited Pt nanoparticles. Interestingly, the Pt nanoparticles on the irradiated surface exhibited ca. 2.5 times higher specific activity than those on the non-irradiated one. X-ray photoelectron spectroscopy suggested the interfacial Pt-C interaction occurring between the irradiated GC and Pt nanoparticles, which should be a reason for improvement of the ORR activity.
Ishizawa, Akihiro*; Idomura, Yasuhiro; Imadera, Kenji*; Kasuya, Naohiro*; Kanno, Ryutaro*; Satake, Shinsuke*; Tatsuno, Tomoya*; Nakata, Motoki*; Nunami, Masanori*; Maeyama, Shinya*; et al.
Purazuma, Kaku Yugo Gakkai-Shi, 92(3), p.157 - 210, 2016/03
The high-performance computer system Helios which is located at The Computational Simulation Centre (CSC) in The International Fusion Energy Research Centre (IFERC) started its operation in January 2012 under the Broader Approach (BA) agreement between Japan and the EU. The Helios system has been used for magnetised fusion related simulation studies in the EU and Japan and has kept high average usage rate. As a result, the Helios system has contributed to many research products in a wide range of research areas from core plasma physics to reactor material and reactor engineering. This project review gives a short catalogue of domestic simulation research projects. First, we outline the IFERC-CSC project. After that, shown are objectives of the research projects, numerical schemes used in simulation codes, obtained results and necessary computations in future.
 O
O thin films on Si (100) by ion beam sputter deposition
thin films on Si (100) by ion beam sputter depositionMao, W.*; Fujita, Masaya*; Chikada, Takumi*; Yamaguchi, Kenji; Suzuki, Akihiro*; Terai, Takayuki*; Matsuzaki, Hiroyuki*
Surface & Coatings Technology, 283, p.241 - 246, 2015/12
Times Cited Count:3 Percentile:12.82(Materials Science, Coatings & Films)Single-phase nanocrystalline thin films of Er O
O (440) has been first prepared using Si (100) substrates by ion beam sputter deposition at 973 K at a pressure of
(440) has been first prepared using Si (100) substrates by ion beam sputter deposition at 973 K at a pressure of  10
10 Pa and
Pa and 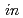 -
-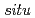 annealing at 1023 K at a pressure of
annealing at 1023 K at a pressure of  10
10 Pa. Er silicides formed during the deposition are eliminated via the annealing, which results in the single phase and the smooth surface of the Er
Pa. Er silicides formed during the deposition are eliminated via the annealing, which results in the single phase and the smooth surface of the Er O
O thin films. The epitaxial relationship between Si (100) and Er
thin films. The epitaxial relationship between Si (100) and Er O
O (110) is clarified by X-ray diffraction and reflection high energy electron diffraction.
(110) is clarified by X-ray diffraction and reflection high energy electron diffraction.
 -tetrafluoroethylene) films using different media
-tetrafluoroethylene) films using different mediaNuryanthi, N.*; Yamaki, Tetsuya; Kitamura, Akane; Koshikawa, Hiroshi; Yoshimura, Kimio; Sawada, Shinichi; Hasegawa, Shin; Asano, Masaharu; Maekawa, Yasunari; Suzuki, Akihiro*; et al.
Transactions of the Materials Research Society of Japan, 40(4), p.359 - 362, 2015/12
The ion-track grafting of a vinylbenzyl chloride (VBC) into a poly(ethylene-co-tetrafluoroethylene) (ETFE) film is necessary for preparing nanostructured hydroxide-ion-conductive electrolyte membranes. A key for success here is to obtain as high graft levels as possible (for higher conductivity) in a smaller number of tracks (for improving the other membrane properties). To this end, therefore, the effect of the medium for the VBC grafting was investigated as part of our continuing effort to optimize the experimental conditions. A 25  m-thick ETFE film was irradiated in a vacuum chamber with 560 MeV
m-thick ETFE film was irradiated in a vacuum chamber with 560 MeV  Xe at different fluences, and then the grafting was performed by immersing the irradiated films in a 20vol% VBC monomer at 60
Xe at different fluences, and then the grafting was performed by immersing the irradiated films in a 20vol% VBC monomer at 60 C. A medium was a mixture of water (H
C. A medium was a mixture of water (H O) and isopropyl alcohol (iPrOH) at different volume ratios. The degree of grafting increased as the H
O) and isopropyl alcohol (iPrOH) at different volume ratios. The degree of grafting increased as the H O content became higher, and reached a maximum in pure H
O content became higher, and reached a maximum in pure H O. These results can be explained by considering the well-known Trommsdorff effect, in which poor solubility of the grafted polymer in polar media leads to an increased polymerization rate probably due to a lower termination rate.
O. These results can be explained by considering the well-known Trommsdorff effect, in which poor solubility of the grafted polymer in polar media leads to an increased polymerization rate probably due to a lower termination rate.
Usui, Takahiro*; Sawada, Akihiko; Amaya, Masaki; Suzuki, Akihiro*; Chikada, Takumi*; Terai, Takayuki*
Journal of Nuclear Science and Technology, 52(10), p.1318 - 1322, 2015/10
Times Cited Count:55 Percentile:97.00(Nuclear Science & Technology)SiC coating is one of countermeasures for the prevention of oxidation and hydrogen embrittlement of fuel claddings because SiC has high resistance of oxidation and hydrogen permeation. Hydrogen permeation and oxidation experiments for the cladding materials with SiC coatings were conducted in unirradiated conditions. The sputtering method was employed to make SiC coatings. In the hydrogen permeation experiment, SUS316 was used as a base material of the coating. SUS316 with SiC coatings showed hydrogen permeation reduction by one order of magnitude. In the oxidation experiments, Zry-4 and SUS316 were used as base materials of the coatings. The weight gain of the Zry-4 specimens with a SiC coating decreased by about one-fifth compared to the uncoated ones. This phenomenon was observed for SUS316 at 750  C as well. The peel-off of the coating was observed in some experiments, and it is considered that the peel-off was caused by the difference of the thermal expansions between coatings and base materials. Thicker coatings showed better oxidation resistance, but thinner coatings showed more tolerance of peel-off.
C as well. The peel-off of the coating was observed in some experiments, and it is considered that the peel-off was caused by the difference of the thermal expansions between coatings and base materials. Thicker coatings showed better oxidation resistance, but thinner coatings showed more tolerance of peel-off.
 -H
-H O mixture
O mixtureTanaka, Nobuyuki; Yamaki, Tetsuya; Asano, Masaharu; Terai, Takayuki*; Onuki, Kaoru
Journal of Membrane Science, 456, p.31 - 41, 2014/04
Times Cited Count:2 Percentile:8.28(Engineering, Chemical)Electro-electrodialysis (EED) has been applied to thermochemical water-splitting iodine-sulfur process for hydrogen production in order to enhance the HI uptake from a HI-I -H
-H O mixture (HIx solution). In this paper, the constitution of the HIx solution absorbed in grafted membranes and in Nafion 212 is evaluated. The affinity of the membranes for the HIx solution is discussed based on the measurement results. The behavior of each component in the membrane was as follows: The strong affinity of the ETFE-St membrane for I
O mixture (HIx solution). In this paper, the constitution of the HIx solution absorbed in grafted membranes and in Nafion 212 is evaluated. The affinity of the membranes for the HIx solution is discussed based on the measurement results. The behavior of each component in the membrane was as follows: The strong affinity of the ETFE-St membrane for I was shown because of the formation of a charge-transfer complex with electron-donating aromatic groups, whereas I
was shown because of the formation of a charge-transfer complex with electron-donating aromatic groups, whereas I absorption by Nafion 212 was minimal. The difference of affinity for the ions can be attributed to the formation of the I
absorption by Nafion 212 was minimal. The difference of affinity for the ions can be attributed to the formation of the I - complex from I- and I
- complex from I- and I absorbed in the grafted membranes, which was precluded in Nafion 212.
absorbed in the grafted membranes, which was precluded in Nafion 212.
 O
O (001) surface; A DFT study
(001) surface; A DFT studyMao, W.*; Chikada, Takumi*; Shimura, Kenichiro*; Suzuki, Akihiro*; Yamaguchi, Kenji; Terai, Takayuki*
Journal of Nuclear Materials, 443(1-3), p.555 - 561, 2013/11
Times Cited Count:4 Percentile:30.44(Materials Science, Multidisciplinary)In this work,  calculations based on density functional theory (DFT) and generalized gradient approximation were performed to investigate the structural and electronic properties of the cubic Er
calculations based on density functional theory (DFT) and generalized gradient approximation were performed to investigate the structural and electronic properties of the cubic Er O
O (001) surface and H adsorption processes on this surface. Several stable adsorption sites were identified, and at the most energetically favorable adsorption sites it was found that H bonds with O atoms at the cubic Er
(001) surface and H adsorption processes on this surface. Several stable adsorption sites were identified, and at the most energetically favorable adsorption sites it was found that H bonds with O atoms at the cubic Er O
O (001) surface with an adsorption energy of 295.68 kJ mol
(001) surface with an adsorption energy of 295.68 kJ mol at coverage 1/8 ML, which was inclined to decrease with the increase of H coverage (
at coverage 1/8 ML, which was inclined to decrease with the increase of H coverage ( 1/4 ML). In addition, the calculations revealed that the dissociative H atom configurations have adsorption energies that are at least 152.64 kJ mol
1/4 ML). In addition, the calculations revealed that the dissociative H atom configurations have adsorption energies that are at least 152.64 kJ mol greater than the H
greater than the H molecule configurations on the surface. These results are discussed in regard of the hydrogen isotope permeation behavior in the tritium permeation barrier in a fusion reactor.
molecule configurations on the surface. These results are discussed in regard of the hydrogen isotope permeation behavior in the tritium permeation barrier in a fusion reactor.
Kato, Sho; Yamaki, Tetsuya; Yamamoto, Shunya; Hakoda, Teruyuki; Kawaguchi, Kazuhiro; Kobayashi, Tomohiro*; Suzuki, Akihiro*; Terai, Takayuki*
Nuclear Instruments and Methods in Physics Research B, 314, p.149 - 152, 2013/11
Times Cited Count:3 Percentile:25.62(Instruments & Instrumentation)We implanted 100 keV W in unpolished GC substrates at nominal fluences up to
in unpolished GC substrates at nominal fluences up to 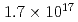 ions/cm
ions/cm . The implanted samples were electrochemically anodized in a NaOH aqueous solution to etch the surface layer. The analyses were performed by X-ray photoelectron spectroscopy (XPS), Rutherford backscattering spectrometry (RBS), and transmission electron microscopy (TEM). XPS W 4f spectra indicated the formation of carbides as reported previously. The electrochemical etching clearly increased the W concentration on the surface. According to the RBS results, half of the implanted W atoms were retained in the substrate, while the rest should escape to the etching solution. The cross-sectional TEM image revealed a uniform distribution of WC particles with a diameter of less than 10 nm just near the surface region.
. The implanted samples were electrochemically anodized in a NaOH aqueous solution to etch the surface layer. The analyses were performed by X-ray photoelectron spectroscopy (XPS), Rutherford backscattering spectrometry (RBS), and transmission electron microscopy (TEM). XPS W 4f spectra indicated the formation of carbides as reported previously. The electrochemical etching clearly increased the W concentration on the surface. According to the RBS results, half of the implanted W atoms were retained in the substrate, while the rest should escape to the etching solution. The cross-sectional TEM image revealed a uniform distribution of WC particles with a diameter of less than 10 nm just near the surface region.
Kitamura, Akane; Kobayashi, Tomohiro*; Sato, Takahiro; Koka, Masashi; Kamiya, Tomihiro; Suzuki, Akihiro*; Terai, Takayuki*
Nuclear Instruments and Methods in Physics Research B, 307, p.614 - 617, 2013/07
Times Cited Count:7 Percentile:46.40(Instruments & Instrumentation)Kato, Sho; Yamaki, Tetsuya; Yamamoto, Shunya; Hakoda, Teruyuki; Kawaguchi, Kazuhiro; Kobayashi, Tomohiro*; Suzuki, Akihiro*; Terai, Takayuki*
Transactions of the Materials Research Society of Japan, 38(1), p.81 - 84, 2013/03
Nanoparticles were formed by 100 keV tungsten-ion implantation in unpolished glassy carbon substrates at nominal fluences of 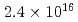 -
- 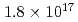 ions/cm
ions/cm . The implanted samples were analyzed by Rutherford backscattering spectrometry, X-ray photoelectron spectroscopy, hydrodynamic voltammetry using a rotating disk electrode, and field emission scanning electron microscopy. A significant sputtering effect changed the depth profile during the course of irradiation and limited the amount of tungsten retainable in the substrate in agreement with our calculated distributions. The nanoparticles were composed of tungsten carbide and dispersed uniformly with diameters of around 10 nm.
. The implanted samples were analyzed by Rutherford backscattering spectrometry, X-ray photoelectron spectroscopy, hydrodynamic voltammetry using a rotating disk electrode, and field emission scanning electron microscopy. A significant sputtering effect changed the depth profile during the course of irradiation and limited the amount of tungsten retainable in the substrate in agreement with our calculated distributions. The nanoparticles were composed of tungsten carbide and dispersed uniformly with diameters of around 10 nm.
Mao, W.*; Chikada, Takumi*; Suzuki, Akihiro*; Terai, Takayuki*; Yamaguchi, Kenji
Journal of Plasma and Fusion Research SERIES, Vol.10, p.27 - 32, 2013/02
A tritium permeation barrier (TPB) is strongly required in fusion blankets for the reduction of loss of fuel and radiological hazard. However, the precise tritium permeation mechanism through the TPB coatings has not been clarified yet, because of their complicated crystal structures. To understand the microscopic mechanism, we have not only prepared and characterized nanostructured ceramic Er O
O thin films, but also studied the energetics and mobility of hydrogen atom in cubic bulk Er
thin films, but also studied the energetics and mobility of hydrogen atom in cubic bulk Er O
O using ab initio density-functional calculations. The estimated diffusion activation energy (
using ab initio density-functional calculations. The estimated diffusion activation energy (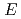
 ) of interstitial H is somewhat higher than the diffusion energy barrier observed experimentally at 873 K. It is then considered that diffusion and permeation of hydrogen and its isotopes through the Er
) of interstitial H is somewhat higher than the diffusion energy barrier observed experimentally at 873 K. It is then considered that diffusion and permeation of hydrogen and its isotopes through the Er O
O coatings are likely to be dominated by the grain boundary rather than by the grain.
coatings are likely to be dominated by the grain boundary rather than by the grain.
Wakai, Eiichi; Kondo, Hiroo; Sugimoto, Masayoshi; Fukada, Satoshi*; Yagi, Juro*; Ida, Mizuho; Kanemura, Takuji; Furukawa, Tomohiro; Hirakawa, Yasushi; Watanabe, Kazuyoshi; et al.
Purazuma, Kaku Yugo Gakkai-Shi, 88(12), p.691 - 705, 2012/12
no abstracts in English
 -H
-H O mixture for IS process
O mixture for IS processTanaka, Nobuyuki; Yamaki, Tetsuya; Asano, Masaharu; Terai, Takayuki*; Onuki, Kaoru
Proceedings of 6th International Topical Meeting on High Temperature Reactor Technology (HTR 2012) (USB Flash Drive), 6 Pages, 2012/10
Research and development of a water-splitting hydrogen-production method, called iodine-sulfur (IS) process, has been conducting as one of the heat applications of high temperature gas-cooled reactor. Among the unit operations in this IS process, we have investigated electro-electrodialysis (EED) using an ion-exchange membrane to concentrate HI in an HI-I -H
-H O mixture. Aiming at maximizing EED performance, new membrane materials were prepared by the radiation-induced graft polymerization and were examined in terms of their proton permeability through the membrane, i.e., transport number and conductivity at different iodine (I
O mixture. Aiming at maximizing EED performance, new membrane materials were prepared by the radiation-induced graft polymerization and were examined in terms of their proton permeability through the membrane, i.e., transport number and conductivity at different iodine (I ) concentrations in the HI-I
) concentrations in the HI-I -H
-H O mixture. The transport number increased and the conductivity decreased with an increase in the feed I
O mixture. The transport number increased and the conductivity decreased with an increase in the feed I molality. The EED model derived by the Nernst-Planck theory suggested that this trend could be explained exclusively by the variation of diffusion coefficient of I
molality. The EED model derived by the Nernst-Planck theory suggested that this trend could be explained exclusively by the variation of diffusion coefficient of I .
.
 -H
-H O mixture using ion exchange membranes
O mixture using ion exchange membranesTanaka, Nobuyuki; Yamaki, Tetsuya; Asano, Masaharu; Maekawa, Yasunari; Terai, Takayuki*; Onuki, Kaoru
Journal of Membrane Science, 411-412, p.99 - 108, 2012/09
Times Cited Count:15 Percentile:43.93(Engineering, Chemical)In the thermochemical water-splitting iodine-sulfur process for hydrogen production, an electro-electrodialysis was applied to increase the HI molality of a HI-I -H
-H O mixture. In order to investigate the temperature dependence of the membrane performance for increasing the HI concentration, a theoretical formula for the performance indexes (proton transport number, initial cell voltage, and ratio of water permeation to proton permeation in the membrane) was derived on the basis of the Nernst-Planck equation and electrophoresis theory. The obtained formula could reproduce the experimental values of the grafted membranes and Nafion 212 in the temperature range 313-373 K and was validated.
O mixture. In order to investigate the temperature dependence of the membrane performance for increasing the HI concentration, a theoretical formula for the performance indexes (proton transport number, initial cell voltage, and ratio of water permeation to proton permeation in the membrane) was derived on the basis of the Nernst-Planck equation and electrophoresis theory. The obtained formula could reproduce the experimental values of the grafted membranes and Nafion 212 in the temperature range 313-373 K and was validated.
Kondo, Hiroo; Furukawa, Tomohiro; Hirakawa, Yasushi; Nakamura, Kazuyuki; Ida, Mizuho; Watanabe, Kazuyoshi; Kanemura, Takuji; Wakai, Eiichi; Horiike, Hiroshi*; Yamaoka, Nobuo*; et al.
Nuclear Fusion, 51(12), p.123008_1 - 123008_12, 2011/12
Times Cited Count:40 Percentile:81.74(Physics, Fluids & Plasmas)The Engineering Validation and Engineering Design Activity (EVEDA) for the International Fusion Materials Irradiation Facility (IFMIF) is proceeded as one of the ITER Broader Approach (BA) activities. The EVEDA Li test loop (ELTL) is aimed at validating stability of the Li target and feasibility of a Li purification system as the key issues. In this paper, the design of the ELTL especially of a target assembly in which the Li target is produced by the contraction nozzle is presented.
Nakamura, Kazuyuki; Furukawa, Tomohiro; Hirakawa, Yasushi; Kanemura, Takuji; Kondo, Hiroo; Ida, Mizuho; Niitsuma, Shigeto; Otaka, Masahiko; Watanabe, Kazuyoshi; Horiike, Hiroshi*; et al.
Fusion Engineering and Design, 86(9-11), p.2491 - 2494, 2011/10
Times Cited Count:11 Percentile:58.47(Nuclear Science & Technology)In IFMIF/EVEDA, tasks for lithium target system are shared to 5 validation tasks (LF1-5) and a design task (LF6). The purpose of LF1 task is to construct and operate the EVEDA lithium test loop, and JAEA has a main responsibility to the performance of the Li test loop. LF2 is a task for the diagnostics of the Li test loop and IFMIF design. Basic research for the diagnostics equipment has been completed, and the construction for the Li test loop will be finished before March in 2011. LF4 is a task for the purification systems with nitrogen and hydrogen. Basic research for the purification equipment has been completed, and the construction of the nitrogen system for the Li test loop will be finished before March in 2011. LF5 is a task for the remote handling system with the target assembly. JAEA has an idea to use the laser beam for cutting and welding of the lip part of the flanges. LF6 is a task for the design of the IFMIF based on the validation experiments of LF1-5.