Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kaneda-Nakashima, Kazuko*; Shirakami, Yoshifumi*; Kadonaga, Yuichiro*; Watabe, Tadashi*; Oe, Kazuhiro*; Yin, X.*; Haba, Hiromitsu*; Shirasaki, Kenji*; Kikunaga, Hidetoshi*; Tsukada, Kazuaki; et al.
International Journal of Molecular Sciences (Internet), 25(2), p.933_1 - 933_14, 2024/01
Times Cited Count:1 Percentile:0.01(Biochemistry & Molecular Biology)Yakushev, A.*; Lens, L.*; D llmann, Ch. E.*; Khuyagbaatar, J.*; J
llmann, Ch. E.*; Khuyagbaatar, J.*; J ger, E.*; Krier, J.*; Runke, J.*; Albers, H. M.*; Asai, Masato; Block, M.*; et al.
ger, E.*; Krier, J.*; Runke, J.*; Albers, H. M.*; Asai, Masato; Block, M.*; et al.
Frontiers in Chemistry (Internet), 10, p.976635_1 - 976635_11, 2022/08
Times Cited Count:9 Percentile:79.28(Chemistry, Multidisciplinary)Flerovium (Fl, element 114) is the heaviest element chemically studied so far. The first chemical experiment on Fl suggested that Fl is a noble-gas-like element, while the second studies suggested that Fl has a volatile-metal-like character. To obtain more reliable conclusion, we performed further experimental studies on Fl adsorption behavior on Si oxide and gold surfaces. The present results suggest that Fl is highly volatile and less reactive than the volatile metal, Hg, but has higher reactivity than the noble gas, Rn.
 At]NaAt and [
At]NaAt and [ I]NaI in an NIS-expressing thyroid cancer mouse model
I]NaI in an NIS-expressing thyroid cancer mouse modelWatabe, Tadashi*; Liu, Y.*; Kaneda, Kazuko*; Sato, Tatsuhiko; Shirakami, Yoshifumi*; Oe, Kazuhiro*; Toyoshima, Atsushi*; Shimosegawa, Eku*; Wang, Y.*; Haba, Hiromitsu*; et al.
International Journal of Molecular Sciences (Internet), 23(16), p.9434_1 - 9434_11, 2022/08
Times Cited Count:7 Percentile:78.48(Biochemistry & Molecular Biology)In this study, we compare the therapeutic effect between [ At]NaAt and [
At]NaAt and [ I]NaI. In vitro analysis of double-stranded DNA break (DSB) and colony formation assay were performed using K1-NIS cells. [
I]NaI. In vitro analysis of double-stranded DNA break (DSB) and colony formation assay were performed using K1-NIS cells. [ At]NaAt induced higher numbers of DSBs and had a reduced colony formation than [
At]NaAt induced higher numbers of DSBs and had a reduced colony formation than [ I]NaI. In K1-NIS mice, dose-dependent therapeutic effects were observed in both [
I]NaI. In K1-NIS mice, dose-dependent therapeutic effects were observed in both [ At]NaAt and [
At]NaAt and [ I]NaI. The superior therapeutic effect of [
I]NaI. The superior therapeutic effect of [ At]NaAt suggests the promising clinical applicability of targeted alpha therapy using [
At]NaAt suggests the promising clinical applicability of targeted alpha therapy using [ At]NaAt in patients with differentiated thyroid cancer refractory to standard [
At]NaAt in patients with differentiated thyroid cancer refractory to standard [ I]NaI treatment.
I]NaI treatment.
G tz, M.*; Yakushev, A.*; G
tz, M.*; Yakushev, A.*; G tz, S.*; Di Nitto, A.*; D
tz, S.*; Di Nitto, A.*; D llmann, Ch. E.*; Asai, Masato; Kindler, B.*; Krier, J.*; Lommel, B.*; Nagame, Yuichiro*; et al.
llmann, Ch. E.*; Asai, Masato; Kindler, B.*; Krier, J.*; Lommel, B.*; Nagame, Yuichiro*; et al.
Radiochimica Acta, 110(2), p.75 - 86, 2022/02
Times Cited Count:2 Percentile:31.78(Chemistry, Inorganic & Nuclear)The study of volatile superheavy element carbonyl complexes requires more efficient methods because the yield of transactinide elements decreases with increasing atomic number. This is achieved by using a newly developed double chamber system to separate the recoil chamber and the reaction one, thereby avoiding the decomposition of reactive molecules by the projectile ion beam, which hinders the synthesis of carbonyl complexes. The feasibility of this method was verified by synthesizing 5d metal short-lived isotopes as homologous element isotopes of the light transactinide elements Sg, Bh, Hs, and Mt at the Japan Atomic Energy Agency tandem accelerator and conducting model experiments.
Yakushev, A.*; Lens, L.*; D llmann, C. E.*; Block, M.*; Nagame, Yuichiro*; Sato, Tetsuya; Toyoshima, Atsushi*; 42 of others*
llmann, C. E.*; Block, M.*; Nagame, Yuichiro*; Sato, Tetsuya; Toyoshima, Atsushi*; 42 of others*
Frontiers in Chemistry (Internet), 9, p.753738_1 - 753738_9, 2021/11
Times Cited Count:10 Percentile:63.67(Chemistry, Multidisciplinary)Nihonium (Nh, element 113) and flerovium (Fl, element 114) are the first superheavy elements in which the 7p shell is occupied. High volatility and inertness were predicted for Fl due to the strong relativistic stabilization of the closed 7p sub-shell, which originates from a large spin-orbit splitting between the 7p
sub-shell, which originates from a large spin-orbit splitting between the 7p and 7p
and 7p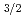 orbitals. One unpaired electron in the outermost 7p
orbitals. One unpaired electron in the outermost 7p sub-shell in Nh is expected to give rise to a higher chemical reactivity. Theoretical predictions of Nh reactivity are discussed, along with results of the first experimental attempts to study Nh chemistry in the gas phase. The experimental observations verify a higher chemical reactivity of Nh atoms compared to its neighbor Fl and call for the development of advanced setups. First tests of a newly developed detection device miniCOMPACT with highly reactive Fr isotopes assure that effective chemical studies of Nh are within reach.
sub-shell in Nh is expected to give rise to a higher chemical reactivity. Theoretical predictions of Nh reactivity are discussed, along with results of the first experimental attempts to study Nh chemistry in the gas phase. The experimental observations verify a higher chemical reactivity of Nh atoms compared to its neighbor Fl and call for the development of advanced setups. First tests of a newly developed detection device miniCOMPACT with highly reactive Fr isotopes assure that effective chemical studies of Nh are within reach.
Ikebe, Yurie*; Oshima, Masumi*; Bamba, Shigeru*; Asai, Masato; Tsukada, Kazuaki; Sato, Tetsuya; Toyoshima, Atsushi*; Bi, C.*; Seto, Hirofumi*; Amano, Hikaru*; et al.
Applied Radiation and Isotopes, 164, p.109106_1 - 109106_7, 2020/10
Times Cited Count:2 Percentile:24.28(Chemistry, Inorganic & Nuclear)Boron Neutron Capture Therapy (BNCT) is a radiotherapy for the treatment of intractable cancer. In BNCT precise determination of  B concentration in whole blood sample before neutron irradiation is crucial for control of the neutron irradiation time and the neutron dosimetry. We have applied the Charged Particle Activation Analysis (CPAA) to non-destructive and accurate determination of
B concentration in whole blood sample before neutron irradiation is crucial for control of the neutron irradiation time and the neutron dosimetry. We have applied the Charged Particle Activation Analysis (CPAA) to non-destructive and accurate determination of  B concentration in whole blood sample. The experiment was performed at JAEA Tandem Accelerator using an 8 MeV proton beam. The 478 keV
B concentration in whole blood sample. The experiment was performed at JAEA Tandem Accelerator using an 8 MeV proton beam. The 478 keV  ray of
ray of  Be produced in the
Be produced in the  B(p,
B(p, )
) Be reaction was used to quantify the
Be reaction was used to quantify the  B, and
B, and  rays of
rays of 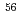 Co originating from the reaction with Fe in blood was used to normalize the
Co originating from the reaction with Fe in blood was used to normalize the  -ray intensity. The results demonstrated that the present CPAA method can be applied to the determination of the
-ray intensity. The results demonstrated that the present CPAA method can be applied to the determination of the  B concentration in the blood sample.
B concentration in the blood sample.
 Bh in the
Bh in the  Cm(
Cm(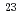 Na,5
Na,5 )
) Bh reaction and its decay properties
Bh reaction and its decay propertiesHaba, Hiromitsu*; Fan, F.*; Kaji, Daiya*; Kasamatsu, Yoshitaka*; Kikunaga, Hidetoshi*; Komori, Yukiko*; Kondo, Narumi*; Kudo, Hisaaki*; Morimoto, Koji*; Morita, Kosuke*; et al.
Physical Review C, 102(2), p.024625_1 - 024625_12, 2020/08
Times Cited Count:6 Percentile:59.56(Physics, Nuclear) decay of
decay of 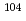 Te with a novel recoil-decay scintillation detector
Te with a novel recoil-decay scintillation detectorXiao, Y.*; Go, S.*; Grzywacz, R.*; Orlandi, R.; Andreyev, A. N.; Asai, Masato; Bentley, M. A.*; de Angelis, G.*; Gross, C. J.*; Hausladen, P.*; et al.
Physical Review C, 100(3), p.034315_1 - 034315_8, 2019/09
Times Cited Count:16 Percentile:84.48(Physics, Nuclear)Chiera, N. M.; Sato, Tetsuya; Tomitsuka, Tomohiro; Asai, Masato; Ito, Yuta; Shirai, Kaori*; Suzuki, Hayato; Tokoi, Katsuyuki; Toyoshima, Atsushi; Tsukada, Kazuaki; et al.
Journal of Radioanalytical and Nuclear Chemistry, 320(3), p.633 - 642, 2019/06
Times Cited Count:1 Percentile:11.15(Chemistry, Analytical)An isothermal gas-chromatographic (IGC) device has been developed and tested for on-line gas phase studies of volatile oxychlorides of short-lived group-5 transition metals. Radioisotopes of niobium and tantalum, produced in nuclear fusion evaporation reactions, are directly flushed into the IGC setup by an inert gas-jet. Oxychloride compounds are formed by the addition of SOCl and O
and O . Parameters influencing the formation and transport of NbOCl
. Parameters influencing the formation and transport of NbOCl and TaOCl
and TaOCl are investigated. For nuclides with half-lives (
are investigated. For nuclides with half-lives (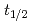 ) of about 30 s, an overall efficiency of 7% is obtained, rendering the IGC setup suitable for the chemical exploration of
) of about 30 s, an overall efficiency of 7% is obtained, rendering the IGC setup suitable for the chemical exploration of  Db(
Db(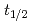 = 34s).
= 34s).
Chiera, N. M.; Sato, Tetsuya; Tomitsuka, Tomohiro; Asai, Masato; Suzuki, Hayato*; Tokoi, Katsuyuki; Toyoshima, Atsushi; Tsukada, Kazuaki; Nagame, Yuichiro
Inorganica Chimica Acta, 486, p.361 - 366, 2019/02
Times Cited Count:3 Percentile:18.6(Chemistry, Inorganic & Nuclear)The formation of NbOCl and TaOCl
and TaOCl and their adsorption behavior on quartz surfaces was explored by applying an isothermal gas-chromatographic method. Trace amounts of short-lived Nb and Ta isotopes were used. Adsorption enthalpy values (
and their adsorption behavior on quartz surfaces was explored by applying an isothermal gas-chromatographic method. Trace amounts of short-lived Nb and Ta isotopes were used. Adsorption enthalpy values ( ) at zero surface coverage of -
) at zero surface coverage of - (NbOCl
(NbOCl ) = 102
) = 102  4 kJ/mol and -
4 kJ/mol and - (TaOCl
(TaOCl ) = 128
) = 128  5 kJ/mol were determined by analyzing the chromatographic behavior of the Nb andTa complexes with a Monte-Carlo simulation method based on an adsorption-desorption kinetic model.By applying an empirical correlation, the experimental
5 kJ/mol were determined by analyzing the chromatographic behavior of the Nb andTa complexes with a Monte-Carlo simulation method based on an adsorption-desorption kinetic model.By applying an empirical correlation, the experimental  values were successively related to the macroscopic standard sublimation enthalpy,
values were successively related to the macroscopic standard sublimation enthalpy, 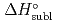 , as a measure of the volatility of each substance. The inferred sublimation enthalpies are in agreement with tabulated thermochemical values. Thus, the linear empirical correlation between
, as a measure of the volatility of each substance. The inferred sublimation enthalpies are in agreement with tabulated thermochemical values. Thus, the linear empirical correlation between  and
and 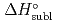 for metal-oxychlorides was updated with the inclusion of the present data. According to the predicted
for metal-oxychlorides was updated with the inclusion of the present data. According to the predicted 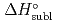 (DbOCl
(DbOCl ), a
), a  (DbOCl
(DbOCl ) value of 135
) value of 135  2 kJ/mol was extrapolated. The future accomplishment of comparative studies with DbOCl
2 kJ/mol was extrapolated. The future accomplishment of comparative studies with DbOCl under the same experimental conditions will provide valuable information on the volatility trend in Group-5 elements, together with an indication on the magnitude of relativistic effects on the electronic structure of dubnium.
under the same experimental conditions will provide valuable information on the volatility trend in Group-5 elements, together with an indication on the magnitude of relativistic effects on the electronic structure of dubnium.
 acidic solutions
acidic solutionsYokoyama, Akihiko*; Kitayama, Yuta*; Fukuda, Yoshiki*; Kikunaga, Hidetoshi*; Murakami, Masashi*; Komori, Yukiko*; Yano, Shinya*; Haba, Hiromitsu*; Tsukada, Kazuaki; Toyoshima, Atsushi*
Radiochimica Acta, 107(1), p.27 - 32, 2019/01
Times Cited Count:1 Percentile:11.15(Chemistry, Inorganic & Nuclear)Hatsukawa, Yuichi*; Hayakawa, Takehito*; Tsukada, Kazuaki; Hashimoto, Kazuyuki*; Sato, Tetsuya; Asai, Masato; Toyoshima, Atsushi; Tanimori, Toru*; Sonoda, Shinya*; Kabuki, Shigeto*; et al.
PLOS ONE (Internet), 13(12), p.e0208909_1 - e0208909_12, 2018/12
Times Cited Count:3 Percentile:30.05(Multidisciplinary Sciences)Imaging of 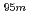 Tc radioisotope was conducted using an electron tracking-Compton camera (ETCC).
Tc radioisotope was conducted using an electron tracking-Compton camera (ETCC). 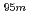 Tc emits 204, 582, and 835 keV
Tc emits 204, 582, and 835 keV  rays, and was produced in the
rays, and was produced in the 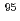 Mo(p,n)
Mo(p,n)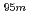 Tc reaction with a
Tc reaction with a 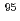 Mo-enriched target. The recycling of the
Mo-enriched target. The recycling of the 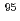 Mo-enriched molybdenum trioxide was investigated, and the recycled yield of
Mo-enriched molybdenum trioxide was investigated, and the recycled yield of 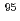 Mo was achieved to be 70% - 90%. The images were obtained with each of the three
Mo was achieved to be 70% - 90%. The images were obtained with each of the three  rays. Results showed that the spatial resolution increases with increasing
rays. Results showed that the spatial resolution increases with increasing  -ray energy, and suggested that the ETCC with high-energy
-ray energy, and suggested that the ETCC with high-energy  -ray emitters such as
-ray emitters such as 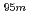 Tc is useful for the medical imaging of deep tissue and organs in the human body.
Tc is useful for the medical imaging of deep tissue and organs in the human body.
Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Beerwerth, R.*; Kaneya, Yusuke*; Makii, Hiroyuki; Mitsukai, Akina*; Nagame, Yuichiro; Osa, Akihiko; Toyoshima, Atsushi; et al.
Journal of the American Chemical Society, 140(44), p.14609 - 14613, 2018/11
Times Cited Count:27 Percentile:69.46(Chemistry, Multidisciplinary)The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
 SO
SO and HF/HCl solutions into toluene with Aliquat336; Sulfate and fluoride complex formation of Mo and W towards chemical studies of seaborgium (Sg)
and HF/HCl solutions into toluene with Aliquat336; Sulfate and fluoride complex formation of Mo and W towards chemical studies of seaborgium (Sg)Toyoshima, Atsushi; Mitsukai, Akina; Tsukada, Kazuaki; Oe, Kazuhiro*; Haba, Hiromitsu*; Komori, Yukiko*; Murakami, Masashi; Kaneya, Yusuke*; Sato, Daisuke*; Asai, Masato; et al.
Journal of Radioanalytical and Nuclear Chemistry, 317(1), p.421 - 430, 2018/07
Times Cited Count:2 Percentile:20.93(Chemistry, Analytical)We have studied extraction behavior of group-6 elements Mo and W to search for suitable conditions for an on-line extraction experiment of their heavier homolog, seaborgium (Sg). Batch-wise extraction of carrier-free radiotracers  Mo and
Mo and 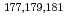 W were carried out from 0.10 - 8.6 M H
W were carried out from 0.10 - 8.6 M H SO
SO and 1.0
and 1.0 10
10 - 5.0 M HF/1.0 M HCl into toluene with a quaternary ammonium compound, Aliquat336. Anionic sulfate complexes of Mo and W with charge - 2 were extracted with Aliquat336 from H
- 5.0 M HF/1.0 M HCl into toluene with a quaternary ammonium compound, Aliquat336. Anionic sulfate complexes of Mo and W with charge - 2 were extracted with Aliquat336 from H SO
SO solutions with concentrations of [H
solutions with concentrations of [H SO
SO ]
]  2 M. In HF/1.0 M HCl, oxyfluoro complexes of Mo and W with charge - 1 were interpreted to be formed and extracted with Aliquat336. From these results, favorable conditions for the extraction of Sg are discussed.
2 M. In HF/1.0 M HCl, oxyfluoro complexes of Mo and W with charge - 1 were interpreted to be formed and extracted with Aliquat336. From these results, favorable conditions for the extraction of Sg are discussed.
Nishio, Katsuhisa; Hirose, Kentaro; Vermeulen, M. J.; Makii, Hiroyuki; Orlandi, R.; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Sato, Tetsuya; Nagame, Yuichiro; et al.
EPJ Web of Conferences, 163, p.00041_1 - 00041_6, 2017/11
Times Cited Count:1 Percentile:61.21(Nuclear Science & Technology)Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Stora, T.*; Sato, Nozomi*; Kaneya, Yusuke; Tsukada, Kazuaki; D llmann, C. E.*; Eberhardt, K.*; Eliav, E.*; et al.
llmann, C. E.*; Eberhardt, K.*; Eliav, E.*; et al.
EPJ Web of Conferences, 131, p.05001_1 - 05001_6, 2016/12
Times Cited Count:0 Percentile:0.9(Chemistry, Inorganic & Nuclear)Ionization efficiency in a surface ionization process depends on the first ionization potential of the atom. Based on the dependence, the ionization potential of the atom can be determined. We measured ionization efficiencies of fermium, einsteinium, mendelevium, and lawrencium by using a newly developed gas-jet coupled surface ion-source. The ionization potential of the elements have not been determined so far due to their low production rates and/or their short half-lives. Based on a relationship between the ionization efficiency and the ionization potential obtained via measurements of short-lived lanthanide isotopes, the ionization potentials of these actinide elements have been successfully determined.
Eichler, R.*; Asai, Masato; Brand, H.*; Chiera, N. M.*; Di Nitto, A.*; Dressler, R.*; D llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
EPJ Web of Conferences, 131, p.07005_1 - 07005_7, 2016/12
Times Cited Count:3 Percentile:72.98(Chemistry, Inorganic & Nuclear)In recent years gas-phase chemical studies assisted by physical pre-separation allowed for the productions and investigations of fragile single molecular species of superheavy elements. The latest highlight is the formation of very volatile hexacarbonyl compound of element 106, Sg(CO) . Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO)
. Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO) , W(CO)
, W(CO) , and Sg(CO)
, and Sg(CO) .
.
 at the single-atom level
at the single-atom levelSteinegger, P.*; Asai, Masato; Dressler, R.*; Eichler, R.*; Kaneya, Yusuke*; Mitsukai, Akina*; Nagame, Yuichiro; Piguet, D.*; Sato, Tetsuya; Sch del, M.; et al.
del, M.; et al.
Journal of Physical Chemistry C, 120(13), p.7122 - 7132, 2016/04
Times Cited Count:23 Percentile:60.73(Chemistry, Physical)A new experimental method "vacuum chromatography" has been developed to measure adsorption enthalpy of superheavy elements, and its feasibility has been examined using short-lived thallium isotopes. The short-lived thallium isotopes were produced at the JAEA tandem accelerator. The thallium ion beam prepared with an on-line isotope separator which ionized and mass-separated the thallium isotopes was injected into an isothermal vacuum chromatography apparatus. A temperature-dependent adsorption property of thallium atom on SiO surface were measured. The adsorption enthalpy of thallium was determined to be 158 kJ/mol. The thallium is a homolog of element 113. Thus, the vacuum chromatography developed in this study enables us to perform chemical experiments for short-lived superheavy elements with half-lives of a order of one second.
surface were measured. The adsorption enthalpy of thallium was determined to be 158 kJ/mol. The thallium is a homolog of element 113. Thus, the vacuum chromatography developed in this study enables us to perform chemical experiments for short-lived superheavy elements with half-lives of a order of one second.
 and W(CO)
and W(CO)
Usoltsev, I.*; Eichler, R.*; Wang, Y.*; Even, J.*; Yakushev, A.*; Haba, Hiromitsu*; Asai, Masato; Brand, H.*; Di Nitto, A.*; D llmann, Ch. E.*; et al.
llmann, Ch. E.*; et al.
Radiochimica Acta, 104(3), p.141 - 151, 2016/03
Times Cited Count:31 Percentile:95.03(Chemistry, Inorganic & Nuclear)Conditions of the production and decomposition of hexacarbonyl complexes of short-lived Mo and W isotopes were investigated to study thermal stability of the heaviest group 6 hexacarbonyl complex Sg(CO) . A tubular flow reactor was tested to decompose the hexacarbonyl complexes and to extract the first bond dissociation energies. A silver was found to be the most appropriate reaction surface to study the decomposition of the group 6 hexacarbonyl. It was found that the surface temperature at which the decomposition occurred was correlated to the first bond dissociation energy of Mo(CO)
. A tubular flow reactor was tested to decompose the hexacarbonyl complexes and to extract the first bond dissociation energies. A silver was found to be the most appropriate reaction surface to study the decomposition of the group 6 hexacarbonyl. It was found that the surface temperature at which the decomposition occurred was correlated to the first bond dissociation energy of Mo(CO) and W(CO)
and W(CO) , indicating that the first bond dissociation energy of Sg(CO)
, indicating that the first bond dissociation energy of Sg(CO) could be determined with this technique.
could be determined with this technique.
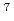 Li +
Li + 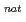 Pb reaction
Pb reactionNishinaka, Ichiro; Yokoyama, Akihiko*; Washiyama, Koshin*; Maeda, Eita*; Watanabe, Shigeki; Hashimoto, Kazuyuki; Ishioka, Noriko; Makii, Hiroyuki; Toyoshima, Atsushi; Yamada, Norihiro*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 304(3), p.1077 - 1083, 2015/06
Times Cited Count:9 Percentile:60.64(Chemistry, Analytical)Production cross sections of astatine isotopes 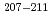 At in the 29-57 MeV
At in the 29-57 MeV 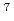 Li induced reaction with
Li induced reaction with 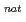 Pb target have been measured by
Pb target have been measured by  - and
- and  -ray spectrometry. Excitation functions of production cross sections have been compared with a statistical model calculation to study the reaction mechanism of
-ray spectrometry. Excitation functions of production cross sections have been compared with a statistical model calculation to study the reaction mechanism of 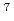 Li +
Li + 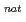 Pb. Considerably small experimental cross sections of
Pb. Considerably small experimental cross sections of 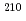 At and
At and 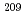 At compared with the calculation were clearly observed at incident energies higher than 44 MeV, indicating that the effects of breakup reaction play a role. A chemical separation of astatine from an irradiated lead target has been studied with a dry-distillation method. A complementary way to produce astatine isotopes has been developed.
At compared with the calculation were clearly observed at incident energies higher than 44 MeV, indicating that the effects of breakup reaction play a role. A chemical separation of astatine from an irradiated lead target has been studied with a dry-distillation method. A complementary way to produce astatine isotopes has been developed.