Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tsurumi, Miwa*; Enoto, Teruaki*; Ikkatai, Yuko*; Wu, T.*; Wang, D.*; Shinoda, Taro*; Nakazawa, Kazuhiro*; Tsuji, Naoki*; Diniz, G.*; Kataoka, Jun*; et al.
Geophysical Research Letters, 50(13), p.e2023GL103612_1 - e2023GL103612_9, 2023/07
Times Cited Count:0 Percentile:0(Geosciences, Multidisciplinary)Horiguchi, Naoki; Yoshida, Hiroyuki; Kitatsuji, Yoshihiro; Hasegawa, Makoto*; Kishimoto, Tadafumi*
Proceedings of 30th International Conference on Nuclear Engineering (ICONE30) (Internet), 7 Pages, 2023/05
From the viewpoint of energy security in Japan and reduction of the environmental load, continuous operation of light water reactors is essential. Since a pH adjuster with enriched Li-7 ions is required for water quality control on PWR, the development of Li-7 enrichment technology is one of the key issues. The multi-channel counter-current electrophoresis (MCCCE) method has been developed as the technology with a low environmental load. To put this method into practical use, it is necessary to understand Li-7 ion behavior in the channel flow and optimize the experimental condition to separate Li-7 and its isotope. In this paper, to understand Li-7 ion behavior in a single channel of the experimental apparatus, a numerical simulation method based on a computational fluid dynamics (CFD) code with a particle tracking method, TPFIT-LPT, was developed. In the method, the motion of multiple ions under the electric field was simulated as a particle with an added velocity by the electric field. The difference in the isotopes was represented by changing of the magnitude of the added velocity. We also considered that although it is impossible to measure the behavior of each ion, it is important to measure the flow velocity of the bulk fluid for the validation of the numerical simulation. We developed a lab-scale experimental apparatus in which the single channel of the actual apparatus was simplified to measure the flow velocity by Particle Image Velocimetry (PIV). We set a pulsation flow condition on the lab-scale experiment, which is one of difficult conditions for the numerical simulation, and measured the velocity. As the result, we confirmed that the pulsation flow was reproduced. We set the measured data as the inlet boundary condition of the numerical simulation and conducted it. As the numerical result, we confirmed the ions affected by the electric field moved upstream with pulsation. We also confirmed the effect of the electric field on the motion of the isotope.
Sakuma, Hiroshi*; Tachi, Yukio; Yotsuji, Kenji; Suehara, Shigeru*; Arima, Tatsumi*; Fujii, Naoki*; Kawamura, Katsuyuki*; Honda, Akira
Clays and Clay Minerals, 65(4), p.252 - 272, 2017/08
Times Cited Count:2 Percentile:16.47(Chemistry, Physical)Structure and stability of montmorillonite edge faces (110), (010), (100), and (130) of the layer charges y = 0.5 and 0.33 are investigated by the first-principles electronic calculations based on the density functional theory. Stacking and single layer models are tested for understanding the effect of stacking on the stability of montmorillonite edge faces. Most stacking layers stabilize the edge faces by making hydrogen bonds between the layers; therefore, the surface energy of stacking layers is reduced rather than the single layer model. This indicates that the surface energy of edge faces should be estimated depending on the swelling conditions. Lowest surface energies of (010) and (130) edge faces were realized by the presence of Mg ions on the edge faces. These edge faces have a strong adsorption site for cations due to local negative charge of the edges.
Kimura, Hideo; Aoyagi, Tetsuo; Sato, Taiichi; Sakai, Manabu; Hikasa, Naoki*; Suzuki, Hitoshi; Tsuji, Minoru
JAEA-Technology 2011-027, 31 Pages, 2011/09
The financial accounting and contract management system of JAEA, built on a commercial ERP package, had suffered from serious problems of high lifecycle cost, poor response, and lack of extensibility, due to the too much customization to the ERP package, and disorganized software structure. To solve those problems, three approaches were applied; (1) conducting thorough analysis of business flow and fit/gap, which enabled removing all customization brought to the ERP package; (2) dividing the system into subsystems, and clearly defining interfaces between the subsystems, which increases the transparency, extensibility and performance of the system; (3) outsourcing the development of the subsystems to multiple venders to reduce the development cost. Those approaches can be useful for developing business information systems using commercial software to save time and cost, while meeting the unique requirements of an organization.
Sugikawa, Susumu; ; ; Nakazaki, Masato; Shirahashi, Koichi; ; *; *; Tsuji, Kenichi*; Tachimori, Shoichi; et al.
JAERI-Tech 97-007, 86 Pages, 1997/03
no abstracts in English
 In
In O
O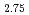 as electrolyte for solid oxide fuel cell analyzed by EXAFS and neutron powder diffraction
as electrolyte for solid oxide fuel cell analyzed by EXAFS and neutron powder diffractionIgawa, Naoki; Kodama, Katsuaki; Taguchi, Tomitsugu*; Tsuji, Takuya; Matsumura, Daiju; Yoshii, Kenji
no journal, ,
The local structure of BaSn In
In O
O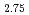 which is one of the excellent solid electrolytes for solid oxide fuel cells was analyzed by using the X-ray absorption spectroscopy followed the neutron diffraction. The average crystal structure analyzed by the neutron diffraction shows that BaSn
which is one of the excellent solid electrolytes for solid oxide fuel cells was analyzed by using the X-ray absorption spectroscopy followed the neutron diffraction. The average crystal structure analyzed by the neutron diffraction shows that BaSn In
In O
O has the space group,
has the space group, 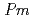 -3
-3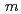 and the Sn and In randomly occupy at same site. However, the result of Extended X-ray Absorption Fine Structure shows that the distance of In-O and Sn-O is different. In addition, the In-O distance is increased by the hydrogen doping while the Sn-O distance does not change. Those results suggest that the Sn and In occupy different sites and therefore the local structural distortion has occurred. In this work, the details of the local structure of BaSn
and the Sn and In randomly occupy at same site. However, the result of Extended X-ray Absorption Fine Structure shows that the distance of In-O and Sn-O is different. In addition, the In-O distance is increased by the hydrogen doping while the Sn-O distance does not change. Those results suggest that the Sn and In occupy different sites and therefore the local structural distortion has occurred. In this work, the details of the local structure of BaSn In
In O
O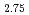 will be discussed.
will be discussed.
Horiguchi, Naoki; Yoshida, Hiroyuki; Kitatsuji, Yoshihiro; Fukumori, Mai*; Takemura, Yuki*; Hasegawa, Makoto*; Kishimoto, Tadafumi*
no journal, ,
no abstracts in English

Yoshii, Kenji; Tsuji, Takuya; Matsumura, Daiju; Nishida, Ginichi*; Inoue, Naoki*; Fukada, Yukimasa*; Yaita, Tsuyoshi
no journal, ,
We have investigated the magnetic properties of Bi-doped perovskite GdCrO , which was reported to be multiferroic below the magnetic transition temperature (170 K). For the low doping levels (about 5-10% Bi at the Gd site), single-phase samples could be prepared at 920
, which was reported to be multiferroic below the magnetic transition temperature (170 K). For the low doping levels (about 5-10% Bi at the Gd site), single-phase samples could be prepared at 920 C, a temperature which is much lower than the firing temperature of GdCrO
C, a temperature which is much lower than the firing temperature of GdCrO (about 1300-1400
(about 1300-1400 C). This result is essentially the same as that of isostructural (Sm,Bi)CrO
C). This result is essentially the same as that of isostructural (Sm,Bi)CrO . Magnetic measurements showed that the magnetic properties were essentially identical to those of GdCrO
. Magnetic measurements showed that the magnetic properties were essentially identical to those of GdCrO . For example, the chromium spins show a ferrimagnetic transition at 170 K. Interestingly, these properties were entirely different from those of (Sm,Bi)CrO
. For example, the chromium spins show a ferrimagnetic transition at 170 K. Interestingly, these properties were entirely different from those of (Sm,Bi)CrO , which showed that the Bi doping led to a drastic lowering of magnetic transition temperature (by 140 K). We have also conducted X-ray absorption measurements to determine the crystallographic site of Bi. The results will be shown at the conference.
, which showed that the Bi doping led to a drastic lowering of magnetic transition temperature (by 140 K). We have also conducted X-ray absorption measurements to determine the crystallographic site of Bi. The results will be shown at the conference.
Matsui, Naoki*; Okubo, Takahiro*; Tachi, Yukio; Sugiura, Yuki; Yotsuji, Kenji*
no journal, ,
no abstracts in English
Matsui, Naoki*; Okubo, Takahiro*; Tachi, Yukio; Yotsuji, Kenji*; Sugiura, Yuki
no journal, ,
no abstracts in English
Horiguchi, Naoki; Yoshida, Hiroyuki; Kitatsuji, Yoshihiro; Fukumori, Mai*; Hasegawa, Makoto*; Kishimoto, Tadafumi*
no journal, ,
A Li-7 enriched pH adjuster is essential for water quality control on PWRs. As the required Li-7 enrichment technology, we have developed the multi-channel counter-current electrophoresis (MCCCE) method. In this research, to understand the behavior of ions in the enrichment experiment, we developed a numerical simulation method of the long-time behavior of the ions based on the 3-dimensional field data of the electricity by commercial software and the flow by TPFIT-LPT. In this work, we applied it to the behavior of the ions in a simplified plate-type flow channel from the experimental one. Since the separation coefficient calculated from the simulation results of Li-7 and Li-6 ions agreed well with the experimental value, we confirmed the applicability of the method.