Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tsuji, Takuya; Matsumura, Daiju; Kobayashi, Toru
SPring-8/SACLA Riyo Kenkyu Seikashu (Internet), 12(5), p.259 - 262, 2024/10
no abstracts in English
Krajewska, A.*; Tsuji, Takuya; Matsumura, Daiju; 15 of others*
Science Advances (Internet), 10(41), p.eadn3880_1 - eadn3880_10, 2024/10
Times Cited Count:1 Percentile:0.00(Multidisciplinary Sciences) , AgNbO
, AgNbO , and KNbO
, and KNbO
Yoneda, Yasuhiro; Kobayashi, Toru; Tsuji, Takuya; Matsumura, Daiju; Saito, Yuji; Noguchi, Yuji*
Japanese Journal of Applied Physics, 63(9), p.09SP12_1 - 09SP12_10, 2024/09
Times Cited Count:1 Percentile:0.00(Physics, Applied)NaNbO , AgNbO
, AgNbO , and KNbO
, and KNbO with ABO
with ABO -type perovskite system is known to possess good ferroelectric properties. We directly determined the rattling space of each atom through local structure analysis. This analysis revealed the bonding sites with large fluctuations varied with varying ion size. Experimental evidence including soft X-ray absorption spectroscopy, indicates that the A-site ions are hybridized with oxygen.
-type perovskite system is known to possess good ferroelectric properties. We directly determined the rattling space of each atom through local structure analysis. This analysis revealed the bonding sites with large fluctuations varied with varying ion size. Experimental evidence including soft X-ray absorption spectroscopy, indicates that the A-site ions are hybridized with oxygen.

Yamamoto, Hajime*; Ikeda, Osamu*; Honda, Takashi*; Kimura, Kenta*; Aoyama, Takuya*; Ogushi, Kenya*; Suzuki, Akio*; Ishii, Kenji*; Matsumura, Daiju; Tsuji, Takuya; et al.
Physical Review Materials (Internet), 8(9), p.094402_1 - 094402_6, 2024/09
Times Cited Count:2 Percentile:56.91(Materials Science, Multidisciplinary) Na
Na TiO
TiO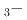 BaTiO
BaTiO solid solutions
solid solutionsYoneda, Yasuhiro; Kobayashi, Toru; Tsuji, Takuya; Matsumura, Daiju; Saito, Yuji; Noguchi, Yuji*
Japanese Journal of Applied Physics, 62(SM), p.SM1006_1 - SM1006_8, 2023/11
Times Cited Count:4 Percentile:50.15(Physics, Applied)Bi Na
Na TiO
TiO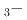 BaTiO
BaTiO (BNT
(BNT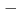 BT) solid solutions have been extensively studied because they exhibit good piezoelectric properties. In addition, a wide variety of phases are observed depending on the BT composition. Soft X-ray absorption spectroscopy and high-energy X-ray diffraction experiments of BNT
BT) solid solutions have been extensively studied because they exhibit good piezoelectric properties. In addition, a wide variety of phases are observed depending on the BT composition. Soft X-ray absorption spectroscopy and high-energy X-ray diffraction experiments of BNT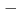 BT solid solutions were performed using synchrotron radiation. From the electronic structure and local structure of BNT
BT solid solutions were performed using synchrotron radiation. From the electronic structure and local structure of BNT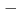 BT solid solution, the substitution effect of BT occurred mainly at the A site, which is the substitution site of Ba. The rhombohedral strain of the TiO
BT solid solution, the substitution effect of BT occurred mainly at the A site, which is the substitution site of Ba. The rhombohedral strain of the TiO octahedron did not change with the change in BT composition, suggesting that the change in the electronic structure at the O-
octahedron did not change with the change in BT composition, suggesting that the change in the electronic structure at the O- absorption edge is due to the change in the hybridization state.
absorption edge is due to the change in the hybridization state.
Matsumura, Daiju; Kimura, Yusaku*; Tsuji, Takuya; Mizuki, Junichiro*
SPring-8/SACLA Riyo Kenkyu Seikashu (Internet), 11(5), p.296 - 299, 2023/11
no abstracts in English
Ikeda, Yoichi*; Umemoto, Yoshihiko*; Matsumura, Daiju; Tsuji, Takuya; Hashimoto, Yuki*; Kitazawa, Takafumi*; Fujita, Masaki*
Materials Transactions, 64(9), p.2254 - 2260, 2023/09
Times Cited Count:4 Percentile:46.68(Materials Science, Multidisciplinary)Yoneda, Yasuhiro; Tsuji, Takuya; Matsumura, Daiju; Okamoto, Yoshihiro; Takaki, Seiya; Takano, Masahide
Physica B; Condensed Matter, 663, p.414960_1 - 414960_9, 2023/08
Times Cited Count:0 Percentile:0.00(Physics, Condensed Matter)We performed various synchrotron X-ray measurements to extract local and average structures of DyN-ZrN solid solutions. We performed the nanoscale structural analysis by combining X-ray absorption fine structure and high-energy X-ray diffraction. The DyN-ZrN solid solution has a rock-salt type cubic crystal structure, and there are instabilities such as the chemical order of the metal site and the distribution of the bond length of the nitrogen site.
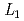 -edge XAFS measurement
-edge XAFS measurementTsuji, Takuya; Matsumura, Daiju; Kobayashi, Toru
SPring-8/SACLA Riyo Kenkyu Seikashu (Internet), 11(4), p.214 - 217, 2023/08
no abstracts in English
 -edge X-ray absorption fine structure study of
-edge X-ray absorption fine structure study of 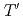 -type
-type 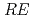
 CuO
CuO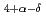 (
(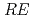 = Rare Earth); Toward unified understanding of electronic state of
= Rare Earth); Toward unified understanding of electronic state of 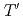 -type cuprate
-type cuprateChen, Y.*; Asano, Shun*; Wang, T.*; Xie, P.*; Kitayama, Shinnosuke*; Ishii, Kenji*; Matsumura, Daiju; Tsuji, Takuya; Taniguchi, Takanori*; Fujita, Masaki*
JPS Conference Proceedings (Internet), 38, p.011050_1 - 011050_6, 2023/05
Ouchi, Kazuki; Matsumura, Daiju; Tsuji, Takuya; Kobayashi, Toru; Otobe, Haruyoshi; Kitatsuji, Yoshihiro
RSC Advances (Internet), 13(24), p.16321 - 16326, 2023/05
Times Cited Count:1 Percentile:11.04(Chemistry, Multidisciplinary)We clarified the chemical state transformation of deposits following the reduction of uranyl ion (U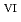 O
O
 ) from the results of electrochemical quartz crystal microbalance, impedance spectra and X-ray absorption fine structure measurements. We propose the following deposition mechanism: (1) U
) from the results of electrochemical quartz crystal microbalance, impedance spectra and X-ray absorption fine structure measurements. We propose the following deposition mechanism: (1) U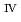 is formed by the disproportionation of U
is formed by the disproportionation of U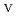 . (2) U
. (2) U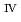 forms U
forms U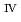 hydroxide deposits, and (3) finally, the hydroxide deposits transform into U
hydroxide deposits, and (3) finally, the hydroxide deposits transform into U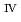 oxide, generally having a larger electrical resistance than the former.
oxide, generally having a larger electrical resistance than the former.
Ikeda, Yusuke*; Matsumura, Daiju; Tsuji, Takuya; Namai, Asuka*; Imoto, Kenta*; Tokoro, Hiroko*; Nakabayashi, Koji*; Okoshi, Shinichi*
Inorganica Chimica Acta, 550, p.121434_1 - 121434_8, 2023/03
Times Cited Count:0 Percentile:0.00(Chemistry, Inorganic & Nuclear)Tsuji, Takuya; Matsumura, Daiju; Kobayashi, Toru; Yaita, Tsuyoshi
SPring-8/SACLA Riyo Kenkyu Seikashu (Internet), 11(1), p.15 - 18, 2023/02
no abstracts in English
Liu, B.*; Feng, R.*; Busch, M.*; Wang, S.*; Wu, H.*; Liu, P.*; Gu, J.*; Bahadoran, A.*; Matsumura, Daiju; Tsuji, Takuya; et al.
ACS Nano, 16(9), p.14121 - 14133, 2022/09
Times Cited Count:97 Percentile:98.70(Chemistry, Multidisciplinary)Uehara, Akihiro*; Matsumura, Daiju; Tsuji, Takuya; Yakumaru, Haruko*; Tanaka, Izumi*; Shiro, Ayumi*; Saito, Hiroyuki*; Ishihara, Hiroshi*; Homma-Takeda, Shino*
Analytical Methods, 14(24), p.2439 - 2445, 2022/06
Times Cited Count:7 Percentile:66.89(Chemistry, Analytical)Uehara, Akihiro*; Shuhui, X.*; Sato, Ryotaro*; Matsumura, Daiju; Tsuji, Takuya; Yakumaru, Haruko*; Shiro, Ayumi*; Saito, Hiroyuki*; Tanaka, Izumi*; Ishihara, Hiroshi*; et al.
Advances in X-Ray Chemical Analysis, Japan, 53, p.223 - 229, 2022/03
no abstracts in English
 ion complex; Presence of a Rh
ion complex; Presence of a Rh Intermediate in Direct Photoreduction
Intermediate in Direct PhotoreductionSaeki, Morihisa*; Matsumura, Daiju; Nakanishi, Ryuzo*; Yomogida, Takumi; Tsuji, Takuya; Saito, Hiroyuki*; Oba, Hironori*
Journal of Physical Chemistry C, 126(12), p.5607 - 5616, 2022/03
Times Cited Count:2 Percentile:12.90(Chemistry, Physical)The reaction mechanism of the direct photoreduction of a Rh ion complex to a Rh
ion complex to a Rh species induced by pulsed ultraviolet laser irradiation was studied using dispersive X-ray absorption fine structure (DXAFS) spectroscopy. The time-resolved X-ray absorption near edge structure (XANES) showed the absence of isosbestic points and suggested that more than two Rh
species induced by pulsed ultraviolet laser irradiation was studied using dispersive X-ray absorption fine structure (DXAFS) spectroscopy. The time-resolved X-ray absorption near edge structure (XANES) showed the absence of isosbestic points and suggested that more than two Rh species contribute toward the direct photoreduction of Rh
species contribute toward the direct photoreduction of Rh . Analysis of the time-resolved XANES data by singular value deposition showed that the direct photoreduction involves three Rh
. Analysis of the time-resolved XANES data by singular value deposition showed that the direct photoreduction involves three Rh species. Multivariate curve resolution by alternating least-squares analysis (MCR-ALS) of the time-resolved XANES data gave pure spectra and concentration profiles of the three Rh
species. Multivariate curve resolution by alternating least-squares analysis (MCR-ALS) of the time-resolved XANES data gave pure spectra and concentration profiles of the three Rh species. The Rh
species. The Rh species were assigned to Rh
species were assigned to Rh , Rh
, Rh , and Rh
, and Rh species based on the features of the pure XANES spectra. The concentration profiles suggested that the direct photoreduction proceeds in the order of Rh
species based on the features of the pure XANES spectra. The concentration profiles suggested that the direct photoreduction proceeds in the order of Rh
 Rh
Rh
 Rh
Rh . A reaction mechanism, which was proposed involving photoreductions of Rh
. A reaction mechanism, which was proposed involving photoreductions of Rh and Rh
and Rh , photoinduced autocatalytic reductions of Rh
, photoinduced autocatalytic reductions of Rh and Rh
and Rh , and photooxidation of Rh
, and photooxidation of Rh , well reproduced the concentration profiles of three Rh
, well reproduced the concentration profiles of three Rh species.
species.
 O
O with electrochemical Li
with electrochemical Li intercalation
intercalationMurase, Satoshi*; Yoshikawa, Yumi*; Fujiwara, Kosuke*; Fukada, Yukimasa*; Teranishi, Takashi*; Kano, Jun*; Fujii, Tatsuo*; Inada, Yasuhiro*; Katayama, Misaki*; Yoshii, Kenji; et al.
Journal of Physics and Chemistry of Solids, 162, p.110468_1 - 110468_6, 2022/03
Times Cited Count:0 Percentile:0.00(Chemistry, Multidisciplinary)We report a trial of the valence control for mixed valence iron triangular oxide YbFe O
O in order to develop an effective technique to control the frustration of charges in strongly correlated electron systems. The electro-chemical doping of Li
in order to develop an effective technique to control the frustration of charges in strongly correlated electron systems. The electro-chemical doping of Li into YbFe
into YbFe O
O was examined on the cell type sample similar to the Li-ion secondary battery cell. Systematic change of the lattice constant, Fe-Fe and Fe-Yb distance were observed with Li doping. Maximum value of the doping was over 300 mAh/g. An EXAFS experiment indicated that Li positioned between Yb octahedron layer (U-layer) and Fe-bipyramidal layer (W-layer). However, detailed change of iron valence state of YbFe
was examined on the cell type sample similar to the Li-ion secondary battery cell. Systematic change of the lattice constant, Fe-Fe and Fe-Yb distance were observed with Li doping. Maximum value of the doping was over 300 mAh/g. An EXAFS experiment indicated that Li positioned between Yb octahedron layer (U-layer) and Fe-bipyramidal layer (W-layer). However, detailed change of iron valence state of YbFe O
O was not clearly observed because of the superimpose of the signal from iron metal nano particles in XANES observation. The results indicate that the electrochemical method might be one of the potential technique to control the frustration of charges in YbFe
was not clearly observed because of the superimpose of the signal from iron metal nano particles in XANES observation. The results indicate that the electrochemical method might be one of the potential technique to control the frustration of charges in YbFe O
O .
.
Tanida, Hajime; Tsuji, Takuya; Kobata, Masaaki
JAEA-Technology 2021-031, 25 Pages, 2022/02
In the decommissioning of the Tokyo Electric Power Company Holdings Fukushima Daiichi Nuclear Power Station, analysis of fuel debris to understand its characteristics is very important. The fuel debris removed for testing and analyzing will be fine particulates. Non-destructive analytical methods using X-rays are effective for such samples, but in order to apply them to fine particles, the X-rays must be focused to the micrometer order. For this purpose, the Kirkpatrick-Baez (KB) mirror was introduced. In this paper, we record the selection, specification, adjustment of the mirror, and write down the example of mapping of elements and evaluation of their valence by this mirror.
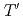 -type La
-type La Eu
Eu CuO
CuO and Nd
and Nd CuO
CuO
Asano, Shun*; Ishii, Kenji*; Matsumura, Daiju; Tsuji, Takuya; Kudo, Kota*; Taniguchi, Takanori*; Saito, Shin*; Sunohara, Toshiki*; Kawamata, Takayuki*; Koike, Yoji*; et al.
Physical Review B, 104(21), p.214504_1 - 214504_7, 2021/12
Times Cited Count:2 Percentile:10.80(Materials Science, Multidisciplinary)