Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Otani, Kyohei; Tsukada, Takashi; Ueno, Fumiyoshi
Materials Transactions, 62(6), p.763 - 769, 2021/06
Times Cited Count:0 Percentile:0(Materials Science, Multidisciplinary)In this study, the iron rust layer formed on the low alloy steel under air-solution alternating conditions was investigated by cross-sectional observation and analysis, and the mechanism of the accelerated corrosion of the steel under alternating conditions was clarified. The observations and analysis showed that the multilayered iron rust layer composed of the red rust layer (FeOOH), rust crust layer (Fe O
O ), inner crystal (Fe
), inner crystal (Fe O
O ), and inner rust layer was formed on the low alloy steel. It can be considered that the multilayered iron rust layer accelerated the cathodic reaction rate of dissolved oxygen under alternating conditions. This acceleration is the reason why the corrosion rate of the low alloy steel under alternating conditions was accelerated.
), and inner rust layer was formed on the low alloy steel. It can be considered that the multilayered iron rust layer accelerated the cathodic reaction rate of dissolved oxygen under alternating conditions. This acceleration is the reason why the corrosion rate of the low alloy steel under alternating conditions was accelerated.
Otani, Kyohei; Tsukada, Takashi; Ueno, Fumiyoshi; Kato, Chiaki
Zairyo To Kankyo, 69(9), p.246 - 252, 2020/09
The purpose of this study was to investigate the effect of artificial sea water concentration on the corrosion rate of carbon steel under air/solution alternating condition, and to clarify the corrosion mechanism of carbon steel that changes with artificial seawater concentration. Mass measurements showed that the corrosion rate of carbon steel in the alternating condition accelerates with increasing concentration in the concentration region between deionized water to 200 times diluted artificial seawater (ASW), and the corrosion rate decreases with increasing concentration in the concentration region between 20 times diluted ASW to undiluted ASW. It can be considered that the reason why the carbon steel corrosion was suppressed in highly concentrated artificial seawater would Mg ions and Ca ions in the artificial seawater precipitate and cover on the surface due to the increase in pH near the surface by oxygen reduction reaction.
Otani, Kyohei; Tsukada, Takashi; Ueno, Fumiyoshi
Zairyo To Kankyo, 68(8), p.205 - 211, 2019/08
In the present study, the iron rust layer formed on the low ally steel in air-solution alternating condition was investigated by cross-sectional observation and analysis, and the mechanism of accelerated corrosion of the steel in the alternating condition was clarified. Observation and analysis showed that the multi-layered iron rust layer composed of red rust layer (FeOOH), rust crust layer (Fe O
O ), inner crystal (Fe
), inner crystal (Fe O
O ), and inner rust layer was formed on the low alloy steel. It can be considered that the multi-layered iron rust layer accelerated the cathodic reaction rate of the steel in the alternating condition. This acceleration would be the reason why the corrosion rate of the low alloy steel in the alternating condition was accelerated.
), and inner rust layer was formed on the low alloy steel. It can be considered that the multi-layered iron rust layer accelerated the cathodic reaction rate of the steel in the alternating condition. This acceleration would be the reason why the corrosion rate of the low alloy steel in the alternating condition was accelerated.
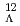 C level structure by
C level structure by  -ray spectroscopy
-ray spectroscopyHosomi, Kenji; Ma, Y.*; Ajimura, Shuhei*; Aoki, Kanae*; Dairaku, Seishi*; Fu, Y.*; Fujioka, Hiroyuki*; Futatsukawa, Kenta*; Imoto, Wataru*; Kakiguchi, Yutaka*; et al.
Progress of Theoretical and Experimental Physics (Internet), 2015(8), p.081D01_1 - 081D01_8, 2015/08
Times Cited Count:14 Percentile:66.59(Physics, Multidisciplinary)Level structure of the 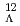 C hypernucleus was precisely determined by means of
C hypernucleus was precisely determined by means of  -ray spectroscopy. We identified four
-ray spectroscopy. We identified four  -ray transitions via the
-ray transitions via the  C
C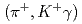 reaction using a germanium detector array, Hyperball2. The spacing of the ground-state doublet
reaction using a germanium detector array, Hyperball2. The spacing of the ground-state doublet 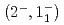 was measured to be
was measured to be 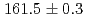 (stat)
(stat)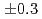 (syst)keV from the direct
(syst)keV from the direct 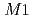 transition. Excitation energies of the
transition. Excitation energies of the 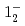 and
and 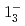 states were measured to be
states were measured to be 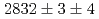 , keV and
, keV and 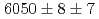 , keV, respectively. The obtained level energies provide definitive references for the reaction spectroscopy of
, keV, respectively. The obtained level energies provide definitive references for the reaction spectroscopy of 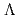 hypernuclei.
hypernuclei.
Otani, Kyohei; Tsukada, Takashi; Ueno, Fumiyoshi
no journal, ,
Previous study reported that corrosion rate of steel under gas/aqueous alternating condition is three times faster than under solution condition. In this study, the mechanism of corrosion acceleration of steel under gas/aqueous alternating condition was clarified by cross-sectional observation and analysis. The cross-sectional image of the rust layer shows that the layer was composed of multi-layer structure and the chemical analysis of Raman spectroscopy clarified that outer layer is FeOOH and inner layer is Fe O
O . These results suggest that FeOOH is formed on the rust initially, and then FeOOH changes to Fe
. These results suggest that FeOOH is formed on the rust initially, and then FeOOH changes to Fe O
O by cathodic reduction reaction. On the other hand, Cl ions concentrated at the rust/metal interface, therefore, anodic Fe dissolution would be accelerated by the concentration of Cl ions. From these reasons, the corrosion of steel under gas/aqueous alternating condition would be accelerated by the cathodic reduction reaction and the anodic Fe dissolution.
by cathodic reduction reaction. On the other hand, Cl ions concentrated at the rust/metal interface, therefore, anodic Fe dissolution would be accelerated by the concentration of Cl ions. From these reasons, the corrosion of steel under gas/aqueous alternating condition would be accelerated by the cathodic reduction reaction and the anodic Fe dissolution.
Otani, Kyohei; Tsukada, Takashi; Ueno, Fumiyoshi; Yamamoto, Masahiro
no journal, ,
It has been reported that the corrosion rate of carbon steel in air-solution alternating environment, however, the mechanism of the accelerated corrosion of carbon steel in the alternating environment has not been clarified. In the present study, the mechanism of accelerated corrosion of carbon steel in the alternating condition is investigated by the cross-sectional observation and analysis for rust layer formed on the steel after the corrosion tests. The observation and analysis showed that the multi-layered rust layer was formed on the steel in the alternating environment, and the multi-layer rust layer accelerated the cathodic reaction. This acceleration of the cathodic reaction is the reason why the corrosion rate of carbon steel was accelerated in air-solution alternating environment.
Tsukada, Takashi; Otani, Kyohei; Terakado, Hiroshi*; Ebata, Koei*; Ueno, Fumiyoshi
no journal, ,
As reported previously, corrosion behavior of carbon steel in the artificial diluted seawater has been examined by the rotating-type corrosion test method and the corrosion rate was accelerated from 3-5 times by the air-solution alternating condition. In this study, an effect of the concentration of diluted seawater at 30 C will be presented.
C will be presented.
Otani, Kyohei; Tsukada, Takashi; Terakado, Hiroshi*; Ebata, Koei*; Ueno, Fumiyoshi
no journal, ,
Inner components of the primary containment vessels (PCVs) in Fukushima Daiichi Nuclear Power Station was observed by remote-controlled robots and the observation showed that carbon steels of the PCVs wall above the contaminated water level was exposed to an air-solution alternating condition. Previous studies have reported that the corrosion rate of carbon steel is accelerated in case of the steel with thin water film exposed in air under the dry/wet condition. This suggests that the corrosion rate of carbon steel will be accelerated in the air-solution alternating condition. However, the corrosion rate of the steel on the corrosion tests which simulated the air-solution alternating condition has not been clarified. In the present study, a novel corrosion test of carbon steel which simulated the air-solution alternating condition was carried out. In order to simulate the alternating condition in the novel corrosion test, the carbon steel was alternately exposed to air and solution by rotating in a water tank which was not completely filled with solution. The surface observations showed that the rust layer formed on the steel after the tests in the alternating condition was thicker than that of the steel rotated always in solution. The mass loss measurements showed that the corrosion rate of carbon steel in the alternating condition was more than three times larger than that of the steel rotated always in solution. A thin water film was confirmed on the steel when the specimen exposed to the air during the tests. It has been reported that the mass transfer of dissolved oxygen to the carbon steel surface is accelerated in case of the carbon steel covered by a thin water film and corrosion of carbon steel is accelerated by the acceleration of oxygen reduction reaction (cathodic reaction). This suggests that the accelerated corrosion of the steel in the alternating condition would be caused by the thin water film on the steel during the tests.
Otani, Kyohei; Tsukada, Takashi; Ueno, Fumiyoshi; Kato, Chiaki
no journal, ,
Corrosion tests of carbon steel was carried out in air-solution alternating condition in previous study. It has been reported that the corrosion rate of carbon steel exposed in air-solution alternating condition was accelerated compare with the steel exposed in solution. In this study, it was investigated that the effect of concentration of model sea water on corrosion rate of carbon steel in air-solution alternating condition. The mass measurement in corrosion tests clarified that the corrosion rate of carbon steel in the 200 times diluted model sea water is fastest during various model sea water. Cross-sectional analysis of iron rust layer formed on carbon steel after corrosion tests indicated that the corrosion rate of carbon steel in model sea water is decreased by precipitation of metal cations containing in model sea water.
Otani, Kyohei; Tsukada, Takashi; Ueno, Fumiyoshi; Kato, Chiaki
no journal, ,
It was reported that the corrosion rate of carbon steel was accelerated under the air/solution alternating condition. In this presentation, it is investigated that the effect of oxygen concentration on the corrosion rate of steel under the air/solution alternating condition. As a result, the corrosion rate of carbon steel under the air/solution alternating condition did not increase linearly with increasing oxygen concentration in the air, and the slope in the low concentration range (0-5%) is greater than in the high concentration range (5-20.8%). This suggests that the corrosion rate may be accelerated around the air/solution interface on the inner surface of the PCV if even a small amount of oxygen is introduced into the PCV during the debris removal process.