Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Aoki, Ryota; Sato, Tetsuya; Uchibaba, Yuta; Miyachi, Yuta*; Gong, G.; Natori, Hina*; Ito, Yuta; Asai, Masato; Tsukada, Kazuaki; Nagame, Yuichiro*
no journal, ,
In order to apply the ion beams of superheavy elements with atomic number over 100 to physics and chemistry research, we constructed an EBGP (Electron Beam Generated Plasma) ion source as an ion source applicable to short-lived single atoms. The EBGP ion source was installed in the JAEA-ISOL (Isotope Separator On-Line), and ion beam generation in various ionization conditions has been conducted with several noble gases. As a result, we confirmed the ion source worked in a stable condition, and an ion beam was successfully generated.
Uchibaba, Yuta; Ito, Yuta; Sato, Tetsuya; Aoki, Ryota; Asai, Masato; Gong, G.; Miyachi, Yuta*; Nagame, Yuichiro*; Natori, Hina*; Tsukada, Kazuaki
no journal, ,
In anticipation of precise mass measurement of superheavy nuclei with an atomic number of 104 or more, we are developing an ion trap for the purpose of improving the accuracy of mass determination by prior identification of ions to be measured by decay spectroscopy and correlation with decay information. In order to capture radiation from trapped unstable nuclear ions at a large solid angle, we have developed a vane electrode type ion trap using a thin plate-like vane electrode. As an indication of performance, the trap lifetime and accumulated charge were evaluated in comparison with a circular electrode type ion trap.
Sato, Tetsuya; Aoki, Ryota; Miyachi, Yuta*; Asai, Masato; Ito, Yuta; Uchibaba, Yuta; Gong, G.; Natori, Hina*; Tsukada, Kazuaki; Nagame, Yuichiro*
no journal, ,
We have been developing an EBGP (Electron Beam Generated Plasma) ion source1 coupled to the gas-jet transport system to produce SHE ion beams for rapid chemical experiments. The EBGP ion source ionizes atoms and molecules by electron impact and is expected to produce ion beams of various elements. We have succeeded to produce ion-beams of several noble gases using the present ion source, and it has been found that the beam intensities increase with increasing atomic number. We report on the ionization of various short-lived nuclides produced in spontaneous fission of 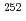 Cf.
Cf.
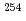 Es target
Es targetAsai, Masato; Tsukada, Kazuaki; Sato, Tetsuya; Ito, Yuta; Nishio, Katsuhisa; Makii, Hiroyuki; Hirose, Kentaro; Orlandi, R.; Suzaki, Fumi; Aoki, Ryota; et al.
no journal, ,
Fission of neutron-rich heavy-actinide nuclei exhibits extremely characteristic fission-fragment mass distributions which differ significantly from those of typical actinide nuclei. In the present study, we have succeeded in producing those neutron-rich heavy-actinide nuclei with multinucleon transfer reactions using an extremely rare and short-lived 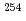 Es target bombarded with heavy-ion beam from the JAEA Tandem Accelerator. The products were mass-separated with an on-line isotope separator (ISOL), and spontaneous fission of
Es target bombarded with heavy-ion beam from the JAEA Tandem Accelerator. The products were mass-separated with an on-line isotope separator (ISOL), and spontaneous fission of  Fm,
Fm, 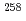 Fm,
Fm, 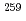 Md,
Md, 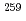 Lr was observed. Fission-fragment mass distributions and total kinetic energy distributions were obtained precisely, which provides us with important information to clarify the characteristic fission mechanism observed in this nuclear region.
Lr was observed. Fission-fragment mass distributions and total kinetic energy distributions were obtained precisely, which provides us with important information to clarify the characteristic fission mechanism observed in this nuclear region.
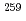 Md
MdAsai, Masato; Ishibashi, Yuichi*; Tsukada, Kazuaki; Sato, Tetsuya; Aoki, Ryota*; Ito, Yuta; Uchibaba, Yuta*; Orlandi, R.; Gong, G.-M.*; Suzaki, Fumi; et al.
no journal, ,
Measurement of mass and total kinetic energy (TKE) distribution for the spontaneous fission (SF) of 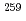 Md has been performed to clarify the fission mechanism in the neutron-rich fermium region.
Md has been performed to clarify the fission mechanism in the neutron-rich fermium region. 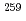 Md nuclei were produced with the
Md nuclei were produced with the 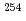 Es target and
Es target and  O beam, and mass-separated with an on-line isotope separator (ISOL). We discuss the mechanism of fission observed in the SF of
O beam, and mass-separated with an on-line isotope separator (ISOL). We discuss the mechanism of fission observed in the SF of 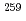 Md on the basis of the observed mass-TKE distribution.
Md on the basis of the observed mass-TKE distribution.
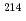 Ra
Ra ions for decay-recoil-correlated spectroscopy
ions for decay-recoil-correlated spectroscopyUchibaba, Yuta; Ito, Yuta; Sato, Tetsuya; Aoki, Ryota; Asai, Masato; Gong, G.; Tsukada, Kazuaki; Natori, Hina; Miyachi, Yuta; Nagame, Yuichiro*
no journal, ,
We have developed a Paul trap with an advanced electrode structure that allows for the detection of radiation and decay recoils over a large solid angle, aiming to perform identifications of radioactive ions by decay spectroscopy and precision mass spectroscopy combined with decay correlation. In order to measure the correlation between radiations and decay recoils from ions stored in the trap, we tested the performance of a semiconductor detector in the vicinity of radio-frequency electrodes and demonstrated in-trap decay measurements using 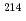 Ra
Ra ions.
ions.
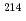 Ra
Ra ions for decay-recoil-correlated spectroscopy
ions for decay-recoil-correlated spectroscopyUchibaba, Yuta; Ito, Yuta; Sato, Tetsuya; Aoki, Ryota; Asai, Masato; Gong, G.; Tsukada, Kazuaki; Natori, Hina; Miyachi, Yuta; Nagame, Yuichiro*
no journal, ,
We have developed a Paul trap with an advanced electrode structure that allows for the detection of radiation and decay recoils over a large solid angle, aiming to perform identifications of radioactive ions by decay spectroscopy and precision mass spectroscopy combined with decay correlation. In order to measure the correlation between radiations and decay recoils from ions stored in the trap, we tested the performance of a semiconductor detector in the vicinity of radio-frequency electrodes and demonstrated in-trap decay measurements using 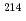 Ra
Ra ions.
ions.
Natori, Hina; Sato, Tetsuya; Asai, Masato; Ito, Yuta; Uchibaba, Yuta; Gong, G.; Tsukada, Kazuaki; Miyachi, Yuta; Nagame, Yuichiro*
no journal, ,
To elucidate the chemical properties of element 106, seaborgium (Sg), we have constructed an off-line isothermal gas chromatographic system. As a model experiment for Sg using the system, gas-phase chemical experiments were performed for volatile oxychlorides of short-lived molybdenum (Mo) isotopes, a homolog of Sg, produced in spontaneous fission of 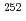 Cf. The optimum experimental conditions were obtained for isothermal gas chromatographic experiments using this apparatus. The observed isothermal gas chromatographic behaviors of short-lived Mo isotopes, we have successfully determined values of the adsorption enthalpies of Mo oxychlorides on the column surface. These values were in good agreement with those estimated from the sublimation enthalpies of the compounds.
Cf. The optimum experimental conditions were obtained for isothermal gas chromatographic experiments using this apparatus. The observed isothermal gas chromatographic behaviors of short-lived Mo isotopes, we have successfully determined values of the adsorption enthalpies of Mo oxychlorides on the column surface. These values were in good agreement with those estimated from the sublimation enthalpies of the compounds.