Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Umebayashi, Tsutomu; Yamaki, Tetsuya; Asai, Keisuke*
Hikari Shokubai; Kiso, Zairyo Kaihatsu, Oyo, p.637 - 643, 2005/05
no abstracts in English
 thin films on glass substrate grown by pulsed laser deposition
thin films on glass substrate grown by pulsed laser depositionChoi, Y.; Yamamoto, Shunya; Umebayashi, Tsutomu; Yoshikawa, Masahito
Solid State Ionics, 172(1-4), p.105 - 108, 2004/08
Times Cited Count:41 Percentile:82.22(Chemistry, Physical)no abstracts in English
 photocatalysts
photocatalystsChoi, Y.; Umebayashi, Tsutomu; Yoshikawa, Masahito
Journal of Materials Science, 39(5), p.1837 - 1839, 2004/03
Times Cited Count:227 Percentile:97.89(Materials Science, Multidisciplinary)Carbon (C)-doped anatase TiO photocatalysts have been prepared by the oxidative annealing of TiC powders at 623 K for 50-100 hrs. From XRD and XPS analysis, it is clear that C is incorporated into O site of the TiO
photocatalysts have been prepared by the oxidative annealing of TiC powders at 623 K for 50-100 hrs. From XRD and XPS analysis, it is clear that C is incorporated into O site of the TiO lattice. Diffuse reflectance spectroscopy (DRS) showed that the optical absorption edge of the C-doped anatase TiO
lattice. Diffuse reflectance spectroscopy (DRS) showed that the optical absorption edge of the C-doped anatase TiO shifted to lower energy compared with that of pure anatase TiO
shifted to lower energy compared with that of pure anatase TiO . The decomposition of methylene blue (MB) adsorbed on the C-doped sample was observed under a visible light range of 420-500 nm. We conclude that substitution of C for O in the TiO
. The decomposition of methylene blue (MB) adsorbed on the C-doped sample was observed under a visible light range of 420-500 nm. We conclude that substitution of C for O in the TiO leads to a photocatalytic decomposition of MB under visible light irradiation.
leads to a photocatalytic decomposition of MB under visible light irradiation.
 /ZnO films by pulsed laser deposition
/ZnO films by pulsed laser depositionYamamoto, Shunya; Choi, Y.; Umebayashi, Tsutomu; Yoshikawa, Masahito
Transactions of the Materials Research Society of Japan, 29(6), p.2701 - 2704, 2004/00
no abstracts in English
 photocatalysts by oxidative annealing of TiC
photocatalysts by oxidative annealing of TiCChoi, Y.; Umebayashi, Tsutomu; Yamamoto, Shunya; Tanaka, Shigeru
Journal of Materials Science Letters, 22(17), p.1209 - 1211, 2003/09
Times Cited Count:47 Percentile:78.45(Materials Science, Multidisciplinary)no abstracts in English
 under visibel light irradiation
under visibel light irradiationUmebayashi, Tsutomu; Asai, Keisuke*; Yamaki, Tetsuya; Yoshikawa, Masahito
Kogyo Zairyo, 51(7), p.34 - 36, 2003/07
no abstracts in English
 by ion implantation
by ion implantationUmebayashi, Tsutomu; Yamaki, Tetsuya; Yamamoto, Shunya; Tanaka, Shigeru; Asai, Keisuke*
Transactions of the Materials Research Society of Japan, 28(2), p.461 - 464, 2003/06
TiO is promising as a photocatalytic material. However, it is active only under UV light irradiation because of its wide band gap (
is promising as a photocatalytic material. However, it is active only under UV light irradiation because of its wide band gap ( 3.0 eV). We recently reported that sulfur (S) doping caused the optical absorption edge of TiO
3.0 eV). We recently reported that sulfur (S) doping caused the optical absorption edge of TiO to be shifted into the lower energy region. Based on the theoretical analyses using first principles band calculations, mixing of the S 3p states with the valence band was found to contribute to the bandgap narrowing. In this study presented here, S-doped TiO
to be shifted into the lower energy region. Based on the theoretical analyses using first principles band calculations, mixing of the S 3p states with the valence band was found to contribute to the bandgap narrowing. In this study presented here, S-doped TiO was prepared by ion implantation and subsequent thermal annealing. S
was prepared by ion implantation and subsequent thermal annealing. S was implanted into the single crystals of rutile TiO
was implanted into the single crystals of rutile TiO with a fluence of 8
with a fluence of 8  10
10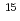 ions/cm
ions/cm . According to the results of RBS/channeling analysis, irradiation damage recovered after the annealing at 600
. According to the results of RBS/channeling analysis, irradiation damage recovered after the annealing at 600  C in air. In the annealed crystal, S atoms occupied O sites to form Ti-S bonds, as assessed by XPS.
C in air. In the annealed crystal, S atoms occupied O sites to form Ti-S bonds, as assessed by XPS.
Umebayashi, Tsutomu; Yamaki, Tetsuya; Yamamoto, Shunya; Miyashita, Atsumi; Tanaka, Shigeru; Sumita, Taishi*; Asai, Keisuke*
Journal of Applied Physics, 93(9), p.5156 - 5160, 2003/05
Times Cited Count:300 Percentile:98.63(Physics, Applied)Sulfur (S)-doped titanium dioxide (TiO ) was synthesized by ion implantation and subsequent thermal annealing. Compared to the pure TiO
) was synthesized by ion implantation and subsequent thermal annealing. Compared to the pure TiO , a photocurrent was observed in the lower energy regions for the S-doped TiO
, a photocurrent was observed in the lower energy regions for the S-doped TiO . Based on the theoretical analyses by the first principles band calculations using the full potential linearized augmented plane wave methods within the generalized gradient approximation, the mixing of the S 3p states with the valence band (VB) was found to contribute to the increasing width of the VB. This leads to the bandgap narrowing in the S-doped TiO
. Based on the theoretical analyses by the first principles band calculations using the full potential linearized augmented plane wave methods within the generalized gradient approximation, the mixing of the S 3p states with the valence band (VB) was found to contribute to the increasing width of the VB. This leads to the bandgap narrowing in the S-doped TiO . Therefore, the photon-to-carrier conversion was induced during irradiation by visible light above 420 nm (
. Therefore, the photon-to-carrier conversion was induced during irradiation by visible light above 420 nm ( 2.9 eV).
2.9 eV).
Yamaki, Tetsuya; Umebayashi, Tsutomu; Sumita, Taishi*; Yamamoto, Shunya; Maekawa, Masaki; Kawasuso, Atsuo; Ito, Hisayoshi
Nuclear Instruments and Methods in Physics Research B, 206(1-4), p.254 - 258, 2003/05
Times Cited Count:136 Percentile:98.89(Instruments & Instrumentation)Single crystalline titanium dioxide (TiO ) rutile were implanted with 200keV F
) rutile were implanted with 200keV F at a nominal fluence of 1
at a nominal fluence of 1 10
10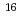 to 1
to 1 10
10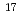 ions cm
ions cm and then thermally annealed in air up to 1200
and then thermally annealed in air up to 1200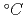 for 5h. The radiation damage and its recovery during the subsequent annealing were analyzed by Rutherford backscattering spectrometry in channeling geometry and variable-energy positron annihilation spectroscopy. The lattice disorder was completely annealed at 1200
for 5h. The radiation damage and its recovery during the subsequent annealing were analyzed by Rutherford backscattering spectrometry in channeling geometry and variable-energy positron annihilation spectroscopy. The lattice disorder was completely annealed at 1200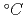 by the diffusion of point defects to the surface acting as a sink. According to the secondary ion mass spectrometry, the F depth profile was shifted to a shallower region along with the damage recovery, finally producing an F-doped layer where the impurity concentration increased steadily towards the surface. The F doping proved to provide a small modification to the conduction-band edge of TiO
by the diffusion of point defects to the surface acting as a sink. According to the secondary ion mass spectrometry, the F depth profile was shifted to a shallower region along with the damage recovery, finally producing an F-doped layer where the impurity concentration increased steadily towards the surface. The F doping proved to provide a small modification to the conduction-band edge of TiO , as assessed by theoretical band calculations.
, as assessed by theoretical band calculations.
Umebayashi, Tsutomu; Yamaki, Tetsuya; Sumita, Taishi*; Yamamoto, Shunya; Tanaka, Shigeru; Asai, Keisuke*
Nuclear Instruments and Methods in Physics Research B, 206, p.264 - 267, 2003/05
Times Cited Count:24 Percentile:81.71(Instruments & Instrumentation)Chromium (Cr) and niobium (Nb) were implanted into single-crystalline titanium dioxide (TiO ; rutile). After annealing at 600
; rutile). After annealing at 600 C for the Cr-implanted sample or at 1000
C for the Cr-implanted sample or at 1000 C for the Nb-implanted sample, the radiation damage was recovered. The implanted metals occupied titanium (Ti) sites in TiO
C for the Nb-implanted sample, the radiation damage was recovered. The implanted metals occupied titanium (Ti) sites in TiO to form metal-oxygen bonds. According to the ultraviolet-ray photoelectron spectra, a localized level due to the implanted metals was formed in band gap of both the crystals. This position was close to the VB edge for the Cr-doped TiO
to form metal-oxygen bonds. According to the ultraviolet-ray photoelectron spectra, a localized level due to the implanted metals was formed in band gap of both the crystals. This position was close to the VB edge for the Cr-doped TiO , while the Nb-doped TiO
, while the Nb-doped TiO had the small peak far from the edge. This is in good agreement with the ab-initio band calculation results. It is considered that the midgap states of Cr- and Nb-doped TiO
had the small peak far from the edge. This is in good agreement with the ab-initio band calculation results. It is considered that the midgap states of Cr- and Nb-doped TiO consist of the Cr t
consist of the Cr t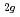 or Ti t
or Ti t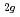 state, respectively.
state, respectively.

Umebayashi, Tsutomu; Yamaki, Tetsuya; Tanaka, Shigeru; Asai, Keisuke*
Chemistry Letters, 32(4), p.330 - 331, 2003/04
Times Cited Count:501 Percentile:99.16(Chemistry, Multidisciplinary)A sulfur (S)-doped titanium dioxide (TiO ) photocatalyst was synthesized by oxidative annealing of titanium disulfide (TiS
) photocatalyst was synthesized by oxidative annealing of titanium disulfide (TiS ). TiS
). TiS turned into anatase TiO
turned into anatase TiO when annealed in air. The residual S atoms occupied O-atom sites in the TiO
when annealed in air. The residual S atoms occupied O-atom sites in the TiO to form Ti-S bonds. The S doping caused the absorption edge of TiO
to form Ti-S bonds. The S doping caused the absorption edge of TiO to be shifted into the lower energy region. Consequently, methylene blue adsorbed on the S-doped TiO
to be shifted into the lower energy region. Consequently, methylene blue adsorbed on the S-doped TiO was photocatalytic decomposed by visible light irradiation.
was photocatalytic decomposed by visible light irradiation.
 based on band calculations
based on band calculationsUmebayashi, Tsutomu; Yamaki, Tetsuya; Ito, Hisayoshi; Asai, Keisuke*
Journal of Physics and Chemistry of Solids, 63(10), p.1909 - 1920, 2002/10
Times Cited Count:627 Percentile:99.67(Chemistry, Multidisciplinary)The electronic structures of titanium dioxide (TiO ) doped with 3d transition metals (V, Cr, Mn, Fe, Co and Ni) have been analyzed by ab-initio band calculations based on the density functional theory with the full-potential linearized-augmented-plane-wave methods. When TiO
) doped with 3d transition metals (V, Cr, Mn, Fe, Co and Ni) have been analyzed by ab-initio band calculations based on the density functional theory with the full-potential linearized-augmented-plane-wave methods. When TiO is doped with V, Cr, Mn, Fe or Co, an occupied level occurs and the electrons localized around each dopant. As the atomic number of the dopant increases, the localized level shifts to lower energy. In contrast, the electrons from Ni dopant are somewhat delocalized, thus significantly contributing to the formation of the valence band with O p and Ti 3d electrons. Based on a comparison with the absorption and photoconductivity data previously reported, we show that the t
is doped with V, Cr, Mn, Fe or Co, an occupied level occurs and the electrons localized around each dopant. As the atomic number of the dopant increases, the localized level shifts to lower energy. In contrast, the electrons from Ni dopant are somewhat delocalized, thus significantly contributing to the formation of the valence band with O p and Ti 3d electrons. Based on a comparison with the absorption and photoconductivity data previously reported, we show that the t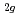 state of the dopant plays a significant role in the photoresponse of TiO
state of the dopant plays a significant role in the photoresponse of TiO under visible light irradiation.
under visible light irradiation.
Umebayashi, Tsutomu; Yamaki, Tetsuya; Ito, Hisayoshi; Asai, Keisuke*
Applied Physics Letters, 81(3), p.454 - 456, 2002/07
Times Cited Count:1351 Percentile:99.93(Physics, Applied)Titanium dioxide (TiO ) doped with sulfur (S) was synthesized by oxidation annealing of titanium disulfide (TiS
) doped with sulfur (S) was synthesized by oxidation annealing of titanium disulfide (TiS ). According to the X-ray diffraction patterns, TiS
). According to the X-ray diffraction patterns, TiS turned into anatase TiO
turned into anatase TiO when annealed at 600
when annealed at 600  C. The residual S atoms occupied O-atom sites in TiO
C. The residual S atoms occupied O-atom sites in TiO to form Ti-S bonds. The S doping caused the absorption edge of TiO
to form Ti-S bonds. The S doping caused the absorption edge of TiO to be shifted into the lower energy region. Based on the theoretical analyses using ab-initio band calculations, mixing of the S 3p states with the valence band was found to contribute to the band gap narrowing.
to be shifted into the lower energy region. Based on the theoretical analyses using ab-initio band calculations, mixing of the S 3p states with the valence band was found to contribute to the band gap narrowing.