Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ikeda, Nobuo*; Wakabayashi, Genichiro*; Uno, Masayoshi*; Yoshinaka, Kazuyuki
Nihon Genshiryoku Gakkai-Shi ATOMO , 65(5), p.304 - 305, 2023/05
, 65(5), p.304 - 305, 2023/05
no abstracts in English
Kumagai, Masayoshi*; Kuroda, Masatoshi*; Matsuno, Takashi*; Harjo, S.; Akita, Koichi*
Materials & Design, 221, p.110965_1 - 110965_8, 2022/09
Times Cited Count:4 Percentile:46.76(Materials Science, Multidisciplinary)Koie, Ryusuke*; Kawaguchi, Munemichi*; Miyahara, Shinya*; Uno, Masayoshi*; Seino, Hiroshi
Proceedings of 29th International Conference on Nuclear Engineering (ICONE 29) (Internet), 4 Pages, 2022/08
In order to investigate removal mechanisms of cesium aerosol from noble gas bubble in sodium pool, we performed a water simulation test to measure the decontamination factors of simulant aerosols with nitrogen gas bubbles rising through the water pool. As a result, it was found that the decontamination factors increased with the increase in the aerosol diameter and the water pool depth.
 and GeO
and GeO liquids
liquidsKawaguchi, Munemichi; Uno, Masayoshi*
Journal of Crystal Growth, 585, p.126590_1 - 126590_7, 2022/05
Phase-field mobility,  , and crystal growth rates in crystallization of 11 oxides or mixed oxides in undercooled silicates, SiO
, and crystal growth rates in crystallization of 11 oxides or mixed oxides in undercooled silicates, SiO and GeO
and GeO liquids were calculated with a simple phase-field model (PFM), and material dependence of the
liquids were calculated with a simple phase-field model (PFM), and material dependence of the  was discussed. Ratios between experimental crystal growth rates and the PFM simulation with
was discussed. Ratios between experimental crystal growth rates and the PFM simulation with 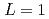 were confirmed to be proportional to a power of
were confirmed to be proportional to a power of 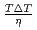 on the solid/liquid interface process during the crystal growth in a log-log plot. We determined that parameters,
on the solid/liquid interface process during the crystal growth in a log-log plot. We determined that parameters, 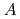 and
and  , of the
, of the 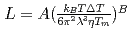 were
were 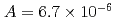 to
to 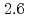 m
m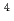 J
J s
s and
and 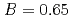 to
to 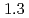 , which were unique for the materials. It was confirmed that our PFM simulation with the determined
, which were unique for the materials. It was confirmed that our PFM simulation with the determined  reproduced quantitively the experimental crystal growth rates. The
reproduced quantitively the experimental crystal growth rates. The 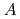 has a proportional relationship with the diffusion coefficient of a cation molar mass average per unit an oxygen molar mass at
has a proportional relationship with the diffusion coefficient of a cation molar mass average per unit an oxygen molar mass at 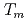 in a log-log graph. The
in a log-log graph. The  depends on the sum of the cation molar mass per the oxygen molar mass,
depends on the sum of the cation molar mass per the oxygen molar mass, 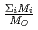 , in a compound. In
, in a compound. In 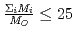 , the
, the  decreases with the cation molar mass increasing. The assumed cause is that the B represents the degree of the temperature dependence of the
decreases with the cation molar mass increasing. The assumed cause is that the B represents the degree of the temperature dependence of the  . Since the cation molar mass is proportional to an inertial resistance of the cation transfer, the
. Since the cation molar mass is proportional to an inertial resistance of the cation transfer, the  decreases with inverse of the cation molar mass. In crystallization of the silicates of heavy cation in
decreases with inverse of the cation molar mass. In crystallization of the silicates of heavy cation in 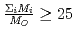 , the
, the  saturates at approximately 0.67, which leads to
saturates at approximately 0.67, which leads to 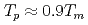 .
.
Doi, Daisuke; Seino, Hiroshi; Miyahara, Shinya*; Uno, Masayoshi*
Journal of Nuclear Science and Technology, 59(2), p.198 - 206, 2022/02
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology)Aoyagi, Mitsuhiro; Takata, Takashi; Uno, Masayoshi*
Nuclear Engineering and Design, 380, p.111258_1 - 111258_11, 2021/08
Times Cited Count:2 Percentile:29.53(Nuclear Science & Technology)Miyahara, Shinya*; Kawaguchi, Munemichi; Seino, Hiroshi; Atsumi, Takuto*; Uno, Masayoshi*
Proceedings of 28th International Conference on Nuclear Engineering (ICONE 28) (Internet), 6 Pages, 2021/08
In a postulated accident of fuel pin failure of sodium cooled fast reactor, a fission product cesium will be released from the failed pin as an aerosol such as cesium iodide and/or cesium oxide together with a fission product noble gas such as xenon and krypton. As the result, the xenon and krypton released with cesium aerosol into the sodium coolant as bubbles have an influence on the removal of cesium aerosol by the sodium pool in a period of bubble rising to the pool surface. In this study, cesium aerosol removal behavior due to inertial deposition, sedimentation and diffusion from a noble gas bubble rising through liquid sodium pool was analyzed by a computer program which deals with the expansion and the deformation of the bubble together with the aerosol absorption considering the effects of particle size distribution and agglomeration in aerosols. In the analysis, initial bubble diameter, sodium pool depth and temperature, aerosol particle diameter and density, initial aerosol concentration in the bubble were changed as parameter, and the results for the sensitivities of these parameters on decontamination factor (DF) of cesium aerosol were compared with the results of the previous study in which the effects of particle size distribution and agglomeration in aerosols were not considered. From the results, it was concluded that the sensitivities of initial bubble diameter, the aerosol particle diameter and density to the DF became significant due to the inertial deposition of agglomerated aerosols. To validate these analysis results, the simulation experiments have been conducted using a simulant particles of cesium aerosol under the condition of room temperature in water pool and air bubble systems. The experimental results were compared with the analysis results calculated under the same condition.
 O-2SiO
O-2SiO , Na
, Na O-2SiO
O-2SiO and K
and K O-2SiO
O-2SiO
Kawaguchi, Munemichi; Uno, Masayoshi*
Journal of the Ceramic Society of Japan, 128(10), p.832 - 838, 2020/10
Times Cited Count:2 Percentile:10.23(Materials Science, Ceramics)This study developed phase-field method (PFM) technique in oxide melt system by using a new mobility coefficient ( ). The crystal growth rates (
). The crystal growth rates (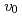 ) obtained by the PFM calculation with the constant
) obtained by the PFM calculation with the constant  were comparable to the thermodynamic driving force in normal growth model. The temperature dependence of the
were comparable to the thermodynamic driving force in normal growth model. The temperature dependence of the  was determined from the experimental crystal growth rates and the
was determined from the experimental crystal growth rates and the 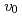 . Using the determined
. Using the determined  , the crystal growth rates (
, the crystal growth rates (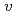 ) in alkali disilicate glasses, Li
) in alkali disilicate glasses, Li O-2SiO
O-2SiO , Na
, Na O-2SiO
O-2SiO and K
and K O-2SiO
O-2SiO were simulated. The temperature dependence of the
were simulated. The temperature dependence of the 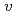 was qualitatively and quantitatively so similar that the PFM calculation results demonstrated the validity of the
was qualitatively and quantitatively so similar that the PFM calculation results demonstrated the validity of the  . Especially, the
. Especially, the 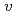 obtained by the PFM calculation appeared the rapid increase just below the thermodynamic melting point (
obtained by the PFM calculation appeared the rapid increase just below the thermodynamic melting point (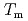 ) and the steep peak at around
) and the steep peak at around 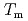 -100 K. Additionally, as the temperature decreased, the
-100 K. Additionally, as the temperature decreased, the 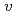 apparently approached zero ms
apparently approached zero ms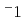 , which is limited by the
, which is limited by the  representing the interface jump process. Furthermore, we implemented the PFM calculation for the variation of the parameter
representing the interface jump process. Furthermore, we implemented the PFM calculation for the variation of the parameter  in the
in the  . As the
. As the  increased from zero to two, the peak of the
increased from zero to two, the peak of the 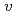 became steeper and the peak temperature of the
became steeper and the peak temperature of the 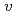 shifted to the high temperature side. The parameters
shifted to the high temperature side. The parameters 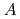 and
and  in the
in the  increased exponentially and decreased linearly as the atomic number of the alkali metal increased due to the ionic potential, respectively. This calculation revealed that the
increased exponentially and decreased linearly as the atomic number of the alkali metal increased due to the ionic potential, respectively. This calculation revealed that the 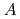 and
and  in the
in the  were close and reasonable for each other.
were close and reasonable for each other.
Uno, Masayoshi*; Nishi, Tsuyoshi*; Takano, Masahide
Comprehensive Nuclear Materials, 2nd Edition, Vol.7, p.202 - 231, 2020/08
On the thermodynamic and thermophysical properties of the actinide nitrides in Comprehensive Nuclear Materials published by Elsevier as the first edition in 2012, we have revised them by adding some brand-new data. The main topics added are the solid solubility of the actinide nitrides into the zirconium nitride matrix for transmutation fuel, the lattice expansion of actinide nitrides induced by self-irradiation damage, the influence of defects accumulation on thermal conductivity, and the thermal expansion in curium nitride lattice.
Ikusawa, Yoshihisa; Hirooka, Shun; Uno, Masayoshi*
2018 GIF Symposium Proceedings (Internet), p.321 - 327, 2020/05
Research and development of Minor actinides (MAs) bearing MOX fuel for fast reactor has been proceeding from the viewpoint of reducing radioactive waste. In order to develop, MA bearing MOX, it is indispensable to clarify the influence of MA addition on irradiation behavior. The addition of Americium (Am) to MOX affects vapor pressure and thermal conductivity, which are important properties from the perspective of evaluating fuel temperature. This is because vapor pressure affects fuel restructuring, and thermal conductivity affects fuel temperature distribution. Focusing on these physical properties, this study evaluates the influence of Am on fuel temperature using irradiation behavior analysis code to contribute to the development of MA-bearing MOX fuel. An increase in Am content decreases the thermal conductivity and increases the oxygen potential of oxide fuel. Because vapor pressure increases with increasing Am content, pore migration is accelerated, and the central void diameter increases with increasing Am content. As a result, after formation of the central void, the influence of Am content on the fuel center temperature is mild. Alpha particles generated by radioactive decay of transuranium elements cause lattice defects in the oxide fuel pellets. It is well known that this phenomenon, which is called self-irradiation, affects thermal conductivity. Since americium is the typical alpha radioactive nucleus, to evaluate fuel temperature of Am-MOX is necessary to take account of the influence of self-irradiation damage on thermal conductivity. Self-irradiation decreases thermal conductivity, and as the Am content increases, the rate of decrease in thermal conductivity is accelerated. Because it recovers with temperature rise, the decrease in thermal conductivity due to self-irradiation damage has very little effect on fuel center temperature. These results suggest that Am-MOX fuel could be irradiated under the same conditions as conventional MOX fuel.
Kawaguchi, Munemichi; Miyahara, Shinya*; Uno, Masayoshi*
Journal of Nuclear Engineering and Radiation Science, 6(2), p.021305_1 - 021305_9, 2020/04
Sodium-concrete reaction (SCR) is one of the important phenomena during severe accidents in sodium-cooled fast reactors (SFRs) owing to the generation of large sources of hydrogen and aerosols in the containment vessel. In this study, SCR experiments with an internal heater were performed to investigate the chemical reaction beneath the internal heater (800 C), which was used to simulate the obstacle and heating effect on SCR. Furthermore, the effects of the internal heater on the self-termination mechanism were discussed. The internal heater on the concrete hindered the transport of Na into the concrete. Therefore, Na could start to react with the concrete at the periphery of the internal heater, and the concrete ablation depth at the periphery was larger than under the internal heater. The high Na pool temperature of 800
C), which was used to simulate the obstacle and heating effect on SCR. Furthermore, the effects of the internal heater on the self-termination mechanism were discussed. The internal heater on the concrete hindered the transport of Na into the concrete. Therefore, Na could start to react with the concrete at the periphery of the internal heater, and the concrete ablation depth at the periphery was larger than under the internal heater. The high Na pool temperature of 800 C increased largely the Na aerosol release rate, which was explained by Na evaporation and hydrogen bubbling, and formed the porous reaction product layer, whose porosity was 0.54-0.59 from the mass balance of Si and image analyzing EPMA mapping. They had good agreement with each other. The porous reaction products decreased the amount of Na transport into the reaction front. The Na concentration around the reaction front became about 30wt.% despite the position of the internal heater. It was found that the Na concentration condition was one of the dominant parameters for the self-termination of SCR, even in the presence of the internal heater.
C increased largely the Na aerosol release rate, which was explained by Na evaporation and hydrogen bubbling, and formed the porous reaction product layer, whose porosity was 0.54-0.59 from the mass balance of Si and image analyzing EPMA mapping. They had good agreement with each other. The porous reaction products decreased the amount of Na transport into the reaction front. The Na concentration around the reaction front became about 30wt.% despite the position of the internal heater. It was found that the Na concentration condition was one of the dominant parameters for the self-termination of SCR, even in the presence of the internal heater.
Okamura, Hiroyuki; Mizuno, Masayoshi*; Hirayama, Naoki*; Shimojo, Kojiro; Naganawa, Hirochika; Imura, Hisanori*
Industrial & Engineering Chemistry Research, 59(1), p.329 - 340, 2020/01
Times Cited Count:15 Percentile:56.16(Engineering, Chemical)The synergistic ionic-liquid extraction and extraction equilibrium of lanthanoid(III) (Ln(III)) ions have been investigated using 2-thenoyltrifluoroacetone (Htta) and trioctylphosphine oxide (TOPO) in the ionic liquid 1-butyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([C mim][Tf
mim][Tf N]). The selective synergistic effect for heavier Ln(III) ions was found using a combination of Htta and TOPO in [C
N]). The selective synergistic effect for heavier Ln(III) ions was found using a combination of Htta and TOPO in [C mim][Tf
mim][Tf N], leading to enhanced separability among Ln(III) ions. The extracted Ln(III) species and the extraction constants in the Htta-TOPO system were determined by three-dimensional extraction equilibrium analysis. The selective synergism for heavier Ln(III) ions in the Htta-TOPO system was ascribed to the formation of hydrophobic, charged adducts, such as Ln(tta)
N], leading to enhanced separability among Ln(III) ions. The extracted Ln(III) species and the extraction constants in the Htta-TOPO system were determined by three-dimensional extraction equilibrium analysis. The selective synergism for heavier Ln(III) ions in the Htta-TOPO system was ascribed to the formation of hydrophobic, charged adducts, such as Ln(tta) (TOPO)
(TOPO)
 and Ln(tta)(TOPO)
and Ln(tta)(TOPO)
 , in [C
, in [C mim][Tf
mim][Tf N].
N].
Kawaguchi, Munemichi; Miyahara, Shinya; Uno, Masayoshi*
Journal of Nuclear Science and Technology, 56(6), p.513 - 520, 2019/06
Times Cited Count:2 Percentile:20.94(Nuclear Science & Technology)This study revealed melting points and thermal conductivities of four samples generated by sodium-concrete reaction (SCR). We prepared the samples using two methods such as firing mixtures of sodium and grinded concrete powder, and sampling depositions after the SCR experiments. In the former, the mixing ratios were determined from the past experiment. The latter simulated the more realistic conditions such as the temperature history and the distribution of Na and concrete. The thermogravimetry-differential thermal analyzer (TG-DTA) measurement showed the melting points were 865-942 C, but those of the samples containing metallic Na couldn't be clarified. In the two more realistic samples, the compression moldings in a furnace were observed. The observation revealed the softening temperature was 800-840
C, but those of the samples containing metallic Na couldn't be clarified. In the two more realistic samples, the compression moldings in a furnace were observed. The observation revealed the softening temperature was 800-840 C and the melting point was 840-850
C and the melting point was 840-850 C, which was 10-20
C, which was 10-20 C lower than the TG-DTA results. The thermodynamics calculation of FactSage 7.2 revealed the temperature of the onset of melting was caused by melting of the some components such as Na
C lower than the TG-DTA results. The thermodynamics calculation of FactSage 7.2 revealed the temperature of the onset of melting was caused by melting of the some components such as Na SiO
SiO and/or Na
and/or Na SiO
SiO . Moreover, the thermal conductivity was
. Moreover, the thermal conductivity was  =1-3W/m-K, which was comparable to xNa
=1-3W/m-K, which was comparable to xNa O-1-xSiO
O-1-xSiO (x=0.5, 0.33, 0.25), and those at 700
(x=0.5, 0.33, 0.25), and those at 700 C were explained by the equation of
C were explained by the equation of 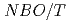 .
.
Doi, Daisuke; Seino, Hiroshi; Miyahara, Shinya*; Uno, Masayoshi*
Journal of Nuclear Science and Technology, 56(6), p.521 - 532, 2019/06
Times Cited Count:1 Percentile:10.65(Nuclear Science & Technology)Ikusawa, Yoshihisa; Morimoto, Kyoichi; Kato, Masato; Saito, Kosuke; Uno, Masayoshi*
Nuclear Technology, 205(3), p.474 - 485, 2019/03
Times Cited Count:2 Percentile:20.94(Nuclear Science & Technology)This study evaluated the effects of plutonium content and self-irradiation on the thermal conductivity of mixed-oxide (MOX) fuel. Samples of UO fuel and various MOX fuels were tested. The MOX fuels had a range of plutonium contents, and some samples were stored for 20 years. The thermal conductivity of these samples was determined from thermal diffusivity measurements taken via laser flash analysis. Although the thermal conductivity decreased with increasing plutonium content, this effect was slight. The effect of self-irradiation was investigated using the stored samples. The reduction in thermal conductivity caused by self-irradiation depended on the plutonium content, its isotopic composition, and storage time. The reduction in thermal conductivity over 20 years' storage can be predicted from the change of lattice parameter. In addition, the decrease in thermal conductivity caused by self-irradiation was recovered with heat treatment, and recovered almost completely at temperatures over 1200 K. From these evaluation results, we formulated an equation for thermal conductivity that is based on the classical phonon-transport model. This equation can predict the thermal conductivity of MOX fuel thermal conductivity by accounting for the influences of plutonium content and self-irradiation.
fuel and various MOX fuels were tested. The MOX fuels had a range of plutonium contents, and some samples were stored for 20 years. The thermal conductivity of these samples was determined from thermal diffusivity measurements taken via laser flash analysis. Although the thermal conductivity decreased with increasing plutonium content, this effect was slight. The effect of self-irradiation was investigated using the stored samples. The reduction in thermal conductivity caused by self-irradiation depended on the plutonium content, its isotopic composition, and storage time. The reduction in thermal conductivity over 20 years' storage can be predicted from the change of lattice parameter. In addition, the decrease in thermal conductivity caused by self-irradiation was recovered with heat treatment, and recovered almost completely at temperatures over 1200 K. From these evaluation results, we formulated an equation for thermal conductivity that is based on the classical phonon-transport model. This equation can predict the thermal conductivity of MOX fuel thermal conductivity by accounting for the influences of plutonium content and self-irradiation.
Aoyagi, Mitsuhiro; Takata, Takashi; Ohno, Shuji; Uno, Masayoshi*
Proceedings of 12th International Topical Meeting on Nuclear Reactor Thermal-Hydraulics, Operation and Safety (NUTHOS-12) (USB Flash Drive), 10 Pages, 2018/10
Heat radiation is one of dominant heat transfer process during a sodium fire event which is a concern in sodium-cooled fast reactor plants. This study aims to model radiation heat transfer from combusting droplets. Radiation energy transport on the combustion flame surface around a sodium droplet is formulated considering emission, absorption and scattering through a similar approach to the formulation of the wall boundary condition. The improved model is tested trough a simple verification analysis and a benchmark analysis on an upward sodium spray combustion experiment. As the result, overestimation of atmospheric temperature and pressure is mitigated by the improved model due to increase in heat transfer to structure.
Kawaguchi, Munemichi; Miyahara, Shinya; Uno, Masayoshi*
Journal of Nuclear Science and Technology, 55(8), p.874 - 884, 2018/08
Times Cited Count:4 Percentile:37.66(Nuclear Science & Technology)As parts of severe accident studies in sodium-cooled fast reactor, experiments were performed to investigate the termination mechanism of sodium-concrete reaction (SCR). In the experiment, the reaction time was controlled to investigate the distribution change of sodium (Na) and the reaction products in the pool and around the reaction front. In the results, the Na around the reaction front decreased from the enough amount with the reaction time. The concentrations were 18-24 wt.% for Na, and 22-18 wt.% for Si after the termination. From the thermodynamics calculations, the stable materials around the reaction front comprised more than 90 wt.% solid products such as Na SiO
SiO , and no Na. Further, the distribution of Na and reaction products could be explained by a steady-state sedimentation-diffusion model. At the early stage of SCR, the reaction products were suspended as particles in the Na pool because of the high H
, and no Na. Further, the distribution of Na and reaction products could be explained by a steady-state sedimentation-diffusion model. At the early stage of SCR, the reaction products were suspended as particles in the Na pool because of the high H -generation rate. As the concrete ablation proceeds, they start settling down due to the decreased H
-generation rate. As the concrete ablation proceeds, they start settling down due to the decreased H -generation rate, thereby allowing SCR termination. It was concluded that SCR termination was caused by the sediment of the reaction products and the lack of Na around the reaction front.
-generation rate, thereby allowing SCR termination. It was concluded that SCR termination was caused by the sediment of the reaction products and the lack of Na around the reaction front.
Kawaguchi, Munemichi; Miyahara, Shinya; Uno, Masayoshi*
Proceedings of 26th International Conference on Nuclear Engineering (ICONE-26) (Internet), 8 Pages, 2018/07
Sodium-concrete reaction (SCR) is one of the important phenomena during severe accidents in sodium-cooled fast reactors (SFRs) owing to the presence of large sources of hydrogen and aerosols in the containment vessel. In this study, SCR experiments with an internal heater (800 C) were performed to investigate the chemical reaction under the internal heater. Furthermore, the effects of the internal heater on the self-termination mechanism were discussed. Because the internal heater hindered the transport of Na, the moisture in the concrete, and reaction products, Na could permeate and react with the surface concrete at the periphery of the internal heater. As the SCR proceeded, the reaction products accumulated under the internal heater and disturbed the Na diffusion. Therefore, the Na concentration under the internal heater decreased relatively lower, and the concrete ablation depth under the internal heater decreased compared to that under the periphery of the internal heater. However, the Na concentration around the reaction front was about 30 wt.% despite the position of the internal heater. The Na concentration was similar to that of Na
C) were performed to investigate the chemical reaction under the internal heater. Furthermore, the effects of the internal heater on the self-termination mechanism were discussed. Because the internal heater hindered the transport of Na, the moisture in the concrete, and reaction products, Na could permeate and react with the surface concrete at the periphery of the internal heater. As the SCR proceeded, the reaction products accumulated under the internal heater and disturbed the Na diffusion. Therefore, the Na concentration under the internal heater decreased relatively lower, and the concrete ablation depth under the internal heater decreased compared to that under the periphery of the internal heater. However, the Na concentration around the reaction front was about 30 wt.% despite the position of the internal heater. The Na concentration was similar to that of Na SiO
SiO , which was almost same as that in our past study. It was found that the Na concentration condition was one of the dominant parameters for the self-termination of SCR, even in the presence of the internal heater.
, which was almost same as that in our past study. It was found that the Na concentration condition was one of the dominant parameters for the self-termination of SCR, even in the presence of the internal heater.
Aoyagi, Mitsuhiro; Takata, Takashi; Ohno, Shuji; Uno, Masayoshi*
Nihon Kikai Gakkai Rombunshu (Internet), 84(859), p.17-00374_1 - 17-00374_13, 2018/03
no abstracts in English
Kawaguchi, Munemichi; Miyahara, Shinya; Uno, Masayoshi*
Netsu Sokutei, 45(1), p.2 - 8, 2018/01
Liquid sodium (Na) has been used as the coolant of fast reactors for the various merits, such as the high thermal conductivity. On the other hand, it is postulated that a steel liner may fail and lead to a sodium-concrete reaction (SCR) during the Na-leak accident. Because of concrete ablation and release of hydrogen gas due to the chemical reactions between Na and concrete components, the SCR is one of the important phenomena in the Na-leak accident. In the study, fundamental experiments related to the SCR were performed using Na and concrete powder. Here, the used concrete powder is milled siliceous concrete which is usually used as the structural concrete in Japanese nuclear power plants. The obvious temperature changes at 3 temperature regions were observed for the reaction process such as Na-melt, NaOH-SiO and Na-H
and Na-H O-SiO
O-SiO reaction, which occurred around 100, 300 and 500
reaction, which occurred around 100, 300 and 500 C, respectively. Especially, the violent reaction around 500
C, respectively. Especially, the violent reaction around 500 C caused the temperature peak to
C caused the temperature peak to 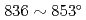 C, and the reaction heat of
C, and the reaction heat of 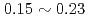 kW/g was estimated under the Na-concrete mixing ratio such as
kW/g was estimated under the Na-concrete mixing ratio such as 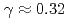 . The main components of the reaction products was identified as Na
. The main components of the reaction products was identified as Na SiO
SiO with X-ray diffraction technique. Moreover, the measured thermophysical properties such as melting point, density, specific heat, thermal conductivity and viscosity were similar to those of
with X-ray diffraction technique. Moreover, the measured thermophysical properties such as melting point, density, specific heat, thermal conductivity and viscosity were similar to those of 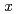 Na
Na O-
O-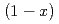 SiO
SiO (
(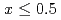 ).
).