Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Miyazaki, Kanako*; Takehara, Masato*; Minomo, Kenta*; Horie, Kenji*; Takehara, Mami*; Yamasaki, Shinya*; Saito, Takumi*; Onuki, Toshihiko*; Takano, Masahide; Shiotsu, Hiroyuki; et al.
Journal of Hazardous Materials, 470(15), p.134104_1 - 134104_11, 2024/05
Times Cited Count:1 Percentile:31.14(Engineering, Environmental)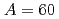
Tamii, Atsushi*; Pellegri, L.*; S derstr
derstr m, P.-A.*; Allard, D.*; Goriely, S.*; Inakura, Tsunenori*; Khan, E.*; Kido, Eiji*; Kimura, Masaaki*; Litvinova, E.*; et al.
m, P.-A.*; Allard, D.*; Goriely, S.*; Inakura, Tsunenori*; Khan, E.*; Kido, Eiji*; Kimura, Masaaki*; Litvinova, E.*; et al.
European Physical Journal A, 59(9), p.208_1 - 208_21, 2023/09
Times Cited Count:8 Percentile:83.85(Physics, Nuclear)no abstracts in English
Fueda, Kazuki*; Komiya, Tatsuki*; Minomo, Kenta*; Horie, Kenji*; Takehara, Mami*; Yamasaki, Shinya*; Shiotsu, Hiroyuki; Onuki, Toshihiko*; Grambow, B.*; Law, G. T. W.*; et al.
Chemosphere, 328, p.138566_1 - 138566_12, 2023/07
Times Cited Count:5 Percentile:50.39(Environmental Sciences)Onuki, Toshihiko*; Ye, J.*; Kato, Tomoaki; Liu, J.; Takano, Masahide; Kozai, Naofumi; Utsunomiya, Satoshi*
Environmental Science; Processes & Impacts, 25(7), p.1204 - 1212, 2023/07
Times Cited Count:2 Percentile:23.37(Chemistry, Analytical)To elucidate chemical forms of Cs and I in microparticles produced via the Fukushima Daiichi Nuclear Power Plant accident and released into the atmosphere, we analyzed Cs and I in condensed vaporized particles (CVP) produced by melting experiments using nuclear fuel components containing CsI with concrete. CVPs consisted of many round particles containing Cs and I of diameters less than several tens of micrometers. Two kinds of particles were present: one containing large amounts of Cs and I, suggesting the presence of CsI, and the other containing small amounts of Cs and I with large Si contents. Most of CsI from both particles were dissolved in water. On the contrary, some fractions of Cs remained from the latter particles. These results suggest that Cs was incorporated in CVPs along with Si to form water low-soluble CVPs
Kato, Tomoaki; Kozai, Naofumi; Tanaka, Kazuya; Kaplan, D. I.*; Utsunomiya, Satoshi*; Onuki, Toshihiko
Journal of Nuclear Science and Technology, 59(5), p.580 - 589, 2022/05
Times Cited Count:6 Percentile:55.57(Nuclear Science & Technology)This study reports the effect of fulvic acids, which is a natural organic substance generally contained in groundwater, on the oxidation states of radioactive iodine anions (iodide and iodate). Iodide and iodate are contained in the contaminated water of the Fukushima Daiichi Nuclear Power Plant and supposed to be removed by activated carbon (AC) via adsorption. When fulvic acids does not exist in the experimental system, the adsorption of iodide on AC was less than that of iodate and their oxidation states after the adsorption were not changed. When fulvic acids existed, a fraction of the adsorbed iodate was reduced to iodide. This result indicates that the reduction of the adsorbed iodate progresses during the storage of the spent AC.
 C control rods in Fukushima Daiichi nuclear reactors during meltdown; B-Li isotopic signatures in cesium-rich microparticles
C control rods in Fukushima Daiichi nuclear reactors during meltdown; B-Li isotopic signatures in cesium-rich microparticlesFueda, Kazuki*; Takami, Ryu*; Minomo, Kenta*; Morooka, Kazuya*; Horie, Kenji*; Takehara, Mami*; Yamasaki, Shinya*; Saito, Takumi*; Shiotsu, Hiroyuki; Onuki, Toshihiko*; et al.
Journal of Hazardous Materials, 428, p.128214_1 - 128214_10, 2022/04
Times Cited Count:12 Percentile:66.41(Engineering, Environmental)Grambow, B.; Nitta, Ayako; Shibata, Atsuhiro; Koma, Yoshikazu; Utsunomiya, Satoshi*; Takami, Ryu*; Fueda, Kazuki*; Onuki, Toshihiko*; Jegou, C.*; Laffolley, H.*; et al.
Journal of Nuclear Science and Technology, 59(1), p.1 - 24, 2022/01
Times Cited Count:32 Percentile:76.55(Nuclear Science & Technology)Onuki, Toshihiko*; Ozaki, Takuo*; Kozai, Naofumi; Utsunomiya, Satoshi*
Behavior of Radionuclides in the Environment I; Function of Particles in Aquatic System, p.67 - 92, 2020/00
It has been experimentally revealed that microorganisms transform radionuclides. For example, cells of microorganisms such as bacteria and yeast accumulate actinides on the surface. Phosphate ions released from microorganism cells precipitate actinides as phosphates. This chapter discusses the role of environmental microorganisms on migration of actinides in the groundwater around Lake Karachai where a lot of radioactive wastes from nuclear facilities of the Soviet Union.
Onuki, Toshihiko*; Satou, Yukihiko; Utsunomiya, Satoshi*
Journal of Nuclear Science and Technology, 56(9-10), p.790 - 800, 2019/09
Times Cited Count:10 Percentile:60.45(Nuclear Science & Technology)Horiike, Takumi*; Dotsuta, Yuma*; Nakano, Yuriko*; Ochiai, Asumi*; Utsunomiya, Satoshi*; Onuki, Toshihiko; Yamashita, Mitsuo*
Applied and Environmental Microbiology, 83(20), p.e00855-17_1 - e00855-17_11, 2017/10
Times Cited Count:19 Percentile:59.78(Biotechnology & Applied Microbiology)Radioactive strontium ( Sr) leaked into the ocean from the Fukushima Daiichi Nuclear Power Plant after the nuclear accident. Since the removal of Sr using general adsorbents is not efficient at high salinity, this study investigated removal of soluble Sr from a saline solution by biogenic carbonate minerals. An isolate, Bacillus sp. strain TK2d, from marine sediment removed more than 99 % of Sr in a saline solution. Sr adsorbed at the cell surface and then carbonate minerals containing Sr developed outside the cells.
Sr) leaked into the ocean from the Fukushima Daiichi Nuclear Power Plant after the nuclear accident. Since the removal of Sr using general adsorbents is not efficient at high salinity, this study investigated removal of soluble Sr from a saline solution by biogenic carbonate minerals. An isolate, Bacillus sp. strain TK2d, from marine sediment removed more than 99 % of Sr in a saline solution. Sr adsorbed at the cell surface and then carbonate minerals containing Sr developed outside the cells.
Onuki, Toshihiko; Kozai, Naofumi; Sakamoto, Fuminori; Utsunomiya, Satoshi*; Kato, Kenji*
Chemistry Letters, 46(5), p.771 - 774, 2017/05
Times Cited Count:0 Percentile:0.00(Chemistry, Multidisciplinary)The sorption behavior of Np(V) by the microbe consortia and by a single pure culture of Fe reducing bacterium was studied at pH between 3 and 7 in resting cell conditions. The sorption of Np(V) by the Fe reducing bacterium obtained in the inert condition and by the consortia in aerated condition were higher than by the Fe reducing bacterium in aerobic condition at pH below 5, strongly suggesting presence of other mechanism than the adsorption on microbial cell surface, i.e. reduction to Np(IV).
Yamasaki, Shinya*; Imoto, Jumpei*; Furuki, Genki*; Ochiai, Asumi*; Onuki, Toshihiko; Sueki, Keisuke*; Namba, Kenji*; Ewing, R. C.*; Utsunomiya, Satoshi*
Science of the Total Environment, 551-552, p.155 - 162, 2016/05
Times Cited Count:36 Percentile:70.46(Environmental Sciences)Cesium-137 ( Cs) of estuary sediment impacted by the FDNPP was measured. Increasing radioactivity was observed from surface to bottom. 90% of the
Cs) of estuary sediment impacted by the FDNPP was measured. Increasing radioactivity was observed from surface to bottom. 90% of the  Cs was strongly bound to clay minerals in the estuary sediments. These results suggest that
Cs was strongly bound to clay minerals in the estuary sediments. These results suggest that  Cs is being transported from contaminated paddy fields to the estuary.
Cs is being transported from contaminated paddy fields to the estuary.
Tanaka, Kazuya; Takahashi, Yoshio*; Fukushi, Keisuke*; Utsunomiya, Satoshi*
Chikyu Kagaku, 49(4), p.169 - 171, 2015/12
Many studies have been carried out since the Fukushima Dai-ichi Nuclear Power Plant (FDNPP) accident. This paper introduces the special issue on the FDNPP accident which includes contributions from various fields covering aerosols, soil, forest, river, ocean and application of new analytical technique to samples collected in Fukushima.
Kaneko, Makoto*; Iwata, Hajime; Shiotsu, Hiroyuki; Masaki, Shota*; Kawamoto, Yuji*; Yamasaki, Shinya*; Nakamatsu, Yuki*; Imoto, Jumpei*; Furuki, Genki*; Ochiai, Asumi*; et al.
Frontiers in Energy Research (Internet), 3, p.37_1 - 37_10, 2015/09
The mobility of the aggregates of submicron-sized sheet aluminosilicate in the surface environment is a key factor controlling the current Cs migration in Fukushima.
Onuki, Toshihiko; Jiang, M.*; Sakamoto, Fuminori; Kozai, Naofumi; Yamasaki, Shinya*; Yu, Q.; Tanaka, Kazuya; Utsunomiya, Satoshi*; Xia, X.*; Yange, K.*; et al.
Geochimica et Cosmochimica Acta, 163, p.1 - 13, 2015/08
Times Cited Count:26 Percentile:61.03(Geochemistry & Geophysics)The association of Ce(III) with the microbial cell surface and the formation of Ce phosphate nano-particles are responsible for suppressing the oxidation of Ce(III) to Ce(IV) in the mixtures.
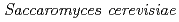
Onuki, Toshihiko; Sakamoto, Fuminori; Yamasaki, Shinya*; Kozai, Naofumi; Shiotsu, Hiroyuki; Utsunomiya, Satoshi*; Watanabe, Naoko*; Kozaki, Tamotsu*
Journal of Environmental Radioactivity, 144, p.127 - 133, 2015/06
Times Cited Count:10 Percentile:27.06(Environmental Sciences)The accumulation of Cs by unicellular fungus of 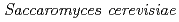 in the presence of minerals has been studied to elucidate the role of microorganisms in the migration of radioactive Cs in the environment. In the presence of minerals in the agar medium, the radioactivity in the yeast cells was in the order of mica
in the presence of minerals has been studied to elucidate the role of microorganisms in the migration of radioactive Cs in the environment. In the presence of minerals in the agar medium, the radioactivity in the yeast cells was in the order of mica  smectite, illite
smectite, illite 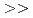 vermiculite, phlogopite, zeolite. This order is inversely correlated to the ratio of the concentration of radioactive Cs between the minerals and the medium solution. These results strongly suggest that the yeast accumulates radioactive Cs competitively with minerals.
vermiculite, phlogopite, zeolite. This order is inversely correlated to the ratio of the concentration of radioactive Cs between the minerals and the medium solution. These results strongly suggest that the yeast accumulates radioactive Cs competitively with minerals.
 nanoparticles on microbial metabolism
nanoparticles on microbial metabolismMasaki, Shota*; Shiotsu, Hiroyuki; Onuki, Toshihiko; Sakamoto, Fuminori; Utsunomiya, Satoshi*
Chemical Geology, 391, p.33 - 41, 2015/01
Times Cited Count:10 Percentile:31.18(Geochemistry & Geophysics)Distinct organic species and intracellular proteins were expressed after exposure of yeast cells to CeNPs. Although cytotoxicity was not caused by CeNPs, the results of the peptide mass fingerprint analysis of the intracellular protein revealed that Eno2p, a glycolysis enzyme, was expressed after the exposure to CeNPs. These results suggest that nanoparticles have the potential to alter microbial metabolism, leading to changes in the compositions of the released substances in the surrounding environment.
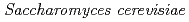 fungal cells; A pH-dependent contribution of phosphoryl functional group
fungal cells; A pH-dependent contribution of phosphoryl functional groupJiang, M. Y.*; Onuki, Toshihiko; Yamasaki, Shinya; Tanaka, Kazuya*; Utsunomiya, Satoshi*
Journal of Radioanalytical and Nuclear Chemistry, 295(3), p.2283 - 2287, 2013/03
Times Cited Count:3 Percentile:24.16(Chemistry, Analytical)The adsorption of Ytterbium on the cells of yeast 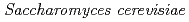 has been studied by batch type experiment by changing solution pH. The Yb adsorption species on the yeast cell wall of the
has been studied by batch type experiment by changing solution pH. The Yb adsorption species on the yeast cell wall of the 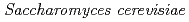 was determined by extended X-ray absorption fine structure (EXAFS) spectroscopy combined with a linear combination analysis at various pHs. The results indicated that the contribution of Yb-phosphoryl species was constant between pH 3 and 5, strongly suggesting that most of the Yb was associated with undeprotonated phosphoryl functional groups.
was determined by extended X-ray absorption fine structure (EXAFS) spectroscopy combined with a linear combination analysis at various pHs. The results indicated that the contribution of Yb-phosphoryl species was constant between pH 3 and 5, strongly suggesting that most of the Yb was associated with undeprotonated phosphoryl functional groups.
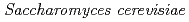
Jiang, M. Y.*; Onuki, Toshihiko; Tanaka, Kazuya*; Kozai, Naofumi; Kamiishi, Eigo; Utsunomiya, Satoshi*
Geochimica et Cosmochimica Acta, 93, p.30 - 46, 2012/09
Times Cited Count:33 Percentile:64.04(Geochemistry & Geophysics)We have investigated the post-adsorption process of ytterbium (Yb) phosphate nano-particle formation by 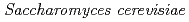 . We have elucidated the nano particle formation by TEM and EXAFS analyses, that adsorbed Yb reacts on the cell surface with the released pohosphate from inside the cell.
. We have elucidated the nano particle formation by TEM and EXAFS analyses, that adsorbed Yb reacts on the cell surface with the released pohosphate from inside the cell.
Kiyanagi, Yoshiaki*; Kino, Koichi*; Furusaka, Michihiro*; Hiraga, Fujio*; Kamiyama, Takashi*; Kato, Kiyoshi*; Igashira, Masayuki*; Katabuchi, Tatsuya*; Mizumoto, Motoharu*; Oshima, Masumi; et al.
Journal of the Korean Physical Society, 59(2), p.1781 - 1784, 2011/08
Times Cited Count:13 Percentile:61.03(Physics, Multidisciplinary)The project of the comprehensive nuclear data research for the development of the advanced reactor systems had been executed successfully by eight organizations from 2005 to 2009. In this project, we constructed the pulsed neutron beamline that was aimed to obtain neutron capture cross-sections of long-lived fission products and minor actinides accurately. The energy spectra, spatial distributions, and pulses of the beam were studied by measurements and simulation calculations, and they were found to be consistent with those of the beamline design. In this paper, we present the overview of the project and the properties of the neutron beam provided by this beam line.