Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 -PuO
-PuO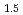 -PuO
-PuO system
systemVinograd, V. L.*; Vauchy, R.
Journal of Nuclear Materials, 619, p.156244_1 - 156244_16, 2026/01
Times Cited Count:0 Percentile:0.00(Materials Science, Multidisciplinary)Thermodynamic properties of (U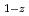 Pu
Pu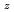 )O
)O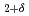 fluorite (FCC) and (U
fluorite (FCC) and (U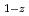 Pu
Pu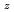 )O
)O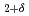 bixbyite (BCC) in the UO
bixbyite (BCC) in the UO -PuO
-PuO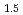 -PuO
-PuO system are assessed by considering phase equilibrium constraints and data on the variation of oxygen to metal ratio (O/M) as a function of the chemical potential of O
system are assessed by considering phase equilibrium constraints and data on the variation of oxygen to metal ratio (O/M) as a function of the chemical potential of O . Thermodynamically, both BCC and FCC are described as ordered solid solutions allowing for a decrease in the configurational entropy of their oxygen/vacancy distributions at the specific values of
. Thermodynamically, both BCC and FCC are described as ordered solid solutions allowing for a decrease in the configurational entropy of their oxygen/vacancy distributions at the specific values of  = -0.5 and
= -0.5 and  = -0.375 (O/M = 1.5 and O/M = 1.625). With this approach, fluorite/bixbyite equilibria in PuO
= -0.375 (O/M = 1.5 and O/M = 1.625). With this approach, fluorite/bixbyite equilibria in PuO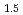 -PuO
-PuO and in UO
and in UO -PuO
-PuO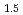 -PuO
-PuO are reproduced well with little effort. Moreover, we show that a large manifold of experimental data on the UO
are reproduced well with little effort. Moreover, we show that a large manifold of experimental data on the UO -PuO
-PuO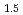 -PuO
-PuO system is consistent with the assumption that Pu/(Pu+U) ratios in individual phases remain equal to the total Pu/(Pu+U) ratio in the system, i.e., no inter-phase U/Pu-partitioning occurs.
system is consistent with the assumption that Pu/(Pu+U) ratios in individual phases remain equal to the total Pu/(Pu+U) ratio in the system, i.e., no inter-phase U/Pu-partitioning occurs.
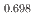 Pu
Pu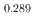 Am
Am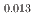 O
O
Vauchy, R.; Horii, Yuta; Hirooka, Shun; Akashi, Masatoshi; Sunaoshi, Takeo*; Nakamichi, Shinya; Saito, Kosuke
Proceedings of 34th Nuclear Energy for New Europe (NENE2025) , p.232 - 238, 2026/01
Vauchy, R.; Horii, Yuta; Hirooka, Shun; Akashi, Masatoshi; Sunaoshi, Takeo*; Nakamichi, Shinya; Saito, Kosuke
Journal of Nuclear Materials, 616, p.156115_1 - 156115_16, 2025/10
Times Cited Count:1 Percentile:59.09(Materials Science, Multidisciplinary)Hirooka, Shun; Vauchy, R.; Horii, Yuta; Sunaoshi, Takeo*; Saito, Kosuke
Proceedings of TopFuel 2025; Nuclear Reactor Fuel Performance Conference (Internet), 10 Pages, 2025/10
Reducing the oxygen-to-metal (O/M) ratio in MOX fuels plays an important role in suppressing the corrosion depth in the cladding due to fuel-cladding chemical interaction (FCCI), which is the key to determining the lifetime of fast reactor MOX fuels. Owing to a number of irradiation and post-irradiation experiments, a clear decreasing trend in the corrosion depth with lower O/M ratio in the as-fabricated MOX pellet was reported, whereas a significant redistribution of the O/M ratio in a pellet driven by the radial temperature gradient during irradiation could supply a higher oxygen potential near the pellet periphery where the FCCI should occur. The reduction of the O/M ratio in the MOX pellet fabrication processes can be achieved by heat treatment by taking high temperature, longer time, and lower oxygen partial pressure in the gas into account; the properties governing the reduction are not sufficiently studied. This study demonstrated the variation of O/M ratio in MOX pellets and the in-situ O/M ratio during the heat treatment was analyzed by using a thermogravimeter, which revealed a decreasing behavior during heating and dwell as well as an increasing behavior in the O/M ratio during the cooling step. Furthermore, the redistribution of O/M ratio, analyzed by Sari's model, was discussed to investigate the O/M ratio and the oxygen potential near the pellet periphery which is likely to have a more direct influence on the FCCI than the as-fabricated O/M ratio. By using the recent oxygen potential data on MOX, it is found that the oxygen potential profile in the radial direction is especially drastic near the pellet periphery and is sensitive to the as-fabricated O/M ratio.
Hirooka, Shun; Vauchy, R.; Horii, Yuta; Sunaoshi, Takeo*; Saito, Kosuke; Ozawa, Takayuki
Proceedings of Workshop on Fuel Performance Assessment and Behaviour for Liquid Metal Cooled Fast Reactors (Internet), 8 Pages, 2025/07
no abstracts in English
Vauchy, R.
Kaku Nenryo, (60-1), p.9 - 11, 2025/02
Plutonium Futures is historically an international conference focusing on plutonium science. As a result, one of the main topics is naturally military applications (nuclear weapons) but nuclear fuels (oxides, carbides, nitrides, metals) and reprocessing are also now a large part the topics presented at the conference.
Vauchy, R.; Hirooka, Shun; Saito, Kosuke
Materials Today Communications (Internet), 41, p.110676_1 - 110676_17, 2024/12
Times Cited Count:0 Percentile:0.00(Materials Science, Multidisciplinary)Oxygen potential measurements of PuO reported in the open literature were reviewed and re-interpreted using the defect chemistry model developed by our team. An empirical, easy-to-use, relationship connecting the O/Pu ratio, the equilibrium oxygen potential, and the temperature is proposed based on the interpolation of the experimental data in the 953-2100 K temperature range. The effect of americium on the oxygen potential of PuO
reported in the open literature were reviewed and re-interpreted using the defect chemistry model developed by our team. An empirical, easy-to-use, relationship connecting the O/Pu ratio, the equilibrium oxygen potential, and the temperature is proposed based on the interpolation of the experimental data in the 953-2100 K temperature range. The effect of americium on the oxygen potential of PuO is also discussed.
is also discussed.
Vauchy, R.; Hirooka, Shun; Horii, Yuta; Ogasawara, Masahiro*; Sunaoshi, Takeo*; Yamada, Tadahisa*; Tamura, Tetsuya*; Murakami, Tatsutoshi
Journal of Nuclear Materials, 599, p.155233_1 - 155233_11, 2024/10
Times Cited Count:2 Percentile:44.79(Materials Science, Multidisciplinary)The fluorite exsolution/recombination in U Pu
Pu O
O (y = 0.30 and 0.45) and PuO
(y = 0.30 and 0.45) and PuO was investigated using differential scanning calorimetry. The results are in relatively good agreement with the literature data, except for plutonia. Our values indicate that the critical temperature of the miscibility gap in Pu-O is 30
was investigated using differential scanning calorimetry. The results are in relatively good agreement with the literature data, except for plutonia. Our values indicate that the critical temperature of the miscibility gap in Pu-O is 30 50 K lower than previously reported. Finally, the systematic experimental procedure allowed us refining the locus of the solvus existing in hypostoichiometric U
50 K lower than previously reported. Finally, the systematic experimental procedure allowed us refining the locus of the solvus existing in hypostoichiometric U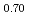 0Pu
0Pu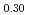 O
O , U
, U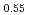 Pu
Pu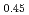 O
O , and PuO
, and PuO dioxides.
dioxides.
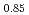 Am
Am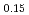 O
O at 1473, 1573, and 1673 K
at 1473, 1573, and 1673 KWatanabe, Masashi; Yokoyama, Keisuke; Vauchy, R.; Kato, Masato; Sugata, Hiromasa*; Seki, Takayuki*; Hino, Tetsushi*
Journal of Nuclear Materials, 599, p.155232_1 - 155232_5, 2024/10
Times Cited Count:3 Percentile:60.19(Materials Science, Multidisciplinary)Oxygen potential data of U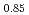 Am
Am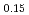 O
O were measured at 1473, 1573, and 1673 K by thermogravimetry. In U
were measured at 1473, 1573, and 1673 K by thermogravimetry. In U An
An O
O , where An stands for Pu or Am, and for a given value of y and Oxygen/Metal ratio, the oxygen potential of U
, where An stands for Pu or Am, and for a given value of y and Oxygen/Metal ratio, the oxygen potential of U Am
Am O
O is higher than that of U
is higher than that of U Pu
Pu O
O . The valence of cations in the hypostoichiometric region is similar to that of Nd-doped UO
. The valence of cations in the hypostoichiometric region is similar to that of Nd-doped UO . At the stoichiometric composition, it is estimated to be Am
. At the stoichiometric composition, it is estimated to be Am , U
, U , and U
, and U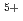 (for charge compensation of Am
(for charge compensation of Am ). The experimental data were analyzed using a defect chemistry model, and a relationship connecting the oxygen-to-metal ratio, the temperature, and the equilibrium oxygen partial pressure was proposed.
). The experimental data were analyzed using a defect chemistry model, and a relationship connecting the oxygen-to-metal ratio, the temperature, and the equilibrium oxygen partial pressure was proposed.
 Pu
Pu O
O powder using master sintering curve theory
powder using master sintering curve theoryNakamichi, Shinya; Sunaoshi, Takeo*; Hirooka, Shun; Vauchy, R.; Murakami, Tatsutoshi
Journal of Nuclear Materials, 595, p.155072_1 - 155072_11, 2024/07
Times Cited Count:1 Percentile:23.55(Materials Science, Multidisciplinary)Kato, Masato; Oki, Takumi; Watanabe, Masashi; Hirooka, Shun; Vauchy, R.; Ozawa, Takayuki; Uwaba, Tomoyuki; Ikusawa, Yoshihisa; Nakamura, Hiroki; Machida, Masahiko
Journal of the American Ceramic Society, 107(5), p.2998 - 3011, 2024/05
Times Cited Count:8 Percentile:49.59(Materials Science, Ceramics) mixed oxides
mixed oxidesVauchy, R.; Matsumoto, Taku; Hirooka, Shun; Uno, Hiroki*; Tamura, Tetsuya*; Arima, Tatsumi*; Inagaki, Yaohiro*; Idemitsu, Kazuya*; Nakamura, Hiroki; Machida, Masahiko; et al.
Journal of Nuclear Materials, 588, p.154786_1 - 154786_13, 2024/01
Times Cited Count:9 Percentile:84.33(Materials Science, Multidisciplinary)Vauchy, R.
Kaku Nenryo, (59-1), p.13 - 15, 2023/12
Vauchy, R.; Hirooka, Shun; Murakami, Tatsutoshi
Materialia, 32, p.101934_1 - 101934_12, 2023/12
Vauchy, R.; Hirooka, Shun; Murakami, Tatsutoshi
Materialia, 32, p.101943_1 - 101943_8, 2023/12
Hirooka, Shun; Horii, Yuta; Sunaoshi, Takeo*; Uno, Hiroki*; Yamada, Tadahisa*; Vauchy, R.; Hayashizaki, Kohei; Nakamichi, Shinya; Murakami, Tatsutoshi; Kato, Masato
Journal of Nuclear Science and Technology, 60(11), p.1313 - 1323, 2023/11
Times Cited Count:5 Percentile:63.07(Nuclear Science & Technology)Additive MOX pellets are fabricated by a conventional dry powder metallurgy method. Nd O
O and Sm
and Sm O
O are chosen as the additive materials to simulate the corresponding soluble fission products dispersed in MOX. Shrinkage curves of the MOX pellets are obtained by dilatometry, which reveal that the sintering temperature is shifted toward a value higher than that of the respective regular MOX. The additives, however, promote grain growth and densification, which can be explained by the effect of oxidized uranium cations covering to a pentavalent state. Ceramography reveals large agglomerates after sintering, and Electron Probe Micro-Analysis confirms that inhomogeneous elemental distribution, whereas XRD reveals a single face-centered cubic phase. Finally, by grinding and re-sintering the specimens, the cation distribution homogeneity is significantly improved, which can simulate spent nuclear fuels with soluble fission products.
are chosen as the additive materials to simulate the corresponding soluble fission products dispersed in MOX. Shrinkage curves of the MOX pellets are obtained by dilatometry, which reveal that the sintering temperature is shifted toward a value higher than that of the respective regular MOX. The additives, however, promote grain growth and densification, which can be explained by the effect of oxidized uranium cations covering to a pentavalent state. Ceramography reveals large agglomerates after sintering, and Electron Probe Micro-Analysis confirms that inhomogeneous elemental distribution, whereas XRD reveals a single face-centered cubic phase. Finally, by grinding and re-sintering the specimens, the cation distribution homogeneity is significantly improved, which can simulate spent nuclear fuels with soluble fission products.
Vauchy, R.; Hirooka, Shun; Watanabe, Masashi; Yokoyama, Keisuke; Murakami, Tatsutoshi
Journal of Nuclear Materials, 584, p.154576_1 - 154576_11, 2023/10
Times Cited Count:8 Percentile:80.73(Materials Science, Multidisciplinary)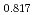 Pu
Pu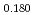 Am
Am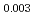 O
O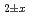 uranium-plutonium-americium mixed oxides at 1573, 1773, and 1873 K
uranium-plutonium-americium mixed oxides at 1573, 1773, and 1873 KVauchy, R.; Sunaoshi, Takeo*; Hirooka, Shun; Nakamichi, Shinya; Murakami, Tatsutoshi; Kato, Masato
Journal of Nuclear Materials, 580, p.154416_1 - 154416_11, 2023/07
Times Cited Count:10 Percentile:86.97(Materials Science, Multidisciplinary)Vauchy, R.; Hirooka, Shun; Watanabe, Masashi; Kato, Masato
Scientific Reports (Internet), 13, p.2217_1 - 2217_8, 2023/02
Times Cited Count:14 Percentile:68.65(Multidisciplinary Sciences) ceramics
ceramicsVauchy, R.; Hirooka, Shun; Watanabe, Masashi; Yokoyama, Keisuke; Sunaoshi, Takeo*; Yamada, Tadahisa*; Nakamichi, Shinya; Murakami, Tatsutoshi
Ceramics International, 49(2), p.3058 - 3065, 2023/01
Times Cited Count:13 Percentile:58.53(Materials Science, Ceramics)