Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tanaka, Kazuya; Yamaji, Keiko*; Masuya, Hayato*; Tomita, Jumpei; Ozawa, Mayumi*; Yamasaki, Shinya*; Tokunaga, Kohei; Fukuyama, Kenjin*; Ohara, Yoshiyuki*; Maamoun, I.*; et al.
Chemosphere, 355, p.141837_1 - 141837_11, 2024/05
In this study, biogenic Mn(IV) oxide was applied to remove Ra from mine water collected from a U mill tailings pond in the Ningyo-toge center. Just 7.6 mg of biogenic Mn(IV) oxide removed more than 98% of the 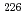 Ra from 3 L of mine water, corresponding to a distribution coefficient of 10
Ra from 3 L of mine water, corresponding to a distribution coefficient of 10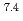 mL/g for Ra at pH 7. The obtained value was convincingly high for practical application of biogenic Mn(IV) oxide in water treatment.
mL/g for Ra at pH 7. The obtained value was convincingly high for practical application of biogenic Mn(IV) oxide in water treatment.
Fueda, Kazuki*; Komiya, Tatsuki*; Minomo, Kenta*; Horie, Kenji*; Takehara, Mami*; Yamasaki, Shinya*; Shiotsu, Hiroyuki; Onuki, Toshihiko*; Grambow, B.*; Law, G. T. W.*; et al.
Chemosphere, 328, p.138566_1 - 138566_12, 2023/07
Times Cited Count:1 Percentile:52.26(Environmental Sciences) C control rods in Fukushima Daiichi nuclear reactors during meltdown; B-Li isotopic signatures in cesium-rich microparticles
C control rods in Fukushima Daiichi nuclear reactors during meltdown; B-Li isotopic signatures in cesium-rich microparticlesFueda, Kazuki*; Takami, Ryu*; Minomo, Kenta*; Morooka, Kazuya*; Horie, Kenji*; Takehara, Mami*; Yamasaki, Shinya*; Saito, Takumi*; Shiotsu, Hiroyuki; Onuki, Toshihiko*; et al.
Journal of Hazardous Materials, 428, p.128214_1 - 128214_10, 2022/04
Times Cited Count:6 Percentile:68.71(Engineering, Environmental)Tanaka, Kazuya; Yamasaki, Shinya*
Chikyu Kagaku, 55(4), p.93 - 95, 2021/12
Ten years have passed since the Fukushima Daiichi Nuclear Power Plant (FDNPP) accident. In this special issue, we compiled review papers on the environmental behavior of the FDNPP-derived radionuclides from various research fields. This special issue shows that various research fields contributed to better understandings on the environmental behavior of the FDNPP-derived radionuclides.

 on Ni-Zn layered hydroxide salt and its application to removal of ReO
on Ni-Zn layered hydroxide salt and its application to removal of ReO
 as a surrogate of TcO
as a surrogate of TcO

Tanaka, Kazuya; Kozai, Naofumi; Yamasaki, Shinya*; Onuki, Toshihiko; Kaplan, D. I.*; Grambow, B.
Applied Clay Science, 182, p.105282_1 - 105282_8, 2019/12
Times Cited Count:16 Percentile:67.61(Chemistry, Physical)In this study, Ni-Zn layered hydroxide salt (LHS) was used for adsorption experiments of ReO
 , as a surrogate of TcO
, as a surrogate of TcO
 , in aqueous solutions with various initial Re and sodium salt concentrations. The maximum adsorption amount of Re was estimated at 127.7 mg/g (6.86
, in aqueous solutions with various initial Re and sodium salt concentrations. The maximum adsorption amount of Re was estimated at 127.7 mg/g (6.86  10
10 eq/g) by fitting adsorption isotherm of ReO
eq/g) by fitting adsorption isotherm of ReO
 to Langmuir plot. The adsorption of ReO
to Langmuir plot. The adsorption of ReO
 at neutral pH was a reversible process by anion exchange, and decreased with increasing Cl
at neutral pH was a reversible process by anion exchange, and decreased with increasing Cl , NO
, NO
 and SO
and SO
 in solution. EXAFS analysis indicated that ReO
in solution. EXAFS analysis indicated that ReO
 was adsorbed as an outer-sphere complex on Ni-Zn LHS. The Ni-Zn LHS is a more robust adsorbent for ReO
was adsorbed as an outer-sphere complex on Ni-Zn LHS. The Ni-Zn LHS is a more robust adsorbent for ReO
 than the Mg-Al LDH in terms of solution pH and tolerance to competing anions, and may be an effective alternative to the traditional and more limited method of removing aqueous TcO
than the Mg-Al LDH in terms of solution pH and tolerance to competing anions, and may be an effective alternative to the traditional and more limited method of removing aqueous TcO
 by reductive precipitation.
by reductive precipitation.
 investigation of radioactive Cs mobility around litter zone in contaminated forest using spent mushroom substrata
investigation of radioactive Cs mobility around litter zone in contaminated forest using spent mushroom substrataOnuki, Toshihiko*; Sakamoto, Fuminori; Kozai, Naofumi; Yamasaki, Shinya*; Sasaki, Yoshito; Niizato, Tadafumi
Journal of Nuclear Science and Technology, 56(9-10), p.814 - 821, 2019/09
Times Cited Count:2 Percentile:21.58(Nuclear Science & Technology)We used the spent mushroom substrata (SMSs) which are a kind of by-product after growing edible mushrooms for the  investigation of radioactive Cs mobility in litter zone in a forest of Fukushima prefecture, Japan. The powder SMS was filled in a plastic net bag of 0.35
investigation of radioactive Cs mobility in litter zone in a forest of Fukushima prefecture, Japan. The powder SMS was filled in a plastic net bag of 0.35 0.55 m, then was placed in a forest for
0.55 m, then was placed in a forest for  6 months under three kinds of different conditions without treatment (No treatment), covered with wooden box (With box), and with zeolite placed on upper position of ground surface (With zeolite). We determined the ratio of radioactivity (TF) in the SMS to that of the soil and litter beneath the SMS bags. TFs of "No treatment" and of "With zeolite" were determined between
6 months under three kinds of different conditions without treatment (No treatment), covered with wooden box (With box), and with zeolite placed on upper position of ground surface (With zeolite). We determined the ratio of radioactivity (TF) in the SMS to that of the soil and litter beneath the SMS bags. TFs of "No treatment" and of "With zeolite" were determined between  0.01 and
0.01 and  0.05 for 6 months. On the other hand, TFs of "With box" were lower by one order at 2 and 4 months than those of "No treatment" and of "With zeolite", and nearly the same values as TFs of "No treatment" and "With zeolite" at 6 months. These results clearly indicate that radioactive Cs accumulates in SMS mainly by throughfall. In addition, for a period of several months, fungi contribute to the accumulation of radioactive Cs in the litter zone, even though radioactive Cs was tightly associated with the soil.
0.05 for 6 months. On the other hand, TFs of "With box" were lower by one order at 2 and 4 months than those of "No treatment" and of "With zeolite", and nearly the same values as TFs of "No treatment" and "With zeolite" at 6 months. These results clearly indicate that radioactive Cs accumulates in SMS mainly by throughfall. In addition, for a period of several months, fungi contribute to the accumulation of radioactive Cs in the litter zone, even though radioactive Cs was tightly associated with the soil.
Sasaki, Hirokazu*; Nishikubo, Hideo*; Nishida, Shinsuke*; Yamazaki, Satoshi*; Nakasaki, Ryusuke*; Isomatsu, Takemi*; Minato, Ryuichiro*; Kinugawa, Kohei*; Imamura, Akihiro*; Otomo, Shinya*; et al.
Furukawa Denko Jiho, (138), p.2 - 10, 2019/02
no abstracts in English
Kuwahara, Akira; Aiba, Yasuaki*; Yamasaki, Shinya*; Nankawa, Takuya; Matsui, Makoto*
Journal of Analytical Atomic Spectrometry, 33(7), p.1150 - 1153, 2018/07
Times Cited Count:7 Percentile:49(Chemistry, Analytical)Although high-temperature plasma sources have been used for direct isotope analysis of solid samples, the spectral resolution of diode laser absorption spectroscopy in high-temperature plasma is limited by the Doppler broadening of atomic absorption lines. Thus, a decrease in translational temperature is necessary to enhance the spectral resolution and distinguish isotope shifts due to mass number. In this study, a supersonic plasma wind tunnel, also called an arc-jet plasma wind tunnel, was used to enhance spectral resolution drastically, and a demonstration was carried out using natural stable xenon isotopes. As a result, the temperature was found to be about 180 K and the spectral resolution was about one order of magnitude higher than that of the conventional high-temperature source. Additionally, the method proposed herein was verified by using two xenon isotopes.
Tanaka, Kazuya; Watanabe, Naoko*; Yamasaki, Shinya*; Sakaguchi, Aya*; Fan, Q.*; Takahashi, Yoshio*
Geochemical Journal, 52(2), p.173 - 185, 2018/00
Times Cited Count:9 Percentile:43.3(Geochemistry & Geophysics)We analyzed riverbed sediments collected at two sites, Yamakiya and Kuroiwa, in Fukushima after the Fukushima accident. The size distributions of K, Rb, and  Csreflected the mineralogy of sediments, where primary host minerals for these alkali elements would be biotite, K-feldspar, and clay minerals. Silt-size fractions contained high
Csreflected the mineralogy of sediments, where primary host minerals for these alkali elements would be biotite, K-feldspar, and clay minerals. Silt-size fractions contained high  Cs and
Cs and  Cs concentrations possibly due to adsorption on clay minerals. Their concentrations decreased with particle size at the Yamakiya site. In contrast, coarse and very coarse sand fractions from the Kuroiwa site showed higher
Cs concentrations possibly due to adsorption on clay minerals. Their concentrations decreased with particle size at the Yamakiya site. In contrast, coarse and very coarse sand fractions from the Kuroiwa site showed higher  Cs and
Cs and  Cs concentrations in comparison to fine - medium sand fractions. The coarse sand fractions contained many weathered biotite grains. Overall, the size distributions of
Cs concentrations in comparison to fine - medium sand fractions. The coarse sand fractions contained many weathered biotite grains. Overall, the size distributions of  Cs and
Cs and  Cs were similar in the sediments, suggesting that the Fukushima-derived radiocesium was distributed into each particle size fraction in response to the distribution of the stable Cs that was controlled by mineralogical composition.
Cs were similar in the sediments, suggesting that the Fukushima-derived radiocesium was distributed into each particle size fraction in response to the distribution of the stable Cs that was controlled by mineralogical composition.
Yamasaki, Shinya*; Tanaka, Kazuya; Kozai, Naofumi; Onuki, Toshihiko
Applied Geochemistry, 78, p.279 - 286, 2017/03
Times Cited Count:4 Percentile:18.68(Geochemistry & Geophysics)This study examined the rate constant for the U(VI) reduction process by three flavin analogues, which are redox-active biomolecules secreted from anaerobic bacteria, to elucidate their substituent group effect on the U(VI) reduction rate by electrochemical methods. The formation of the U(IV) was monitored by UV-vis spectrometry in the presence of the flavins. The rate constant for the U(VI) reduction by the flavins was determined. The apparent reduction potential of U(VI) increased about 0.2 V in the presence of the mediators, which strongly suggests that the biological electron mediator makes the U(VI) reduction possible even under more oxidative conditions.
Hosokawa, Shinya*; Kimura, Koji*; Yamasaki, Michiaki*; Kawamura, Yoshihito*; Yoshida, Koji*; Inui, Masanori*; Tsutsui, Satoshi*; Baron, A. Q. R.*; Kawakita, Yukinobu; Ito, Shinichi*
Journal of Alloys and Compounds, 695, p.426 - 432, 2017/02
Times Cited Count:3 Percentile:16.72(Chemistry, Physical)Yamasaki, Shinya*; Imoto, Jumpei*; Furuki, Genki*; Ochiai, Asumi*; Onuki, Toshihiko; Sueki, Keisuke*; Namba, Kenji*; Ewing, R. C.*; Utsunomiya, Satoshi*
Science of the Total Environment, 551-552, p.155 - 162, 2016/05
Times Cited Count:32 Percentile:70.18(Environmental Sciences)Cesium-137 ( Cs) of estuary sediment impacted by the FDNPP was measured. Increasing radioactivity was observed from surface to bottom. 90% of the
Cs) of estuary sediment impacted by the FDNPP was measured. Increasing radioactivity was observed from surface to bottom. 90% of the  Cs was strongly bound to clay minerals in the estuary sediments. These results suggest that
Cs was strongly bound to clay minerals in the estuary sediments. These results suggest that  Cs is being transported from contaminated paddy fields to the estuary.
Cs is being transported from contaminated paddy fields to the estuary.
Yu, Q.; Onuki, Toshihiko; Tanaka, Kazuya; Kozai, Naofumi; Yamasaki, Shinya*; Sakamoto, Fuminori; Tani, Yukinori*
Geochimica et Cosmochimica Acta, 174, p.1 - 12, 2016/02
Times Cited Count:18 Percentile:58.95(Geochemistry & Geophysics)Although microorganisms possess high sorption capability for lanthanides (Lns), their biological response affecting Lns migration is unclear. We investigated the effects of microbial activity on transformation of Lns by contact of Lns with Aeremonium strictum under metabolically active condition with Mn(II). A biomolecule that specifically complex to Ce(IV) was found to be released from the fungal cell, facilitating the desorption of Ce(IV) from Mn oxide. This biomolecule was not associated with any other trivalent Lns or Fe, which differed from those non-nuclide-specific organic substances released from resting cells, as reported previously.
 -ray irradiation facilities
-ray irradiation facilitiesAgematsu, Takashi; Uno, Sadanori; Hanaya, Hiroaki; Yamagata, Ryohei; Seito, Hajime; Nagao, Yuto; Yamasaki, Shota; Kawashima, Ikuo*; Yagi, Norihiko*; Takagi, Masahide*; et al.
JAEA-Review 2015-022, JAEA Takasaki Annual Report 2014, P. 173, 2016/02
The electron accelerator and the three  -ray irradiation facilities of Takasaki Advanced Radiation Research Institute were operated for various research subjects according to the operation plan in FY 2014. The number of research subjects in FY 2014 decreased at the electron accelerator with operation stop by accelerator trouble, and increased slightly at the
-ray irradiation facilities of Takasaki Advanced Radiation Research Institute were operated for various research subjects according to the operation plan in FY 2014. The number of research subjects in FY 2014 decreased at the electron accelerator with operation stop by accelerator trouble, and increased slightly at the  -ray irradiation facilities as compared with FY 2013, and research subjects were 205 and 857, respectively. Among these research subjects, 35 and 172 related to recovery from the Fukushima Daiichi Nuclear disaster, respectively, were performed at the electron accelerator and the
-ray irradiation facilities as compared with FY 2013, and research subjects were 205 and 857, respectively. Among these research subjects, 35 and 172 related to recovery from the Fukushima Daiichi Nuclear disaster, respectively, were performed at the electron accelerator and the  -ray irradiation facilities, respectively.
-ray irradiation facilities, respectively.
 Co
Co  -ray irradiation facilities
-ray irradiation facilitiesUno, Sadanori; Hanaya, Hiroaki; Yamagata, Ryohei; Seito, Hajime; Nagao, Yuto; Yamasaki, Shota; Agematsu, Takashi; Kawashima, Ikuo*; Yagi, Norihiko*; Takagi, Masahide*; et al.
JAEA-Review 2015-022, JAEA Takasaki Annual Report 2014, P. 172, 2016/02
This paper reports on the operation status of the  -ray irradiation facilities and the electron accelerator in fiscal 2014 at the Takasaki Advanced Radiation Research Institute of JAEA. The
-ray irradiation facilities and the electron accelerator in fiscal 2014 at the Takasaki Advanced Radiation Research Institute of JAEA. The  -ray irradiation facilities were operated smoothly, whereas the electron accelerator stopped for five months because of successive troubles of discharge due to deterioration of equipment over thirty-four years. The annual operation times of the
-ray irradiation facilities were operated smoothly, whereas the electron accelerator stopped for five months because of successive troubles of discharge due to deterioration of equipment over thirty-four years. The annual operation times of the  -ray irradiation facilities of the first, second and food buildings were 19,907, 13,831 and 7,293 hours, respectively, and that of the electron accelerator was 746 hours. The
-ray irradiation facilities of the first, second and food buildings were 19,907, 13,831 and 7,293 hours, respectively, and that of the electron accelerator was 746 hours. The  -ray irradiation facilities were replenished with new five sources of 2.22 PBq, and twenty old sources were removed for disposal. The electron accelerator was repaired for each trouble, however, the complete recovery has not yet been achieved.
-ray irradiation facilities were replenished with new five sources of 2.22 PBq, and twenty old sources were removed for disposal. The electron accelerator was repaired for each trouble, however, the complete recovery has not yet been achieved.
Uno, Sadanori; Hanaya, Hiroaki; Yamasaki, Shota; Yamagata, Ryohei; Seito, Hajime; Nagao, Yuto; Agematsu, Takashi; Kawashima, Ikuo*; Yagi, Norihiko*; Takagi, Masahide*; et al.
Dai-28-Kai Tandemu Kasokuki Oyobi Sono Shuhen Gijutsu No Kenkyukai Hokokushu, p.121 - 123, 2015/12
no abstracts in English
Kaneko, Makoto*; Iwata, Hajime; Shiotsu, Hiroyuki; Masaki, Shota*; Kawamoto, Yuji*; Yamasaki, Shinya*; Nakamatsu, Yuki*; Imoto, Jumpei*; Furuki, Genki*; Ochiai, Asumi*; et al.
Frontiers in Energy Research (Internet), 3, p.37_1 - 37_10, 2015/09
The mobility of the aggregates of submicron-sized sheet aluminosilicate in the surface environment is a key factor controlling the current Cs migration in Fukushima.
Onuki, Toshihiko; Jiang, M.*; Sakamoto, Fuminori; Kozai, Naofumi; Yamasaki, Shinya*; Yu, Q.; Tanaka, Kazuya; Utsunomiya, Satoshi*; Xia, X.*; Yange, K.*; et al.
Geochimica et Cosmochimica Acta, 163, p.1 - 13, 2015/08
Times Cited Count:20 Percentile:57.4(Geochemistry & Geophysics)The association of Ce(III) with the microbial cell surface and the formation of Ce phosphate nano-particles are responsible for suppressing the oxidation of Ce(III) to Ce(IV) in the mixtures.
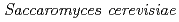
Onuki, Toshihiko; Sakamoto, Fuminori; Yamasaki, Shinya*; Kozai, Naofumi; Shiotsu, Hiroyuki; Utsunomiya, Satoshi*; Watanabe, Naoko*; Kozaki, Tamotsu*
Journal of Environmental Radioactivity, 144, p.127 - 133, 2015/06
Times Cited Count:9 Percentile:27.12(Environmental Sciences)The accumulation of Cs by unicellular fungus of 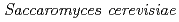 in the presence of minerals has been studied to elucidate the role of microorganisms in the migration of radioactive Cs in the environment. In the presence of minerals in the agar medium, the radioactivity in the yeast cells was in the order of mica
in the presence of minerals has been studied to elucidate the role of microorganisms in the migration of radioactive Cs in the environment. In the presence of minerals in the agar medium, the radioactivity in the yeast cells was in the order of mica  smectite, illite
smectite, illite 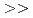 vermiculite, phlogopite, zeolite. This order is inversely correlated to the ratio of the concentration of radioactive Cs between the minerals and the medium solution. These results strongly suggest that the yeast accumulates radioactive Cs competitively with minerals.
vermiculite, phlogopite, zeolite. This order is inversely correlated to the ratio of the concentration of radioactive Cs between the minerals and the medium solution. These results strongly suggest that the yeast accumulates radioactive Cs competitively with minerals.
Kozai, Naofumi; Yamasaki, Shinya; Onuki, Toshihiko
Journal of Radioanalytical and Nuclear Chemistry, 299(3), p.1571 - 1579, 2014/03
Times Cited Count:4 Percentile:30.92(Chemistry, Analytical)To elucidate the sorption behavior of americium(III) on bentonite simplified desorption experiments were applied to the solid phases collected after the sorption experiments. The sorption-desorption behavior was examined in the final pH range from 2 to 8. The desorption experiments revealed that most of the Am was sorbed on the montmorillonite moiety of the bentonite. The sorption of Am on montmorillonite was divided into two types: one was the "exchangeable" sorption, in which the sorbed Am was desorbed with a 1M KCl aqueous solution, and the rest was the "unexchangeable" sorption. The exchangeable sorption was ion exchange of mostly Am . The unexchangeable sorption was the strong sorption of Am hydroxides. An accessory iron mineral, pyrite, might be involved in the Am sorption on bentonite at neutral pH.
. The unexchangeable sorption was the strong sorption of Am hydroxides. An accessory iron mineral, pyrite, might be involved in the Am sorption on bentonite at neutral pH.