Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Li, F.*; Tang, X.*; Fei, Y.*; Zhang, J.*; Liu, J.*; Lang, P.*; Che, G.*; Zhao, Z.*; Zheng, Y.*; Fang, Y.*; et al.
Journal of the American Chemical Society, 147(17), p.14054 - 14059, 2025/04
Times Cited Count:1 Percentile:0.00(Chemistry, Multidisciplinary)We synthesized a crystalline graphane nanoribbon (GANR) via pressure-induced polymerization of 2,2'-bipyrazine (BPZ). By performing Rietveld refinement of in situ neutron diffraction data, nuclear magnetic resonance spectroscopy, infrared spectra, and theoretical calculation, we found that BPZ experienced Diels-Alder polymerization between the 
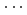
 stacked aromatic rings, and formed extended boat-GANR structures with exceptional long-range order. The unreacted -C=N- groups bridge the two ends of the boat, and ready for further functionalization. The GANR has a bandgap of 2.25 eV, with booming photoelectric response (
stacked aromatic rings, and formed extended boat-GANR structures with exceptional long-range order. The unreacted -C=N- groups bridge the two ends of the boat, and ready for further functionalization. The GANR has a bandgap of 2.25 eV, with booming photoelectric response (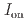 /
/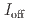 =18.8). Our work highlights that the high-pressure topochemical polymerization is a promising method for the precise synthesis of graphane with specific structure and desired properties.
=18.8). Our work highlights that the high-pressure topochemical polymerization is a promising method for the precise synthesis of graphane with specific structure and desired properties.
Rajeev, H. S.*; Hu, X.*; Chen, W.-L.*; Zhang, D.*; Chen, T.*; Kofu, Maiko*; Kajimoto, Ryoichi; Nakamura, Mitsutaka; Chen, A. Z.*; Johnson, G. C.*; et al.
Journal of the Physical Society of Japan, 94(3), p.034602_1 - 034602_14, 2025/03
Times Cited Count:0 Percentile:0.00(Physics, Multidisciplinary)Liu, P.-F.*; Li, X.*; Li, J.*; Zhu, J.*; Tong, Z.*; Kofu, Maiko*; Nirei, Masami; Xu, J.*; Yin, W.*; Wang, F.*; et al.
National Science Review, 11(12), p.nwae216_1 - nwae216_10, 2024/12
Times Cited Count:13 Percentile:94.32(Multidisciplinary Sciences)Ying, H.*; Yang, X.*; He, H.*; Yan, A.*; An, K.*; Ke, Y.*; Wu, Z.*; Tang, S.*; Zhang, Z.*; Dong, H.*; et al.
Scripta Materialia, 250, p.116181_1 - 116181_7, 2024/09
Times Cited Count:2 Percentile:36.18(Nanoscience & Nanotechnology)Igarashi, Junya*; Ninomiya, Kazuhiko*; Zheng, J.*; Zhang, Z. J.*; Fukuda, Miho*; Aono, Tatsuo*; Minowa, Haruka*; Yoshikawa, Hideki*; Sueki, Keisuke*; Satou, Yukihiko; et al.
Environmental Science & Technology, 58(33), p.14823 - 14830, 2024/08
Times Cited Count:0 Percentile:0.00(Engineering, Environmental)Zhang, Z.*; Hattori, Takanori; Song, R.*; Yu, D.*; Mole, R.*; Chen, J.*; He, L.*; Zhang, Z.*; Li, B.*
Journal of Applied Physics, 136(3), p.035105_1 - 035105_8, 2024/07
Times Cited Count:2 Percentile:35.22(Physics, Applied)Solid-state refrigeration using barocaloric materials is environmentally friendly and highly efficient, making it a subject of global interest over the past decade. Here, we report giant barocaloric effects in sodium hexafluorophosphate (NaPF ) and sodium hexafluoroarsenate (NaAsF
) and sodium hexafluoroarsenate (NaAsF ) that both undergo a cubic-to-rhombohedral phase transition near room temperature. We have determined that the low-temperature phase structure of NaPF
) that both undergo a cubic-to-rhombohedral phase transition near room temperature. We have determined that the low-temperature phase structure of NaPF is a rhombohedral structure with space group R
is a rhombohedral structure with space group R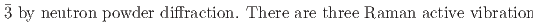 and NaAsF
and NaAsF , i.e., F
, i.e., F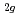 , E
, E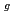 , and A
, and A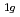 . The phase transition temperature varies with pressure at a rate of dT
. The phase transition temperature varies with pressure at a rate of dT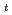 /dP = 250 and 310 K/GPa for NaPF
/dP = 250 and 310 K/GPa for NaPF and NaAsF
and NaAsF . The pressure-induced entropy changes of NaPF
. The pressure-induced entropy changes of NaPF and NaAsF
and NaAsF are determined to be around 45.2 and 35.6J kg
are determined to be around 45.2 and 35.6J kg K
K , respectively. The saturation driving pressure is about 40 MPa. The pressure-dependent neutron powder diffraction suggests that the barocaloric effects are related to the pressure-induced cubic-to-rhombohedral phase transitions.
, respectively. The saturation driving pressure is about 40 MPa. The pressure-dependent neutron powder diffraction suggests that the barocaloric effects are related to the pressure-induced cubic-to-rhombohedral phase transitions.
Zeng, Z.*; Zhou, C.*; Zhou, H.*; Han, L.*; Chi, R.*; Li, K.*; Kofu, Maiko; Nakajima, Kenji; Wei, Y.*; Zhang, W.*; et al.
Nature Physics, 20(7), p.1097 - 1102, 2024/07
Times Cited Count:12 Percentile:94.31(Physics, Multidisciplinary)Wang, S.*; Wang, J.*; Zhang, S.*; Wei, D.*; Chen, Y.*; Rong, X.*; Gong, W.; Harjo, S.; Liu, X.*; Jiao, Z.*; et al.
Journal of Materials Science & Technology, 185, p.245 - 258, 2024/06
Times Cited Count:17 Percentile:98.00(Materials Science, Multidisciplinary)
Liao, J.*; Huang, Z.*; Shangguan, Y.*; Zhang, B.*; Cheng, S.*; Xu, H.*; Kajimoto, Ryoichi; Kamazawa, Kazuya*; Bao, S.*; Wen, J.*
Physical Review B, 109(22), p.224411_1 - 224411_10, 2024/06
Times Cited Count:0 Percentile:0.00(Materials Science, Multidisciplinary)Baccou, J.*; Glantz, T.*; Ghione, A.*; Sargentini, L.*; Fillion, P.*; Damblin, G.*; Sueur, R.*; Iooss, B.*; Fang, J.*; Liu, J.*; et al.
Nuclear Engineering and Design, 421, p.113035_1 - 113035_16, 2024/05
Times Cited Count:6 Percentile:96.49(Nuclear Science & Technology)Guo, B.*; Chen, H.*; Chong, Y.*; Mao, W.; Harjo, S.; Gong, W.; Zhang, Z.*; Jonas, J. J.*; Tsuji, Nobuhiro*
Acta Materialia, 268, p.119780_1 - 119780_11, 2024/04
Times Cited Count:10 Percentile:94.16(Materials Science, Multidisciplinary)Li, X.*; Zhu, R.*; Xin, J.*; Luo, M.*; Shang, S.-L.*; Liu, Z.-K.*; Yin, C.*; Funakoshi, Kenichi*; Dippenaar, R. J.*; Higo, Yuji*; et al.
CALPHAD; Computer Coupling of Phase Diagrams and Thermochemistry, 84, p.102641_1 - 102641_6, 2024/03
Times Cited Count:0 Percentile:0.00(Thermodynamics) FCC hierarchical martensite transformation under dynamic impact in FeMnAlNiTi alloy
FCC hierarchical martensite transformation under dynamic impact in FeMnAlNiTi alloyLi, C.*; Fang, W.*; Yu, H. Y.*; Peng, T.*; Yao, Z. T.*; Liu, W. G.*; Zhang, X.*; Xu, P. G.; Yin, F.*
Materials Science & Engineering A, 892, p.146096_1 - 146096_11, 2024/02
Times Cited Count:6 Percentile:79.11(Nanoscience & Nanotechnology)Matsushima, Yu*; Zhang, Z.*; Ohashi, Yuri*; Hatakeyama, Tsunagu*; Xiao, G.*; Funato, Takumi*; Matsuo, Mamoru; Kaiju, Hideo*
Applied Physics Letters, 124(2), p.022404_1 - 022404_7, 2024/01
Times Cited Count:3 Percentile:70.50(Physics, Applied) neutron diffraction
neutron diffractionZhou, Y.*; Song, W.*; Zhang, F.*; Wu, Y.*; Lei, Z.*; Jiao, M.*; Zhang, X.*; Dong, J.*; Zhang, Y.*; Yang, M.*; et al.
Journal of Alloys and Compounds, 971, p.172635_1 - 172635_7, 2024/01
Times Cited Count:3 Percentile:23.52(Chemistry, Physical)Zhang, A.*; Deng, K.*; Sheng, J.*; Liu, P.*; Kumar, S.*; Shimada, Kenya*; Jiang, Z.*; Liu, Z.*; Shen, D.*; Li, J.*; et al.
Chinese Physics Letters, 40(12), p.126101_1 - 126101_8, 2023/12
Times Cited Count:11 Percentile:85.96(Physics, Multidisciplinary)Zhang, Z.*; Gong, W.; Kawakita, Yukinobu; 9 of others*
Physical Review Materials (Internet), 7(12), p.125402_1 - 125402_9, 2023/12
Times Cited Count:2 Percentile:20.33(Materials Science, Multidisciplinary)Lloveras, P.*; Zhang, Z.*; Zeng, M.*; Barrio, M.*; Kawakita, Yukinobu; Yu, D.*; Lin, S.*; Li, K.*; Moya, X.*; Tamarit, J.-L.*; et al.
Barocaloric Effects in the Solid State; Materials and methods, p.7_1 - 7_30, 2023/10
Times Cited Count:232 Percentile:99.37(Multidisciplinary Sciences)As Chapter 1 of the ebook entitled as "Barocaloric Effects in the Solid State", various plastic crystals (PC) showing colossal barocaloric (BC) effect are introduced. A method to determine the BC response in PCs, thermodynamic origin of BC effects, spectroscopic insights from quasi-elastic neutron scattering and application of PCs are explained.
Bao, S.*; Gu, Z.-L.*; Shangguan, Y.*; Huang, Z.*; Liao, J.*; Zhao, X.*; Zhang, B.*; Dong, Z.-Y.*; Wang, W.*; Kajimoto, Ryoichi; et al.
Nature Communications (Internet), 14, p.6093_1 - 6093_9, 2023/09
Times Cited Count:21 Percentile:92.84(Multidisciplinary Sciences)Shangguan, Y.*; Bao, S.*; Dong, Z.-Y.*; Xi, N.*; Gao, Y.-P.*; Ma, Z.*; Wang, W.*; Qi, Z.*; Zhang, S.*; Huang, Z.*; et al.
Nature Physics, 19(12), p.1883 - 1889, 2023/09
Times Cited Count:19 Percentile:93.50(Physics, Multidisciplinary)