Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yamashita, Keishiro*; Komatsu, Kazuki*; Hattori, Takanori; Machida, Shinichi*; Kagi, Hiroyuki*
Acta Crystallographica Section B; Structural Science, Crystal Engineering and Materials (Internet), 80(6), p.695 - 705, 2024/12
Times Cited Count:0 Percentile:0.00(Chemistry, Multidisciplinary)So far, the odd hydration number has been missing in the water-rich magnesium chloride hydrate series (MgCl

 H
H O). In this study, we have identified magnesium chloride heptahydrate, MgCl
O). In this study, we have identified magnesium chloride heptahydrate, MgCl
 7H
7H O (or MgCl
O (or MgCl
 7D
7D O) which forms at high pressures and high temperatures of above 2 GPa and above 300 K, respectively. Its structure has been determined by a combination of
O) which forms at high pressures and high temperatures of above 2 GPa and above 300 K, respectively. Its structure has been determined by a combination of 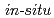 single crystal X-ray diffraction at 2.5 GPa and 298 K and powder neutron diffraction at 3.1 GPa and 300 K. The results showed an orientational disorder of water molecules, which was also examined by the density-functional-theory calculations. The disorder involves the reconnection of hydrogen bonds, which differs from those in water ice phases and known disordered salt hydrates. The shrinkage by compression occurs mainly in one direction. In the plane perpendicular to this most compressible direction, oxygen and chlorine atoms are in a hexagonal-like arrangement.
single crystal X-ray diffraction at 2.5 GPa and 298 K and powder neutron diffraction at 3.1 GPa and 300 K. The results showed an orientational disorder of water molecules, which was also examined by the density-functional-theory calculations. The disorder involves the reconnection of hydrogen bonds, which differs from those in water ice phases and known disordered salt hydrates. The shrinkage by compression occurs mainly in one direction. In the plane perpendicular to this most compressible direction, oxygen and chlorine atoms are in a hexagonal-like arrangement.
Yamashita, Keishiro*; Nakayama, Kazuya*; Komatsu, Kazuki*; Ohara, Takashi; Munakata, Koji*; Hattori, Takanori; Sano, Asami; Kagi, Hiroyuki*
Acta Crystallographica Section B; Structural Science, Crystal Engineering and Materials (Internet), 79(5), p.414 - 426, 2023/10
Times Cited Count:2 Percentile:41.77(Chemistry, Multidisciplinary)The structure of a recently-found hyperhydrated form of sodium chloride, NaCl 13H(D)
13H(D) O, has been determined by
O, has been determined by  single-crystal neutron diffraction at 1.7 GPa and 298 K. It has large hydrogen-bond networks and some water molecules have distorted bonding features such as bifurcated hydrogen bonds and five-coordinated water molecules. The hydrogen-bond network has similarities to ice VI in terms of network topology and disordered hydrogen bonds. Assuming the equivalence of network components connected by pseudo symmetries, the overall network structure of this hydrate can be expressed by breaking it down into smaller structural units which correspond to the ice VI network structure. This hydrogen-bond network contains orientational disorder of water molecules in contrast to the known salt hydrates. Here, we present an example for further insights into a hydrogen-bond network containing ionic species.
single-crystal neutron diffraction at 1.7 GPa and 298 K. It has large hydrogen-bond networks and some water molecules have distorted bonding features such as bifurcated hydrogen bonds and five-coordinated water molecules. The hydrogen-bond network has similarities to ice VI in terms of network topology and disordered hydrogen bonds. Assuming the equivalence of network components connected by pseudo symmetries, the overall network structure of this hydrate can be expressed by breaking it down into smaller structural units which correspond to the ice VI network structure. This hydrogen-bond network contains orientational disorder of water molecules in contrast to the known salt hydrates. Here, we present an example for further insights into a hydrogen-bond network containing ionic species.
Kawasaki, Takuro; Takahashi, Miwako*; Kiyanagi, Ryoji; Ohara, Takashi
Acta Crystallographica Section C; Structural Chemistry (Internet), 78(12), p.743 - 748, 2022/12
Times Cited Count:0 Percentile:0.00(Chemistry, Multidisciplinary)Terasawa, Yukana*; Ohara, Takashi; Sato, Sota*; Yoshida, Satoshi*; Asahi, Toru*
Acta Crystallographica Section E; Crystallographic Communications (Internet), 78(3), p.306 - 312, 2022/03
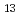 } polyoxo cluster U
} polyoxo cluster U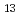 O
O Cl
Cl (MeO)
(MeO)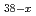 (x = 2.3, MeO = methoxide)
(x = 2.3, MeO = methoxide)Fichter, S.*; Radoske, T.*; Ikeda, Atsushi
Acta Crystallographica Section E; Crystallographic Communications (Internet), 77(8), p.847 - 852, 2021/08
 '(O'3)-type sodium cobalt oxide Na
'(O'3)-type sodium cobalt oxide Na CoO
CoO (
(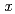
 0.78)
0.78)Miyazaki, Yuzuru*; Igawa, Naoki; Yubuta, Kunio*
Acta Crystallographica Section B; Structural Science, Crystal Engineering and Materials (Internet), 77(3), p.371 - 377, 2021/06
Times Cited Count:2 Percentile:24.24(Chemistry, Multidisciplinary)Crystal structure of  '(O'3)-type layered sodium cobalt oxide Na
'(O'3)-type layered sodium cobalt oxide Na CoO
CoO (
(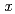
 0.78) was analyzed using the (3+1)-dimensional superspace approach to the neutron diffraction data. The crystal structure is described based on the superspace group
0.78) was analyzed using the (3+1)-dimensional superspace approach to the neutron diffraction data. The crystal structure is described based on the superspace group 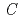 2 =
2 = 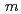 (
(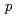 0
0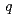 )00, wherein the positions of Na atoms are most significantly modulated in the monoclinic
)00, wherein the positions of Na atoms are most significantly modulated in the monoclinic 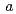 -direction to form an ordered arrangement. Such a positional modulation causes a quasi-periodic shift of Na atoms from the centers of the NaO
-direction to form an ordered arrangement. Such a positional modulation causes a quasi-periodic shift of Na atoms from the centers of the NaO polyhedra between undulated CoO
polyhedra between undulated CoO sheets, changing the form of the NaO
sheets, changing the form of the NaO polyhedron from an octahedral coordination (O) to a trigonal prismatic one (P). Where the Na atoms are most significantly shifted, the neighboring Na atoms are located at almost touching distances and yielding Na-deficient sites. As a result, a typical sequence of NaO
polyhedron from an octahedral coordination (O) to a trigonal prismatic one (P). Where the Na atoms are most significantly shifted, the neighboring Na atoms are located at almost touching distances and yielding Na-deficient sites. As a result, a typical sequence of NaO octahedra with -O-O-O-P-
octahedra with -O-O-O-P-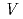
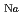 -P- along the a-axis is realized.
-P- along the a-axis is realized.
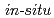 X-ray and neutron diffraction methods
X-ray and neutron diffraction methodsYamashita, Keishiro*; Komatsu, Kazuki*; Hattori, Takanori; Machida, Shinichi*; Kagi, Hiroyuki*
Acta Crystallographica Section C; Structural Chemistry (Internet), 75(12), p.1605 - 1612, 2019/12
Times Cited Count:11 Percentile:68.40(Chemistry, Multidisciplinary)A crystal structure of a high-pressure phase of magnesium chloride hexahydrate (MgCl
 6H
6H O-II) and its deuterated counterpart (MgCl
O-II) and its deuterated counterpart (MgCl
 6D
6D O-II) have been identified for the first time by in-situ single-crystal X-ray and powder neutron diffraction. The crystal structure was analyzed by the Rietveld method for the neutron diffraction pattern based on the initial structure determined by single-crystal X-ray diffraction. This high-pressure phase has a similar framework to that in the known ambient-pressure phase, but exhibits some structural changes with symmetry reduction caused by a subtle modification in the hydrogen-bond network around the Mg(H
O-II) have been identified for the first time by in-situ single-crystal X-ray and powder neutron diffraction. The crystal structure was analyzed by the Rietveld method for the neutron diffraction pattern based on the initial structure determined by single-crystal X-ray diffraction. This high-pressure phase has a similar framework to that in the known ambient-pressure phase, but exhibits some structural changes with symmetry reduction caused by a subtle modification in the hydrogen-bond network around the Mg(H O)
O) octahedra. These structural features reflect the strain in the high-pressure phases of MgCl
octahedra. These structural features reflect the strain in the high-pressure phases of MgCl hydrates.
hydrates.
 baddeleyite involving softenings of bulk modulus and atom vibrations
baddeleyite involving softenings of bulk modulus and atom vibrationsFukui, Hiroshi*; Fujimoto, Manato*; Akahama, Yuichi*; Sano, Asami; Hattori, Takanori
Acta Crystallographica Section B; Structural Science, Crystal Engineering and Materials (Internet), 75(4), p.742 - 749, 2019/08
Times Cited Count:14 Percentile:76.15(Chemistry, Multidisciplinary)Monoclinic ZrO baddeleyite exhibits anomalous softenings of bulk modulus and atom vibrations with compression. We have investigated the pressure evolution of the structure by neutron powder diffraction combined with ab-initio calculations. The present results showed that the anomalous pressure response of the bulk modulus is related not to the change in the bonding characters but to the deformation of an oxygen sublattice, especially one of layers made of oxygens in the crystallographic
baddeleyite exhibits anomalous softenings of bulk modulus and atom vibrations with compression. We have investigated the pressure evolution of the structure by neutron powder diffraction combined with ab-initio calculations. The present results showed that the anomalous pressure response of the bulk modulus is related not to the change in the bonding characters but to the deformation of an oxygen sublattice, especially one of layers made of oxygens in the crystallographic 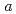 * plane. The layer consists of two parallelograms; one is rotating with little distortion and the other is being distorted with increasing pressure. This deformation of this layer makes one of Zr-O distances long, resulting in the softening of some atom vibrational modes.
* plane. The layer consists of two parallelograms; one is rotating with little distortion and the other is being distorted with increasing pressure. This deformation of this layer makes one of Zr-O distances long, resulting in the softening of some atom vibrational modes.
Yano, Naomine*; Yamada, Taro*; Hosoya, Takaaki*; Ohara, Takashi; Tanaka, Ichiro*; Niimura, Nobuo*; Kusaka, Katsuhiro*
Acta Crystallographica Section D; Structural Biology (Internet), 74(11), p.1041 - 1052, 2018/11
Times Cited Count:16 Percentile:73.72(Biochemical Research Methods)Chatake, Toshiyuki*; Fujiwara, Satoru
Acta Crystallographica Section D; Structural Biology (Internet), 72(1), p.71 - 82, 2016/01
Times Cited Count:6 Percentile:40.33(Biochemical Research Methods)
Hirano, Yu; Kimura, Shigenobu*; Tamada, Taro
Acta Crystallographica Section D, 71(7), p.1572 - 1581, 2015/07
Times Cited Count:3 Percentile:27.99(Biochemical Research Methods)Mammalian microsomal cytochrome 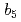 has multiple electron-transfer partners that function in various electron-transfer reactions. Four crystal structures of the solubilized haem-binding domain of cytochrome
has multiple electron-transfer partners that function in various electron-transfer reactions. Four crystal structures of the solubilized haem-binding domain of cytochrome 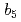 from porcine liver were determined at sub-angstrom resolution (0.76-0.95
from porcine liver were determined at sub-angstrom resolution (0.76-0.95  ) in two crystal forms for both the oxidized and reduced states. The high-resolution structures clearly displayed the electron density of H atoms in some amino-acid residues. Unrestrained refinement of bond lengths revealed that the protonation states of the haem propionate group may be involved in regulation of the haem redox properties. The haem Fe coordination geometry did not show significant differences between the oxidized and reduced structures. However, structural differences between the oxidized and reduced states were observed in the hydrogen-bond network around the axial ligand His68. The hydrogen-bond network could be involved in regulating the redox states of the haem group.
) in two crystal forms for both the oxidized and reduced states. The high-resolution structures clearly displayed the electron density of H atoms in some amino-acid residues. Unrestrained refinement of bond lengths revealed that the protonation states of the haem propionate group may be involved in regulation of the haem redox properties. The haem Fe coordination geometry did not show significant differences between the oxidized and reduced structures. However, structural differences between the oxidized and reduced states were observed in the hydrogen-bond network around the axial ligand His68. The hydrogen-bond network could be involved in regulating the redox states of the haem group.
 -lactamase from the moderate halophile
-lactamase from the moderate halophile  sp.560 and the discovery of a Cs
sp.560 and the discovery of a Cs -selective binding site
-selective binding siteArai, Shigeki; Yonezawa, Yasushi*; Okazaki, Nobuo*; Matsumoto, Fumiko*; Shibazaki, Chie; Shimizu, Rumi; Yamada, Mitsugu*; Adachi, Motoyasu; Tamada, Taro; Kawamoto, Masahide*; et al.
Acta Crystallographica Section D, 71(3), p.541 - 554, 2015/03
Times Cited Count:8 Percentile:52.42(Biochemical Research Methods)The crystal structure of halophilic  -lactamase from
-lactamase from  sp.560 (HaBLA) was determined using X-ray crystallography. Moreover, the locations of bound Sr
sp.560 (HaBLA) was determined using X-ray crystallography. Moreover, the locations of bound Sr and Cs
and Cs ions were identified by anomalous X-ray diffraction. The location of one Cs
ions were identified by anomalous X-ray diffraction. The location of one Cs specific binding site was identified on HaBLA even in the presence of 9-fold molar excess of Na
specific binding site was identified on HaBLA even in the presence of 9-fold molar excess of Na (90 mM Na
(90 mM Na /10 mM Cs
/10 mM Cs ). This Cs
). This Cs binding site is formed by two main-chain O atoms and an aromatic ring of a side chain of Trp. An aromatic ring of Trp interacts with Cs
binding site is formed by two main-chain O atoms and an aromatic ring of a side chain of Trp. An aromatic ring of Trp interacts with Cs by the cation-
by the cation- interaction. The observation of a selective and high-affinity Cs
interaction. The observation of a selective and high-affinity Cs binding site provides important information that is useful for designing artificial Cs
binding site provides important information that is useful for designing artificial Cs binding sites useful in bioremediation of radioactive isotopes.
binding sites useful in bioremediation of radioactive isotopes.
Komatsu, Kazuki*; Shinozaki, Ayako*; Machida, Shinichi*; Matsubayashi, Takuto*; Watanabe, Mao*; Kagi, Hiroyuki*; Sano, Asami; Hattori, Takanori
Acta Crystallographica Section B; Structural Science, Crystal Engineering and Materials (Internet), 71(1), p.74 - 80, 2015/02
Times Cited Count:21 Percentile:80.09(Chemistry, Multidisciplinary)Magnesium dichloride decahydrate (MgCl 10H
10H O) and its deuterated counterpart (MgCl
O) and its deuterated counterpart (MgCl 10D
10D O) are identified for the first time by in-situ powder synchrotron X-ray and spallation neutron diffraction. These substances are crystallized from a previously unidentified nanocrystalline compound, which originates from an amorphous state at low temperature. A combination of a recently developed autoindexing procedure and the charge-flipping method reveals that the crystal structure of MgCl 10H
O) are identified for the first time by in-situ powder synchrotron X-ray and spallation neutron diffraction. These substances are crystallized from a previously unidentified nanocrystalline compound, which originates from an amorphous state at low temperature. A combination of a recently developed autoindexing procedure and the charge-flipping method reveals that the crystal structure of MgCl 10H O consists of an ABCABC... sequence of Mg(H
O consists of an ABCABC... sequence of Mg(H O)
O) octahedra. The Cl
octahedra. The Cl anions and remaining water molecules unconnected to the Mg
anions and remaining water molecules unconnected to the Mg cations bind the octahedra, similar to other water-rich magnesium dichloride hydrates. The D positions in MgCl
cations bind the octahedra, similar to other water-rich magnesium dichloride hydrates. The D positions in MgCl 10D
10D O, determined by the difference Fourier methods using the neutron powder diffraction patterns at 2.5 GPa, show the features such as bifurcated hydrogen bonds and tetrahedrally coordinated O atoms.
O, determined by the difference Fourier methods using the neutron powder diffraction patterns at 2.5 GPa, show the features such as bifurcated hydrogen bonds and tetrahedrally coordinated O atoms.
 sp.593
sp.593Arai, Shigeki; Yonezawa, Yasushi*; Ishibashi, Matsujiro*; Matsumoto, Fumiko*; Adachi, Motoyasu; Tamada, Taro; Tokunaga, Hiroko*; Blaber, M.; Tokunaga, Masao*; Kuroki, Ryota
Acta Crystallographica Section D, 70(3), p.811 - 820, 2014/03
Times Cited Count:13 Percentile:63.41(Biochemical Research Methods)In order to clarify the structural basis of halophilic characteristics of an alkaline phosphatase derived from the moderate halophile  sp.593 (HaAP), the tertiary structure of HaAP was determined to 2.1
sp.593 (HaAP), the tertiary structure of HaAP was determined to 2.1 resolution by X-ray crystallography. Structural properties of surface negative charge and core hydrophobicity are shown to be intermediate between halophile and non-halophile characteristics, and may explain the unique functional adaptation to a wide-range of salt concentration.
resolution by X-ray crystallography. Structural properties of surface negative charge and core hydrophobicity are shown to be intermediate between halophile and non-halophile characteristics, and may explain the unique functional adaptation to a wide-range of salt concentration.
 ; Assignment of bound diatomic molecules as O
; Assignment of bound diatomic molecules as O
Murakawa, Takeshi*; Hayashi, Hideyuki*; Sunami, Tomoko; Kurihara, Kazuo; Tamada, Taro; Kuroki, Ryota; Suzuki, Mamoru*; Tanizawa, Katsuyuki*; Okajima, Toshihide*
Acta Crystallographica Section D, 69(12), p.2483 - 2494, 2013/12
Times Cited Count:14 Percentile:67.17(Biochemical Research Methods)The crystal structure of a Cu amine oxidase from 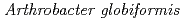 was determined at 1.08
was determined at 1.08  resolution with the use of low-molecular-weight polyethylene glycol (LMW PEG; average molecular weight
resolution with the use of low-molecular-weight polyethylene glycol (LMW PEG; average molecular weight  200) as a cryoprotectant. The final crystallographic
200) as a cryoprotectant. The final crystallographic 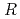 -factor and
-factor and 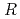
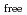 value are 13.0% and 15.0%, respectively. Several molecules of LMW PEG were found to occupy cavities in the protein interior including the active site, which resulted in the marked reduction of the overall
value are 13.0% and 15.0%, respectively. Several molecules of LMW PEG were found to occupy cavities in the protein interior including the active site, which resulted in the marked reduction of the overall 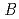 factor and consequently led to a sub-atomic resolution structure for a relatively large protein with a monomer molecular weight of
factor and consequently led to a sub-atomic resolution structure for a relatively large protein with a monomer molecular weight of  70,000. About 40% of all the presumed hydrogen atoms were observed as clear electron densities in the
70,000. About 40% of all the presumed hydrogen atoms were observed as clear electron densities in the 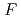
 -
- 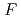
 difference map. Multiple minor conformers were also identified for many residues. Anisotropic displacement fluctuations were evaluated in the active site that contains a post-translationally derived quinone cofactor and a Cu atom. Furthermore, diatomic molecules, most likely molecular oxygen, are bound to the protein, one of which is located in the region that has been previously proposed as an entry route for the substrate dioxygen from the central cavity of the dimer interface to the active site.
difference map. Multiple minor conformers were also identified for many residues. Anisotropic displacement fluctuations were evaluated in the active site that contains a post-translationally derived quinone cofactor and a Cu atom. Furthermore, diatomic molecules, most likely molecular oxygen, are bound to the protein, one of which is located in the region that has been previously proposed as an entry route for the substrate dioxygen from the central cavity of the dimer interface to the active site.
Kanao, Tadayoshi*; Kosaka, Megumi*; Yoshida, Kyoya*; Nakayama, Hisayuki*; Tamada, Taro; Kuroki, Ryota; Yamada, Hidenori; Takada, Jun*; Kamimura, Kazuo*
Acta Crystallographica Section F, 69(6), p.692 - 694, 2013/06
Times Cited Count:10 Percentile:58.18(Biochemical Research Methods)Tetrathionate hydrolase (4THase) from the iron- and sulfur-oxidizing bacterium  catalyses the disproportionate hydrolysis of tetrathionate to elemental sulfur, thiosulfate and sulfate. The gene encoding 4THase (
catalyses the disproportionate hydrolysis of tetrathionate to elemental sulfur, thiosulfate and sulfate. The gene encoding 4THase ( ) was expressed as inclusion bodies in recombinant
) was expressed as inclusion bodies in recombinant  . Recombinant
. Recombinant 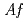 -Tth was activated by refolding under acidic conditions and was then purified to homogeneity. The recombinant protein was crystallized in 20 m
-Tth was activated by refolding under acidic conditions and was then purified to homogeneity. The recombinant protein was crystallized in 20 m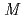 glycine buffer pH 10 containing 50 m
glycine buffer pH 10 containing 50 m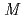 sodium chloride and 33%(
sodium chloride and 33%(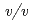 ) PEG 1000 using the hanging-drop vapour-diffusion method. The crystal was a hexagonal cylinder with dimensions of 0.2
) PEG 1000 using the hanging-drop vapour-diffusion method. The crystal was a hexagonal cylinder with dimensions of 0.2  0.05
0.05  0.05 mm. X-ray crystallographic analysis showed that the crystal diffracted to 2.15
0.05 mm. X-ray crystallographic analysis showed that the crystal diffracted to 2.15  resolution and belongs to space group
resolution and belongs to space group 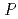 3
3 or
or 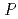 3
3 , with unit-cell parameters
, with unit-cell parameters 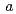 =
= 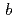 = 92.1,
= 92.1,  = 232.6
= 232.6  .
.
Tokuhisa, Atsushi*; Taka, Junichiro*; Kono, Hidetoshi; Go, Nobuhiro*
Acta Crystallographica Section A, 68(3), p.366 - 381, 2012/05
Times Cited Count:20 Percentile:80.73(Chemistry, Multidisciplinary) HB8
HB8Okazaki, Nobuo; Adachi, Motoyasu; Tamada, Taro; Kurihara, Kazuo; Oga, Takushi*; Kamiya, Nobuo*; Kuramitsu, Seiki*; Kuroki, Ryota
Acta Crystallographica Section F, 68(1), p.49 - 52, 2012/01
Times Cited Count:2 Percentile:31.75(Biochemical Research Methods)Fukuda, Yota*; Tamada, Taro; Takami, Hideto*; Suzuki, Shinichiro*; Inoue, Tsuyoshi*; Nojiri, Masaki
Acta Crystallographica Section F, 67(6), p.692 - 695, 2011/06
Times Cited Count:10 Percentile:66.87(Biochemical Research Methods) sp. A-471
sp. A-471Okazaki, Nobuo; Arimori, Takao; Nakazawa, Masami*; Miyatake, Kazutaka*; Ueda, Mitsuhiro*; Tamada, Taro
Acta Crystallographica Section F, 67(4), p.494 - 497, 2011/04
Times Cited Count:3 Percentile:40.30(Biochemical Research Methods)