Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
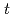 = 0.01-600
= 0.01-600 C,
C,  = 1-3000 bars,
= 1-3000 bars, 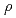
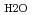 = 0.35-1.1 g cm
= 0.35-1.1 g cm , and
, and 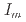 = 0
= 0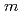
Walker, C. S.*; Arthur, R. C.*; Anraku, Sohtaro; Sasamoto, Hiroshi; Mihara, Morihiro
Applied Geochemistry, 175, p.106086_1 - 106086_17, 2024/11
Times Cited Count:0 Percentile:0.00(Geochemistry & Geophysics)The thermodynamic properties and revised Helgeson-Kirkham-Flowers equation of state (r-H-K-F EoS) parameters of the hydrated (Si(OH) (aq), SiO(OH)
(aq), SiO(OH)
 and SiO
and SiO (OH)
(OH)
 ) and fictive dehydrated (SiO
) and fictive dehydrated (SiO (aq), HSiO
(aq), HSiO
 and SiO
and SiO
 ) monomeric silicon species are used extensively to describe the pH, composition, temperature, and pressure dependence of formation/breakdown reactions of all silicon-bearing compounds globally. Experimental log10 equilbrium constant, K values describing the formation reactions of the hydrated and dehydrated monomeric silicon species were therefore compiled from the literature, extrapolated to zero ionic strength by specific ion interaction theory as required and used to derive their thermodynamic properties and r-H-K-F EoS parameters. Consideration of all formation reactions in the same study provides a collective, internally consistent update to the thermodynamic properties and r-H-K-F EoS parameters of the monomeric silicon species that can provide a satisfactory match to the experimental log10 K values at
) monomeric silicon species are used extensively to describe the pH, composition, temperature, and pressure dependence of formation/breakdown reactions of all silicon-bearing compounds globally. Experimental log10 equilbrium constant, K values describing the formation reactions of the hydrated and dehydrated monomeric silicon species were therefore compiled from the literature, extrapolated to zero ionic strength by specific ion interaction theory as required and used to derive their thermodynamic properties and r-H-K-F EoS parameters. Consideration of all formation reactions in the same study provides a collective, internally consistent update to the thermodynamic properties and r-H-K-F EoS parameters of the monomeric silicon species that can provide a satisfactory match to the experimental log10 K values at 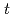 = 0.01-600
= 0.01-600 C,
C,  = 1-3000 bars,
= 1-3000 bars, 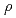
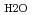 = 0.35-1.1 g cm
= 0.35-1.1 g cm , and zero ionic strength. These temperature and pressure limits comfortably bracket t=0.01-100
, and zero ionic strength. These temperature and pressure limits comfortably bracket t=0.01-100 C and P =1-270 bars relecant to the geological disposal of radioactive wastes at depths of up to 1 km.
C and P =1-270 bars relecant to the geological disposal of radioactive wastes at depths of up to 1 km.
Kato, Yuto*; Sasaki, Takayuki*; Tonna, Ryutaro*; Kobayashi, Taishi*; Okamoto, Yoshihiro
Applied Geochemistry, 175, p.106196_1 - 106196_9, 2024/11
Times Cited Count:1 Percentile:0.00(Geochemistry & Geophysics)Mei, H.; Aoyagi, Noboru; Saito, Takumi*; Tanaka, Kazuya; Sugiura, Yuki; Tachi, Yukio
Applied Geochemistry, 162, p.105926_1 - 105926_8, 2024/02
Times Cited Count:2 Percentile:69.68(Geochemistry & Geophysics)Arthur, R.*; Sasamoto, Hiroshi; Alt-Epping, P.*; Tachi, Yukio
Applied Geochemistry, 155, p.105707_1 - 105707_8, 2023/08
Times Cited Count:2 Percentile:32.37(Geochemistry & Geophysics)The experience gained in modeling the evolution, from past to present, of natural tracer profiles in geologic media can help support safety assessment of disposal concepts for radioactive wastes in deep geologic repository. Solute-transport models were developed in the present study using a forward modeling approach constrained by boundary conditions inferred from the paleo-hydrogeological evolution of the Horonobe area in Hokkaido, Japan. Apparent differences in transport behavior at the two boreholes location considered in this study, which were situated only about 1 km apart, appear to have resulted from relatively small differences in accessible porosity and hydraulic conductivity, which in turn may have been controlled by local differences in fracture density and fracture connectivity.
Mochizuki, Akihito; Ishii, Eiichi
Applied Geochemistry, 155, p.105737_1 - 105737_15, 2023/08
Times Cited Count:3 Percentile:32.37(Geochemistry & Geophysics)Understanding the difference in groundwater flow between glacial and interglacial periods is crucial for predicting the impact of future climate changes on groundwater movement. This study assesses the difference in groundwater flow between the last glacial period (LGP) and the postglacial period (PGP) in fractured mudstones of the Horonobe area, Japan, by combining the data for stable isotopes ( D and
D and 
 O) and Cl
O) and Cl concentration of fracture and pore waters with radiocarbon (
concentration of fracture and pore waters with radiocarbon ( C) age. The isotopic compositions of fractures and pore waters indicate that groundwater at 28
C) age. The isotopic compositions of fractures and pore waters indicate that groundwater at 28 250 m deep in a borehole closest to the recharge area comprises meteoric water, recharged under the same climates as the present. The fracture water has isotopic compositions more similar to meteoric water than the matrix pore water near the fracture. The
250 m deep in a borehole closest to the recharge area comprises meteoric water, recharged under the same climates as the present. The fracture water has isotopic compositions more similar to meteoric water than the matrix pore water near the fracture. The  C age of fracture water suggests meteoric water recharge during the PGP. At greater depths in the borehole and sampling points in other boreholes, the isotopic compositions indicate the mixing of glacial meteoric and altered connate water, with the fracture water having comparable isotopic compositions with the matrix pore water. The recharge timing of meteoric water is inferred to be the LGP or before based on
C age of fracture water suggests meteoric water recharge during the PGP. At greater depths in the borehole and sampling points in other boreholes, the isotopic compositions indicate the mixing of glacial meteoric and altered connate water, with the fracture water having comparable isotopic compositions with the matrix pore water. The recharge timing of meteoric water is inferred to be the LGP or before based on  C dating. These results suggest that the meteoric water recharged during the PGP flows at a shallow depth, whereas the meteoric water recharged during the LGP intruded to greater depths. This result is consistent with previous inferences from surface geophysical and geological surveys that the depths of local valleys during the LGP were greater by
C dating. These results suggest that the meteoric water recharged during the PGP flows at a shallow depth, whereas the meteoric water recharged during the LGP intruded to greater depths. This result is consistent with previous inferences from surface geophysical and geological surveys that the depths of local valleys during the LGP were greater by  50 m than the present ones and enhanced the downward hydraulic gradient. Combining the chemical and isotopic compositions of groundwater with
50 m than the present ones and enhanced the downward hydraulic gradient. Combining the chemical and isotopic compositions of groundwater with  C age helps assess the groundwater flow during the LGP and PGP in fractured rocks.
C age helps assess the groundwater flow during the LGP and PGP in fractured rocks.
Abe, Takeyasu; Iida, Yoshihisa; Sasamoto, Hiroshi; Ishii, Eiichi
Proceedings of Water-Rock Interaction (WRI-17)/ Applied Isotope Geochemistry (AIG-14) (Internet), 6 Pages, 2023/08
Cation exchange is one of important elementary reactions among water-rock interactions in sedimentary rocks. In order to model non-constant Na-K-Ca-Mg-H exchange selectivity of sedimentary rocks, exchange isotherms and pH dependence on cation exchange capacity of smectite were analyzed following active fraction model. As the result of comparison of exchangeable cationic composition between measured and calculated, selectivity coefficients determined in this study were preferred to those presented by previous studies. Using the coefficients and groundwater hydrochemistry reported previously, expected compositions of exchangeable cations were calculated. The results suggested that pH and total Na-K-Ca-Mg-H concentration of groundwater are important factors to interpret observed compositional variation in exchangeable base cations.
Murota, Kento*; Aoyagi, Noboru; Mei, H.; Saito, Takumi*
Applied Geochemistry, 152, p.105620_1 - 105620_11, 2023/05
Times Cited Count:5 Percentile:57.51(Geochemistry & Geophysics)Mizuno, Takashi; Suzuki, Yohei*; Milodowski, A. E.*; Iwatsuki, Teruki
Applied Geochemistry, 150, p.105571_1 - 105571_11, 2023/03
Times Cited Count:2 Percentile:32.37(Geochemistry & Geophysics)Anaerobic oxidation of methane (AOM) affects both the redox conditions and carbon cycle in groundwater. However, examples of studies on crystalline rock deep in terrestrial subsurface as well as the potential host rock for geological disposal are few. Therefore, we conducted a paleohydrogeological study on fracture-filling calcite in the Toki Granite. The 
 O
O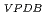 value (-32.7 to -0.59 permil) revealed that the groundwater that precipitated the calcite was groundwater derived from hydrothermal fluid, freshwater that came from the surface, and seawater that penetrated during marine transgression. On the other hand,
value (-32.7 to -0.59 permil) revealed that the groundwater that precipitated the calcite was groundwater derived from hydrothermal fluid, freshwater that came from the surface, and seawater that penetrated during marine transgression. On the other hand, 
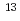 C
C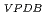 (-56.6 to 6.0 permil) was wider than the isotopic range of DIC that originated from hydrothermal, freshwater, and seawater sources (-25 to 2 permil). Calcite with
(-56.6 to 6.0 permil) was wider than the isotopic range of DIC that originated from hydrothermal, freshwater, and seawater sources (-25 to 2 permil). Calcite with 
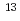 C
C that was lighter than -25 permil was believed to have precipitated DIC, which was provided by AOM. In contrast to previous studies, the Mizunami AOM calcite was precipitated in a freshwater environment, indicating that various processes could have generated AOM in crystalline rocks deep in the terrestrial subsurface.
that was lighter than -25 permil was believed to have precipitated DIC, which was provided by AOM. In contrast to previous studies, the Mizunami AOM calcite was precipitated in a freshwater environment, indicating that various processes could have generated AOM in crystalline rocks deep in the terrestrial subsurface.
Ochs, M.*; Dolder, F.*; Tachi, Yukio
Applied Geochemistry, 136, p.105161_1 - 105161_11, 2022/01
Times Cited Count:9 Percentile:65.01(Geochemistry & Geophysics)Various types of radioactive wastes and environments contain organic substances that can stabilize the aqueous complexes with radionuclides and therefore lead to a decrease of sorption. The present study focuses on testing a methodology to quantify sorption reduction factors (SRFs) in the presence of organic ligands for cement systems. Three approaches for the estimation of SRFs; (1) analogy with solubility enhancement factors, (2) radionuclide speciation based on the thermodynamic calculations, and (3) experimental sorption data in ternary systems, were coupled and tested for the representative organic ligands (ISA and EDTA) and selected key radionuclides (actinides). Our approach allows to critically evaluate the dependence of SRFs for various systems on the chosen method of quantification, in accordance with the data availability for a given systems. The reliable SRFs can only be derived from the sorption measurements in ternary systems. SRF often need to be derived in the absence of such direct evidence, and estimations need to be made based on analogies and speciation information. However, such estimates may be subject to substantial uncertainties.
Mei, H.; Aoyagi, Noboru; Saito, Takumi*; Kozai, Naofumi; Sugiura, Yuki; Tachi, Yukio
Applied Geochemistry, 136, p.105178_1 - 105178_8, 2022/01
Times Cited Count:20 Percentile:90.04(Geochemistry & Geophysics)Wilson, J.*; Bateman, K.; Tachi, Yukio
Applied Geochemistry, 130, p.104979_1 - 104979_19, 2021/07
Times Cited Count:21 Percentile:53.85(Geochemistry & Geophysics)The concept of deep geological disposal will include the multiple use of cement-based materials. In the case of argillaceous host rocks, the presence of hyperalkaline cement porefluid results in the destabilization of primary minerals in the argillite, resulting in the development of a zone of alteration at cement-rock interfaces. The process understanding gained from experimental, analogue, and modelling studies has been reviewed, and remaining areas of uncertainty identified. Although there is a reasonably good understanding of the mineral assemblages that are likely to occur due to cement-rock interactions, there are still some areas where a degree of uncertainty remains, in particular: the evolution of cement-argillite interfaces at T  25
25 C; the rates at which secondary minerals form; the extent of pore clogging due to secondary mineral precipitation; the implications of alteration for radionuclide transport.
C; the rates at which secondary minerals form; the extent of pore clogging due to secondary mineral precipitation; the implications of alteration for radionuclide transport.
Watanabe, Takahiro; Tsuchiya, Noriyoshi*; Yamasaki, Shinichi*; Sawai, Yuki*; Hosoda, Norihiro*; Nara, Fumiko*; Nakamura, Toshio*; Komai, Takeshi*
Applied Geochemistry, 118, p.104644_1 - 104644_11, 2020/07
Times Cited Count:18 Percentile:72.08(Geochemistry & Geophysics)Possible tsunami inundation areas can be predicted from the distribution of paleo tsunami deposits, which are mainly composed of marine-derived sands and muds on land. Such exotic sandy and muddy layers have been identified by multiple approaches. However, there still remain uncertainties and other useful proxies need to be investigated for the identification of tsunami deposits. Here we show the geochemical signatures of modern tsunami deposits from the Pacific coast of Northeast Japan (2011 Tohoku tsunami), as well as those paleo tsunami deposits, which were taken from the Tohoku District (Jogan, ca. 1080 calibrated ages before present [cal BP] and Yayoi, ca. 2000 cal BP). A geochemical ternary diagram (Seawater-Rock-(As+Metals)) enables the weathering trend of tsunami deposits over ca. 2000 years in the Sendai Plain in the Tohoku to be shown. In the paleo tsunami layers from the Tohoku, the Na/Ti atomic ratios markedly increased to 23.4, and the average values were 19.3 3.0 (Jogan and Yayoi tsunami deposits), which were clearly higher than those of other layers (soil deposits, 10.3
3.0 (Jogan and Yayoi tsunami deposits), which were clearly higher than those of other layers (soil deposits, 10.3 3.5 on average). These results show that the Na/Ti ratio is a useful indicator of marine incursions in our case. Our rapid and simple method using the Na/Ti ratio can be easily applied to the Tohoku, and it may contribute to the detection of unrecorded muddy tsunami deposits.
3.5 on average). These results show that the Na/Ti ratio is a useful indicator of marine incursions in our case. Our rapid and simple method using the Na/Ti ratio can be easily applied to the Tohoku, and it may contribute to the detection of unrecorded muddy tsunami deposits.
Nakata, Kotaro*; Hasegawa, Takuma*; Solomon, D. K.*; Miyakawa, Kazuya; Tomioka, Yuichi*; Ota, Tomoko*; Matsumoto, Takuya*; Hama, Katsuhiro; Iwatsuki, Teruki; Ono, Masahiko*; et al.
Applied Geochemistry, 104, p.60 - 70, 2019/05
Times Cited Count:8 Percentile:36.15(Geochemistry & Geophysics)no abstracts in English
Tanaka, Kazuya; Kaplan, D. I.*; Onuki, Toshihiko
Applied Geochemistry, 85(Part B), p.119 - 120, 2017/10
Times Cited Count:0 Percentile:0.00(Geochemistry & Geophysics)We have prepared a special issue for Applied Geochemistry entitled "Transformation and Fate of Natural and Anthropogenic Radionuclides in the Environments". Here, we present 13 peer-reviewed articles on the general theme of natural and anthropogenic radionuclides in different environments. At the same time, these articles cover various topics of field research on the distribution of radionuclides, as well as laboratory experiments on adsorption and redox chemistry of these. The articles have been written by the attendees of the session at the Goldschmidt 2016 held in Yokohama, Japan, and by other authors who submitted their manuscripts to Applied Geochemistry focusing on the theme of the special issue.
Iwatsuki, Teruki; Munemoto, Takashi*; Kubota, Mitsuru*; Hayashida, Kazuki; Kato, Toshihiro*
Applied Geochemistry, 82, p.134 - 145, 2017/05
Times Cited Count:9 Percentile:33.59(Geochemistry & Geophysics)This study investigated the behavior of rare earth elements (REEs) associated with suspended particles in deep granitic groundwater and in a sealed drift at a depth of 500 m in the Mizunami Underground Research Laboratory (URL) in Japan. Approximately 10% 60% of REEs in groundwater are associated with suspended particles. Carbonate particles in groundwater are most likely derived from in situ precipitation of supersaturated carbonate minerals such as calcite. Thermodynamic calculations show that the dissolved REE carbonate complexes in the closed drift decreased in the drift closure period. These complexes may have been absorbed or co-precipitated within the shotcrete on the drift wall. The usage of cement based materials would generate environmental conditions in which REEs are fundamentally immobile in and around the underground facilities.
60% of REEs in groundwater are associated with suspended particles. Carbonate particles in groundwater are most likely derived from in situ precipitation of supersaturated carbonate minerals such as calcite. Thermodynamic calculations show that the dissolved REE carbonate complexes in the closed drift decreased in the drift closure period. These complexes may have been absorbed or co-precipitated within the shotcrete on the drift wall. The usage of cement based materials would generate environmental conditions in which REEs are fundamentally immobile in and around the underground facilities.
Yamasaki, Shinya*; Tanaka, Kazuya; Kozai, Naofumi; Onuki, Toshihiko
Applied Geochemistry, 78, p.279 - 286, 2017/03
Times Cited Count:6 Percentile:23.48(Geochemistry & Geophysics)This study examined the rate constant for the U(VI) reduction process by three flavin analogues, which are redox-active biomolecules secreted from anaerobic bacteria, to elucidate their substituent group effect on the U(VI) reduction rate by electrochemical methods. The formation of the U(IV) was monitored by UV-vis spectrometry in the presence of the flavins. The rate constant for the U(VI) reduction by the flavins was determined. The apparent reduction potential of U(VI) increased about 0.2 V in the presence of the mediators, which strongly suggests that the biological electron mediator makes the U(VI) reduction possible even under more oxidative conditions.
Miyakawa, Kazuya; Ishii, Eiichi; Hirota, Akinari*; Komatsu, Daisuke*; Ikeya, Kosuke*; Tsunogai, Urumu*
Applied Geochemistry, 76, p.218 - 231, 2017/01
Times Cited Count:24 Percentile:66.73(Geochemistry & Geophysics)no abstracts in English
Umeda, Koji; Asamori, Koichi; Kusano, Tomohiro
Applied Geochemistry, 37, p.134 - 141, 2013/10
Times Cited Count:21 Percentile:49.61(Geochemistry & Geophysics)This study was undertaken to elucidate spatial and temporal changes of  He/
He/ He ratios around a source region before and after an inland earthquake using both new and existing helium isotope data from hot spring and drinking water wells. Most gas samples near the seismic source region are characterized by increase in postseismic
He ratios around a source region before and after an inland earthquake using both new and existing helium isotope data from hot spring and drinking water wells. Most gas samples near the seismic source region are characterized by increase in postseismic  He/
He/ He ratios. In contrast, the postseismic ratios decrease by up to about 30% away from the seismic source region. Episodic faulting could release accumulated radiogenic helium from host rocks, or enhance the transfer of mantle volatiles through permeable fault zones, such that subsequent fluid flow proximal to the source region could then explain the spatio-temporal variations in
He ratios. In contrast, the postseismic ratios decrease by up to about 30% away from the seismic source region. Episodic faulting could release accumulated radiogenic helium from host rocks, or enhance the transfer of mantle volatiles through permeable fault zones, such that subsequent fluid flow proximal to the source region could then explain the spatio-temporal variations in  He/
He/ He ratios.
He ratios.
Sasamoto, Hiroshi; Arthur, R. C.*; Hama, Katsuhiro
Applied Geochemistry, 26(8), p.1464 - 1477, 2011/08
Times Cited Count:20 Percentile:46.51(Geochemistry & Geophysics)It is important to evaluate the undisturbed hydrochemical conditions in deep underground for considering the initial geochemical setting of performance assessment of high level radioactive waste. In the present study, groundwater data obtained thorough the initial surface-based investigation at the Horonobe URL were used to check the correlation and reliability of data based on empirical, geostatistical and geochemical modeling techniques. Applicability of these techniques were discussed in the present study and the representative undisturbed hydrochemical conditions were estimated.
Savage, D.*; Soler, J. M.*; Yamaguchi, Kohei; Walker, C.; Honda, Akira; Inagaki, Manabu; Watson, C.*; Wilson, J.*; Benbow, S.*; Gaus, I.*; et al.
Applied Geochemistry, 26(7), p.1138 - 1152, 2011/07
Times Cited Count:20 Percentile:46.51(Geochemistry & Geophysics)The use of cement and concrete as fracture grouting or as tunnel seals in a geological disposal facility for rad wastes creates potential issues concerning chemical reactivity. From a long term safety perspective, it is desirable to be able model these interactions and changes quantitatively. As part of the LCS (Long-term Cement Studies) project programme, a modelling inter-comparison has been conducted, involving the modelling of two experiments describing cement hadration and cement-rock reaction, with teams representing the NDA (UK), Posiva (Finland), and JAEA. This modelling exercise showed that the dominant reaction pathways in the two experiments are fairly well understood and are consistent between the different modelling teams, although significant differences existed amongst the precise parameterisation. Future modelling exercises of this type should focus on a suitable natural or industrial analogue that might aid assessing mineral-fluid reactions at these longer timescales.