Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 resolution
resolutionUemura, Takuya*; Kita, Akiko*; Watanabe, Yoshihiko*; Adachi, Motoyasu; Kuroki, Ryota; Morimoto, Yukio*
Biochemical and Biophysical Research Communications, 469(2), p.158 - 163, 2016/01
Times Cited Count:18 Percentile:53.10(Biochemistry & Molecular Biology)Matsuo, Tatsuhito; Arata, Toshiaki*; Oda, Toshiro*; Nakajima, Kenji; Kawamura, Seiko; Kikuchi, Tatsuya; Fujiwara, Satoru
Biochemical and Biophysical Research Communications, 459(3), p.493 - 497, 2015/04
Times Cited Count:5 Percentile:15.16(Biochemistry & Molecular Biology)Hata, Kuniki; Urushibara, Ayumi; Yamashita, Shinichi; Shikazono, Naoya; Yokoya, Akinari; Katsumura, Yosuke*
Biochemical and Biophysical Research Communications, 434(2), p.341 - 345, 2013/05
Times Cited Count:8 Percentile:22.50(Biochemistry & Molecular Biology)Fujiwara, Satoru; Plazanet, M.*; Oda, Toshiro*
Biochemical and Biophysical Research Communications, 431(3), p.542 - 546, 2013/02
Times Cited Count:4 Percentile:10.38(Biochemistry & Molecular Biology)Matsumoto, Fumiko; Maeda, Kayo*; Chatake, Toshiyuki*; Maeda, Yuichiro*; Fujiwara, Satoru
Biochemical and Biophysical Research Communications, 382(1), p.205 - 209, 2009/04
Times Cited Count:15 Percentile:36.64(Biochemistry & Molecular Biology)Two cardiomyopathy-causing mutations, E244D and K247R, in human cardiac troponin T (TnT) are located in the coiled-coil region of the Tn-core domain. To elucidate effects of mutations in this region on the regulatory function of Tn, we measured Ca -dependent ATPase activity of myofibrils containing various mutants of TnT at these residues. The results confirmed that the mutant E244D increases the maximum ATPase activity without changing the Ca
-dependent ATPase activity of myofibrils containing various mutants of TnT at these residues. The results confirmed that the mutant E244D increases the maximum ATPase activity without changing the Ca sensitivity. The mutant K247R was shown for the first time to have the effect similar to the mutant E244D. Furthermore, various TnT mutants (E244D, E244M, E244A, E244K, K247R, K247E, and K247A) showed various effects on the maximum ATPase activity while the Ca
sensitivity. The mutant K247R was shown for the first time to have the effect similar to the mutant E244D. Furthermore, various TnT mutants (E244D, E244M, E244A, E244K, K247R, K247E, and K247A) showed various effects on the maximum ATPase activity while the Ca sensitivity was unchanged. Molecular dynamics simulations of the Tn-core containing these TnT mutants suggested that the hydrogen-bond network formed by the side chains of neighboring residues around residues 244 and 247 is important for Tn to function properly.
sensitivity was unchanged. Molecular dynamics simulations of the Tn-core containing these TnT mutants suggested that the hydrogen-bond network formed by the side chains of neighboring residues around residues 244 and 247 is important for Tn to function properly.
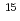 N NMR relaxation measurements
N NMR relaxation measurementsYoshida, Yuichiro*; Okuri, Takatoshi*; Takeda, Chika*; Kuroki, Ryota; Izuhara, Kenji*; Imoto, Taiji*; Ueda, Tadashi*
Biochemical and Biophysical Research Communications, 358(1), p.292 - 297, 2007/06
Times Cited Count:4 Percentile:9.61(Biochemistry & Molecular Biology)The single nucleotide polymorphism interleukin-13 (IL-13) R110Q is associated with severe bronchial asthma because its lower affinity leads to the augmentation of local IL-13 concentration, resulting in an increase in the signal transduction via IL-13R. Since the mutation site does not directly bind to IL-13 R 2, we carried out NMR relaxation analyses of the wild-type IL-13 and IL-13 R110Q in order to examine whether the R110Q mutation affects the internal motions in IL-13 molecules. The results showed that the internal motion in the micro- to millisecond time scale on helix D, which is suggested to be important for the interaction between IL-13 and IL-13R
2, we carried out NMR relaxation analyses of the wild-type IL-13 and IL-13 R110Q in order to examine whether the R110Q mutation affects the internal motions in IL-13 molecules. The results showed that the internal motion in the micro- to millisecond time scale on helix D, which is suggested to be important for the interaction between IL-13 and IL-13R 2, is increased in IL-13-R110Q compared with that in the wild-type IL-13. It therefore appears that the difference in the internal motions on helix D between the wild-type IL-13 and IL-13-R110Q may be involved in their affinity differences with IL-13R
2, is increased in IL-13-R110Q compared with that in the wild-type IL-13. It therefore appears that the difference in the internal motions on helix D between the wild-type IL-13 and IL-13-R110Q may be involved in their affinity differences with IL-13R 2.
2.

Hua, Y.*; Narumi, Issei; Gao, G.*; Tian, B.*; Sato, Katsuya; Kitayama, Shigeru; Shen, B.*
Biochemical and Biophysical Research Communications, 306(2), p.354 - 360, 2003/06
Times Cited Count:157 Percentile:95.88(Biochemistry & Molecular Biology)We have identified a unique deinococcal gene, 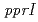 , as a general switch for downstream DNA repair and protection pathways, from a natural mutant, in which
, as a general switch for downstream DNA repair and protection pathways, from a natural mutant, in which 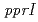 is disrupted by a transposon. Complete functional disruption of the gene in wild-type leads to dramatic sensitivity to ionizing radiation. Radioresistance of the disruptant could be fully restored by complementation with
is disrupted by a transposon. Complete functional disruption of the gene in wild-type leads to dramatic sensitivity to ionizing radiation. Radioresistance of the disruptant could be fully restored by complementation with 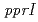 . In response to radiation stress, PprI can significantly and specifically induce the gene expression of
. In response to radiation stress, PprI can significantly and specifically induce the gene expression of 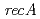 and
and 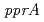 and enhance the enzyme activities of catalases. These results strongly suggest that PprI plays a crucial role in regulating multiple DNA repair and protection pathways in response to radiation stress.
and enhance the enzyme activities of catalases. These results strongly suggest that PprI plays a crucial role in regulating multiple DNA repair and protection pathways in response to radiation stress.
; ; ; Shibabe, Sadami; ;
Biochemical and Biophysical Research Communications, 172(2), p.576 - 581, 1990/10
Times Cited Count:14 Percentile:39.21(Biochemistry & Molecular Biology)no abstracts in English
; ; Ishido, Akio; ; Kume, Tamikazu; Ishigaki, Isao;
Biochemical and Biophysical Research Communications, 158(2), p.603 - 609, 1989/01
Times Cited Count:9 Percentile:30.29(Biochemistry & Molecular Biology)no abstracts in English