Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kinoshita, Ryoma; Sasaki, Yuji; Kaneko, Masashi; Matsumiya, Masahiko*; Shinoku, Kota*; Shiroishi, Hidenobu*
Hydrometallurgy, 222, p.106159_1 - 106159_12, 2023/10
Times Cited Count:2 Percentile:23.52(Metallurgy & Metallurgical Engineering)Solvent extraction is conducted using a total of 20 metals revealing high stability constants with Cl and hexahexyl-nitrilotriacetamide (NTAamide(C6)) extractant. The metals used here may behave as anions at high Cl concentrations, and NTAamide(C6), which contains a tertiary N atom, is protonated under acidic conditions. Most of the metal ions in this study display higher distribution ratios (D(M)) from HCl than those from HNO , and exhibit 1:1 stoichiometries with NTAamide. Following the experimental results, the association constants and distribution coefficients of the group 12 elements are calculated via ion-pair extraction modeling using density functional theory calculations, and the simulations of D yield calculated values with the same trend as that of the measured values.
, and exhibit 1:1 stoichiometries with NTAamide. Following the experimental results, the association constants and distribution coefficients of the group 12 elements are calculated via ion-pair extraction modeling using density functional theory calculations, and the simulations of D yield calculated values with the same trend as that of the measured values.
Nguyen, T. H.*; Le Ba, T.*; Tran, C. T.*; Nguyen, T. T.*; Doan, T. T. T.*; Do, V. K.; Watanabe, Masayuki; Pham, Q. M.*; Hoang, S. T.*; Nguyen, D. V.*; et al.
Hydrometallurgy, 213, p.105933_1 - 105933_11, 2022/08
Times Cited Count:18 Percentile:80.29(Metallurgy & Metallurgical Engineering)A continuous counter-current extraction for the selective recovery of thorium (Th) and uranium (U) from the Yen Phu (Vietnam) rare earth concentrate leach solutions was systematically studied. The primary amine N1923 was used as an extractant which was prepared in the isoparaffin IP-2028 diluent. Thorium and uranium were selectively recovered in a hydrometallurgical circuit established by continuous mixer-settler extraction, scrubbing, and back-extraction at the laboratory scale. The desired purity of Th and U can be achieved by managing the volume ratio of organic to aqueous phase (O/A ratio) in the corresponding steps. Highly pure Th and U were recovered from the pregnant back-extraction liquor and the raffinate, respectively, which have satisfactory properties for further processing of the subsequent nuclear materials.
Matsumiya, Masahiko*; Nomizu, Daiki*; Tsuchida, Yusuke*; Sasaki, Yuji
Hydrometallurgy, 199, p.105539_1 - 105539_8, 2021/02
Times Cited Count:6 Percentile:32.21(Metallurgy & Metallurgical Engineering)The synergistic solvent extraction of lanthanide(III) with mixtures of di-(2-ethylhexyl)phosphoric acid (D2EHPA, A) and monoisodecyl phosphoric acid (MIDPA, B) in phosphonium-based ionic liquid was investigated. In the case of D2EHPA or MIDPA single extractant system, Ln(III) (Ln = Pr and Nd) was extracted as [LnA HA] or [LnB
HA] or [LnB HB], respectively, the extracted species of Tb(III) or Dy(III) were determined by slope analysis. According to the equilibrium constants (
HB], respectively, the extracted species of Tb(III) or Dy(III) were determined by slope analysis. According to the equilibrium constants (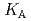 ,
, 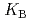 and
and 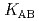 ) and the formation constants (
) and the formation constants (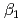 ,
, 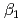 and
and 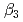 ), it was found that the extracted complex [TbHA
), it was found that the extracted complex [TbHA B
B or [DyHA
or [DyHA B
B ] was more stable than [LnA
] was more stable than [LnA HA] or [LnB
HA] or [LnB HB]. The synergistic extraction effects were investigated to study the possibility of separating Dy(III) from Pr(III) and Nd(III) according to their separation factors.
HB]. The synergistic extraction effects were investigated to study the possibility of separating Dy(III) from Pr(III) and Nd(III) according to their separation factors.
Sasaki, Yuji; Morita, Keisuke; Saeki, Morihisa*; Hisamatsu, Shugo; Yoshizuka, Kazuharu*
Hydrometallurgy, 169, p.576 - 584, 2017/05
Times Cited Count:16 Percentile:55.22(Metallurgy & Metallurgical Engineering)The novel tridentate extractant including soft donor has developed and examined. The extractant, tetraoctyl-thiodiglycolamide (TDGA), is analogous structure to tetraoctyl-diglycolamide (TODGA) and methylimino-dioctylacetamide (MIDOA), but with sulfur donor instead of ether oxygen or nitrogen atoms of TODGA or MIDOA. From the present work, TDGA can extract silver, palladium, gold, and mercury from acidic solutions to n-dodecane. In addition to these results, the distribution ratios of hard and soft acid metals by using TDGA, TODGA, and MIDOA are compared, where the metal-complexations with each donor atom are investigated. 1H-NMR and IR studies for the metal-TDGA complexes indicate the role on donor atoms, S and N, of TDGA.
Fujimoto, Jun*; Tanaka, Kazuya; Watanabe, Naoko*; Takahashi, Yoshio*
Hydrometallurgy, 166, p.80 - 86, 2016/12
Times Cited Count:7 Percentile:32.09(Metallurgy & Metallurgical Engineering)We examined recovery of REEs in Fe-Mn nodules by using 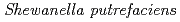 (Fe-reducing bacterium). In this method, Fe-Mn nodule decomposition and REE recovery were achieved simultaneously in a single solution system. Fe-Mn nodules were reductively decomposed in NaCl solution under anaerobic conditions with daily addition of sodium lactate as an electron donor. During the decomposition of Fe-Mn nodule, REEs released from the Fe-Mn nodule were adsorbed on bacterial cells. Of the conditions studied here, the best REE adsorption rates were obtained with 0.5M NaCl solution at pH7 with daily addition of 1 mmol sodium lactate.
(Fe-reducing bacterium). In this method, Fe-Mn nodule decomposition and REE recovery were achieved simultaneously in a single solution system. Fe-Mn nodules were reductively decomposed in NaCl solution under anaerobic conditions with daily addition of sodium lactate as an electron donor. During the decomposition of Fe-Mn nodule, REEs released from the Fe-Mn nodule were adsorbed on bacterial cells. Of the conditions studied here, the best REE adsorption rates were obtained with 0.5M NaCl solution at pH7 with daily addition of 1 mmol sodium lactate.
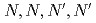 -tetraoctyl-thiodiglycolamide
-tetraoctyl-thiodiglycolamideSasaki, Yuji; Suzuki, Tomoya; Morita, Keisuke; Yoshizuka, Kazuharu*
Hydrometallurgy, 159, p.107 - 109, 2016/01
Times Cited Count:5 Percentile:24.71(Metallurgy & Metallurgical Engineering)The novel tridentate extractant including soft donor has developed and examined. The extractant, tetraoctyl-thiodiglycolamide (S-DGA), is analogous structure to tetraoctyl-diglycolamide (TODGA), but with sulfur donor instead of ether oxygen of TODGA. S-DGA has unique property to extract silver from acidic solutions to n-dodecane with relatively high D values. We investigated the extraction behavior of Ag in various acids, HNO , H
, H SO
SO , and HClO
, and HClO .
.
Yu, Q.; Sasaki, Keiko*
Hydrometallurgy, 150, p.253 - 258, 2014/12
Times Cited Count:9 Percentile:39.33(Metallurgy & Metallurgical Engineering)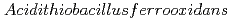
Sasaki, Keiko*; Takatsugi, Koichiro*; Kaneko, Kenji*; Kozai, Naofumi; Onuki, Toshihiko; Tuovinen, O. H.*; Hirajima, Tsuyoshi*
Hydrometallurgy, 104(3-4), p.424 - 431, 2010/10
Times Cited Count:23 Percentile:69.13(Metallurgy & Metallurgical Engineering)no abstracts in English