Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 C and 500
C and 500 C
CTakagi, Honoka*; Yabutsuka, Takeshi*; Hayashida, Hirotoshi*; Song, F.; Kai, Tetsuya; Shinohara, Takenao; Kurita, Keisuke; Iikura, Hiroshi; Yamamoto, Norio*; Nakajima, Minoru*; et al.
Solid State Ionics, 417, p.116716_1 - 116716_7, 2024/12
Times Cited Count:2 Percentile:0.00(Chemistry, Physical) -dispersed LATP by means of neutron radiography
-dispersed LATP by means of neutron radiographySong, F.*; Chen, H.*; Hayashida, Hirotoshi*; Kai, Tetsuya; Shinohara, Takenao; Yabutsuka, Takeshi*; Yao, Takeshi*; Takai, Shigeomi*
Solid State Ionics, 377, p.115873_1 - 115873_6, 2022/04
Times Cited Count:4 Percentile:29.55(Chemistry, Physical) S)
S) (SiS
(SiS )
)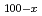 superionic glasses
superionic glassesMori, Kazuhiro*; Iwase, Kenji*; Oba, Yojiro; Ikeda, Kazutaka*; Otomo, Toshiya*; Fukunaga, Toshiharu*
Solid State Ionics, 344, p.115141_1 - 115141_10, 2020/01
Times Cited Count:14 Percentile:55.94(Chemistry, Physical)no abstracts in English
 (LiPO
(LiPO )
)
Kartini, E.*; Nakamura, Mitsutaka; Arai, Masatoshi; Inamura, Yasuhiro; Nakajima, Kenji; Maksum, T.*; Hoggowiranto, W.*; Putra, T. Y. S. P.*
Solid State Ionics, 262, p.833 - 836, 2014/09
Times Cited Count:19 Percentile:58.45(Chemistry, Physical)We present the neutron scattering study of superionic glass (LiI) (LiPO
(LiPO )
) by using the inelastic neutron spectrometer AMATERAS at J-PARC. From the inelastic scattering, low energy excitation or the Boson Peak appears at
by using the inelastic neutron spectrometer AMATERAS at J-PARC. From the inelastic scattering, low energy excitation or the Boson Peak appears at  6.5 meV. The extra quasielastic scattering was observed in the energy range 1 meV
6.5 meV. The extra quasielastic scattering was observed in the energy range 1 meV  E
E  3 meV at 285 K but not at 20 K. The Q dependence of the dynamical structure factor S(Q,E) can be approximated to the long-wave acoustic like density fluctuation.
3 meV at 285 K but not at 20 K. The Q dependence of the dynamical structure factor S(Q,E) can be approximated to the long-wave acoustic like density fluctuation.
 O
O measured by neutron radiography
measured by neutron radiographyTakai, Shigeomi*; Yoshioka, Kazuya*; Iikura, Hiroshi; Matsubayashi, Masahito; Yao, Takeshi*; Esaka, Takao*
Solid State Ionics, 256, p.93 - 96, 2014/03
Times Cited Count:39 Percentile:77.37(Chemistry, Physical) Ba
Ba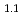 Ga
Ga Mg
Mg O
O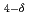
Hamao, Naoki*; Kitamura, Naoto*; Ito, Takanori*; Igawa, Naoki; Idemoto, Yasushi*; Daido, Hiroyuki; Kaku, Masanori*; Kubodera, Masakazu*
Solid State Ionics, 253, p.123 - 129, 2013/12
Times Cited Count:3 Percentile:14.25(Chemistry, Physical)La Ba
Ba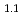 Ga
Ga Mg
Mg O
O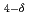 which is one of the candidates for solid electrolyte of solid oxide fuel cell was synthesized by means of a conventional solid-state reaction, and investigated by conductivity measurements, the Rietveld and maximum entropy method analyses using neutron and synchrotron X-ray diffraction data. Conductivity measurements indicated that simultaneous Ba and Mg substitutions for LaBaGaO
which is one of the candidates for solid electrolyte of solid oxide fuel cell was synthesized by means of a conventional solid-state reaction, and investigated by conductivity measurements, the Rietveld and maximum entropy method analyses using neutron and synchrotron X-ray diffraction data. Conductivity measurements indicated that simultaneous Ba and Mg substitutions for LaBaGaO were effective way to improve the protonic conductivity. From the Rietveld and maximum entropy method analyses, it was confirmed that La
were effective way to improve the protonic conductivity. From the Rietveld and maximum entropy method analyses, it was confirmed that La Ba
Ba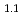 Ga
Ga Mg
Mg O
O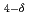 kept the same crystal structure as LaBaGaO
kept the same crystal structure as LaBaGaO even at elevated temperature, i.e. 1000 K, and that protons existed around the O3 site and formed hydrogen bonds. It is also found that oxygens in LaBaGaO
even at elevated temperature, i.e. 1000 K, and that protons existed around the O3 site and formed hydrogen bonds. It is also found that oxygens in LaBaGaO -based samples formed Ga-O
-based samples formed Ga-O tetrahedra, and the bonding strength was varied by the partial substitutions.
tetrahedra, and the bonding strength was varied by the partial substitutions.
 O from diffuse neutron scattering measurement
O from diffuse neutron scattering measurementWada, Tamotsu*; Sakuma, Takashi*; Sakai, Ryutaro*; Uehara, Hiroyuki*; Xianglian*; Takahashi, Haruyuki*; Kamishima, Osamu*; Igawa, Naoki; Danilkin, S. A.*
Solid State Ionics, 225, p.18 - 21, 2012/10
Times Cited Count:14 Percentile:49.31(Chemistry, Physical)Diffuse neutron scattering measurement on Ag O crystals was performed at 8 K and 295 K. The observed diffuse scattering intensities were analyzed by including the correlation effects among thermal displacements of atoms in Ag
O crystals was performed at 8 K and 295 K. The observed diffuse scattering intensities were analyzed by including the correlation effects among thermal displacements of atoms in Ag O. Using the values of correlation effects among neighboring atoms and the values of Debye-Waller temperature parameter, force constants among first, second and third nearest neighboring atoms have been evaluated.
O. Using the values of correlation effects among neighboring atoms and the values of Debye-Waller temperature parameter, force constants among first, second and third nearest neighboring atoms have been evaluated.
Sakuma, Takashi*; Xianglian*; Shimizu, Norifumi*; Mohapatra, S. R.*; Isozaki, Nobuhiro*; Uehara, Hiroyuki*; Takahashi, Haruyuki*; Basar, K.*; Igawa, Naoki; Kamishima, Osamu*
Solid State Ionics, 192(1), p.54 - 57, 2011/06
Times Cited Count:15 Percentile:51.84(Chemistry, Physical)Diffuse neutron scattering measurements have been performed on powder KBr. The oscillatory scheme of the diffuse scattering intensity is explained by the values of correlation effects among thermal displacements of 1st, 2nd and 3rd nearest neighboring atoms which are almost the same as those in other ionic crystals. Force constants among 1st, 2nd and 3rd nearest neighboring atoms are obtained from the correlation effects at 90 K are 0.83 eV/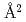 , 0.50 eV/
, 0.50 eV/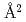 and 0.41 eV/
and 0.41 eV/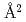 , respectively.
, respectively.
 -LnO
-LnO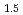 solid solutions (M
solid solutions (M =Ce, Th; Ln
=Ce, Th; Ln = Lanthanide), 2; Detailed local-structure and ionic-conductivity analysis
= Lanthanide), 2; Detailed local-structure and ionic-conductivity analysisNakamura, Akio
Solid State Ionics, 181(37-38), p.1631 - 1653, 2010/12
Times Cited Count:19 Percentile:60.28(Chemistry, Physical) -LnO
-LnO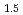 solid solutions (M
solid solutions (M =Ce, Th; Ln
=Ce, Th; Ln =Lanthanide), 1; Model description and lattice-parameter data analysis
=Lanthanide), 1; Model description and lattice-parameter data analysisNakamura, Akio
Solid State Ionics, 181(35-36), p.1543 - 1564, 2010/11
Times Cited Count:27 Percentile:69.82(Chemistry, Physical)Sakuma, Takashi*; Mohapatra, S. R.*; Yokokawa, Jo*; Shimizu, Norifumi*; Isozaki, Nobuhiro*; Uehara, Hiroyuki*; Xianglian*; Basar, K.*; Takahashi, Haruyuki*; Kamishima, Osamu*; et al.
Proceedings of 12th Asian Conference on Solid State Ionics and 15th Chinese Conference on Solid State Ionics, p.439 - 445, 2010/00
Diffuse neutron scattering intensities from ionic crystals, covalent crystals and metal crystals are analyzed by including the correlation effects among thermal displacements of atoms into the function describing background intensity. The obtained values of correlation effects among first nearest neighboring atoms are 0.7 near room temperature. The values of the correlation effects do not depend much on the type of the crystal binding near room temperature. The values of correlation effects decrease rapidly with the increase of inter-atomic distances. The correlation effects also decrease with the decrease of temperature.
 Sc
Sc O
O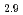
Shimoyama, Tomotaka*; Tojo, Takeo*; Kawaji, Hitoshi*; Atake, Toru*; Fukazawa, Hiroshi; Igawa, Naoki
Solid State Ionics, 180(6-8), p.560 - 562, 2009/05
Times Cited Count:6 Percentile:28.33(Chemistry, Physical)The neutron diffraction of BaZr Sc
Sc O
O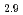 with and without proton were measured and analyzed by Rietveld method and Maximum Entropy Method. We found that the location of proton is 12
with and without proton were measured and analyzed by Rietveld method and Maximum Entropy Method. We found that the location of proton is 12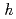 site near oxygen ion. The heat capacity of sample with proton is larger than that without proton. This difference is discussed in term of the changes of the crystal structure and lattice vibrations due to the dissolution of proton.
site near oxygen ion. The heat capacity of sample with proton is larger than that without proton. This difference is discussed in term of the changes of the crystal structure and lattice vibrations due to the dissolution of proton.
Xianglian*; Siagian, S.*; Basar, K.*; Sakuma, Takashi*; Takahashi, Haruyuki*; Igawa, Naoki; Ishii, Yoshinobu*
Solid State Ionics, 180(6-8), p.480 - 482, 2009/05
Times Cited Count:3 Percentile:15.86(Chemistry, Physical)Correlation effects among thermal vibrations of atoms are very important to realize the thermal properties of materials. We discuss the inter-atomic distance and temperature dependence of the correlation effects in ionic crystal, AgBr, CuBr, CuI and CuCl, by neutron diffraction method. From the Rietveld analysis of those diffraction data the lattice constants and Deby-Waller parameters were determined. The diffuse scattering of diffraction data was analyzed with the correlation effect,  . It is found that the
. It is found that the  is 0.6-0.75 for the inter-atomic distance around 2.5
is 0.6-0.75 for the inter-atomic distance around 2.5 and 0.4-0.5 for 4
and 0.4-0.5 for 4 for all ionic crystals. The value of
for all ionic crystals. The value of  decreases with the decrease of temperature.
decreases with the decrease of temperature.
Kobayashi, Kiyoshi*; Matsushita, Yoshitaka*; Igawa, Naoki; Izumi, Fujio*; Nishimura, Chikashi*; Miyoshi, Shogo*; Oyama, Yukiko*; Yamaguchi, Shu*
Solid State Ionics, 179(38), p.2209 - 2215, 2008/11
Times Cited Count:26 Percentile:68.65(Chemistry, Physical)A novel synthesis method of lanthanum silicate apatite was developed by a sol-gel method using an aqueous solution system. The processes of apatite phase formation were investigated by XRD and TG-DTA. Lanthanum dioxide carbonate was found in the sample below 873 K and lanthanum silicate apatite was formed above 1073 K. It was founded that the profiles of powder XRD and neutron diffraction could be refined by the oxy-apatite structure with the space group 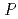 6
6 /
/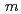 .
.
Sawada, Shinichi; Yamaki, Tetsuya; Nishimura, Hidetoshi*; Asano, Masaharu; Suzuki, Akihiro*; Terai, Takayuki*; Maekawa, Yasunari
Solid State Ionics, 179(27-32), p.1611 - 1614, 2008/09
Times Cited Count:13 Percentile:49.33(Chemistry, Physical)We measured the self-diffusion coefficient of water, D, in proton exchange membranes (PEMs) based on crosslinked polytetrafluoroethylene (PTFE) films by a radiotracer-permeation method using tritium labeled water (HTO). When the crosslinking density of the PTFE matrix increased, the D value became smaller. This decreasing trend coincided with that of a water-content parameter,  , defined as the number of water molecules per sulfonic acid (SO
, defined as the number of water molecules per sulfonic acid (SO H) group. According to the model assumption of hydrophilic regions, the relationship between the D and
H) group. According to the model assumption of hydrophilic regions, the relationship between the D and  indicates that the amount of free water in the crosslinked-PTFE PEMs could be lower than that in the non-crosslinked one, thereby restricting the mobility of water molecules through strong interactions between the majority of water and SO
indicates that the amount of free water in the crosslinked-PTFE PEMs could be lower than that in the non-crosslinked one, thereby restricting the mobility of water molecules through strong interactions between the majority of water and SO H groups.
H groups.
 adsorption and dissociation on Pt electrode
adsorption and dissociation on Pt electrodeSuzuki, Chikashi; Nakagiri, Toshio
Solid State Ionics, 179(21-26), p.855 - 861, 2008/09
Times Cited Count:3 Percentile:16.10(Chemistry, Physical)no abstracts in English

Xianglian*; Basar, K.*; Honda, Hiroyuki*; Siagian, S.*; Ohara, Kota*; Sakuma, Takashi*; Takahashi, Haruyuki*; Igawa, Naoki; Ishii, Yoshinobu*
Solid State Ionics, 179(21-26), p.776 - 779, 2008/09
Times Cited Count:3 Percentile:16.10(Chemistry, Physical)Diffuse X-ray and neutron scattering from powder PbF are measured at 15 and 294K. Those oscillatory profile of diffuse scattering is explained by including the correlation effects among thermal displacements of atoms. The contribution of correlated thermal vibration of Pb-Pb atoms located at far-off distance to the oscillatory profile of diffuse scattering intensities is first confirmed.
are measured at 15 and 294K. Those oscillatory profile of diffuse scattering is explained by including the correlation effects among thermal displacements of atoms. The contribution of correlated thermal vibration of Pb-Pb atoms located at far-off distance to the oscillatory profile of diffuse scattering intensities is first confirmed.
 Ca
Ca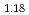 Nb
Nb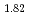 O
O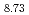
Shimoyama, Tomotaka*; Tojo, Takeo*; Kawaji, Hitoshi*; Atake, Toru*; Igawa, Naoki; Ishii, Yoshinobu*
Solid State Ionics, 179(7-8), p.231 - 235, 2008/04
Times Cited Count:15 Percentile:53.35(Chemistry, Physical)The neutron diffraction study was carried out to determine the location of proton conducting mixed perovskite-type oxide Ba Ca
Ca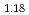 Nb
Nb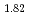 O
O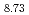 at 300K. The obtained data have been analyzed by the Rietveld method and the maximum entropy method. The crystal structure of the sample is refined on the basis of the cubic structure with the space group
at 300K. The obtained data have been analyzed by the Rietveld method and the maximum entropy method. The crystal structure of the sample is refined on the basis of the cubic structure with the space group 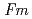 -3
-3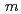 . We found that oxide ion locates at 24
. We found that oxide ion locates at 24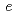 site and deuterium 96
site and deuterium 96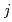 site, respectively. The distance between oxide ion and deuterium sites is about 0.72
site, respectively. The distance between oxide ion and deuterium sites is about 0.72 , which is shorter than the normal distance of OD group. The oxygen in OD group is shifted from 24
, which is shorter than the normal distance of OD group. The oxygen in OD group is shifted from 24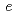 site slightly toward the opposite side of deuterium and the deuterium is shifted to the Ca/Nb mixed site.
site slightly toward the opposite side of deuterium and the deuterium is shifted to the Ca/Nb mixed site.
Sakuma, Takashi*; Xianglian*; Basar, K.*; Siagian, S.*; Shimizu, Norifumi*; Takahashi, Haruyuki*; Igawa, Naoki
Proceedings of 11th Asian Conference on Solid State Ionics, p.45 - 52, 2008/00
The diffuse neutron scattering intensities for CuI and AgBr were analyzed by the theory including the thermal correction effects up to 3rd nearest neighboring atoms. When we include the temperature dependence of the values of correlation effects, the obtained results by the refinement were improved than the former. The values of correlation effects decrease rapidly with the increase of inter-atomic distance. The values also decrease with the decrease of temperature.
 In
In O
O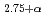 at 77-473 K by neutron powder diffraction
at 77-473 K by neutron powder diffractionIto, Tsuyoshi*; Nagasaki, Takanori*; Iwasaki, Kota*; Yoshino, Masahito*; Matsui, Tsuneo*; Fukazawa, Hiroshi; Igawa, Naoki; Ishii, Yoshinobu
Solid State Ionics, 178(7-10), p.607 - 613, 2007/04
Times Cited Count:7 Percentile:34.13(Chemistry, Physical)no abstracts in English