Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 CsI +
CsI +  Cs
Cs 
 Cs + I
Cs + I Cs
CsKobayashi, Takanori*; Matsuoka, Leo*; Yokoyama, Keiichi
Computational and Theoretical Chemistry, 1150, p.40 - 48, 2019/02
Times Cited Count:1 Percentile:1.61(Chemistry, Physical)One of important research targets in the development of cesium isotope separation system is design of recovery process of cesium atom. Relevant to this research target, the reaction cross section and reaction rate constant of a cesium exchange reaction through collision of the cesium iodide molecules with cesium atoms are calculated by a quasi-classical trajectory calculation based on a potential energy surface obtained by quantum chemistry calculations. Consequently, the rate constant is calculated to be 3.6  10
10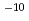 cm
cm molecule
molecule s
s , as large as collision rate in the present condition. In addition, slightly positive temperature dependence is observed in the rate constant. This behavior is explained with the long-range attractive force and effect of subsequent dissociation process.
, as large as collision rate in the present condition. In addition, slightly positive temperature dependence is observed in the rate constant. This behavior is explained with the long-range attractive force and effect of subsequent dissociation process.
 thin film as selective absorbent for cesium isotope separation using X-ray photoelectron spectroscopy
thin film as selective absorbent for cesium isotope separation using X-ray photoelectron spectroscopySekiguchi, Tetsuhiro; Yokoyama, Keiichi; Uozumi, Yuki; Asaoka, Hidehito; Yano, Masahiro; Yaita, Tsuyoshi
no journal, ,
For nuclear transmutation of cesium-135 (Cs-135), which is long-lived fission product, we are developing selective absorbent which takes only Cs atom in, but does not CsI. In this study, absorbing property of Cs atom onto the surface of fullerene (C60) film has been investigated using synchrotron-based angle-dependent X-ray photoelectron spectroscopy. The results were compared with those of CsI. It was found that [Cs-3d]/[C-1s] ratio depends moderately on emission angle for the sample of Cs on C60, while [Cs-3d]/[C-1s] ratio depends strongly on angle for the sample of CsI on C60. The results suggest that Cs penetrates into C60 bulk deeply, while CsI deposits on shallow surface.