Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Machida, Akihiko*; Saito, Hiroyuki*; Aoki, Katsutoshi*; Komatsu, Kazuki*; Hattori, Takanori; Sano, Asami; Funakoshi, Kenichi*; Machida, Shinichi*; Sato, Toyoto*; Orimo, Shinichi*
Physical Review B, 111(22), p.224413_1 - 224413_6, 2025/06
Times Cited Count:1The crystal and magnetic structures of antiferromagnetic Mn deuterides formed by hydrogenating Mn metal at high temperature and high pressure, fcc  -MnDx and hcp
-MnDx and hcp 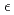 -MnDx, were investigated by in-situ neutron powder diffraction. Deuterium atoms partially occupied the octahedral interstitial positions of the fcc and hcp metal lattices. The site occupancies increased rapidly with decreasing temperature from
-MnDx, were investigated by in-situ neutron powder diffraction. Deuterium atoms partially occupied the octahedral interstitial positions of the fcc and hcp metal lattices. The site occupancies increased rapidly with decreasing temperature from  700 to
700 to  450 K and remained down to 300 K. N
450 K and remained down to 300 K. N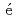 el temperature of 543(10) K was determined for
el temperature of 543(10) K was determined for  -MnD
-MnD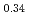 . For
. For 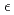 -MnD
-MnD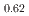 , saturation magnetic moment and N
, saturation magnetic moment and N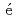 el temperature were determined to be 0.82(1)
el temperature were determined to be 0.82(1) 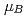 and 347(3) K, respectively. The N
and 347(3) K, respectively. The N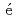 el temperatures determined for
el temperatures determined for  -MnD
-MnD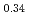 and
and 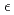 -MnD
-MnD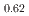 are consistent with those predicted by the respective Slater-Pauling curves proposed in previous studies. The updated N
are consistent with those predicted by the respective Slater-Pauling curves proposed in previous studies. The updated N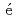 el temperatures provide insights into the development of more accurate Slater-Pauling curves based on electronic band structure calculations.
el temperatures provide insights into the development of more accurate Slater-Pauling curves based on electronic band structure calculations.
Hu, F.-F.*; Qin, T.-Y.*; Ao, N.*; Xu, P. G.; Su, Y. H.; Parker, J. D.*; Shinohara, Takenao; Shobu, Takahisa; Kang, G.-Z.*; Ren, M.-M.; et al.
Journal of Traffic and Transportation Engineering, 25(2), p.75 - 93, 2025/04
Li, F.*; Tang, X.*; Fei, Y.*; Zhang, J.*; Liu, J.*; Lang, P.*; Che, G.*; Zhao, Z.*; Zheng, Y.*; Fang, Y.*; et al.
Journal of the American Chemical Society, 147(17), p.14054 - 14059, 2025/04
Times Cited Count:1 Percentile:0.00(Chemistry, Multidisciplinary)We synthesized a crystalline graphane nanoribbon (GANR) via pressure-induced polymerization of 2,2'-bipyrazine (BPZ). By performing Rietveld refinement of in situ neutron diffraction data, nuclear magnetic resonance spectroscopy, infrared spectra, and theoretical calculation, we found that BPZ experienced Diels-Alder polymerization between the 
 stacked aromatic rings, and formed extended boat-GANR structures with exceptional long-range order. The unreacted -C=N- groups bridge the two ends of the boat, and ready for further functionalization. The GANR has a bandgap of 2.25 eV, with booming photoelectric response (
stacked aromatic rings, and formed extended boat-GANR structures with exceptional long-range order. The unreacted -C=N- groups bridge the two ends of the boat, and ready for further functionalization. The GANR has a bandgap of 2.25 eV, with booming photoelectric response (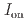 /
/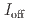 =18.8). Our work highlights that the high-pressure topochemical polymerization is a promising method for the precise synthesis of graphane with specific structure and desired properties.
=18.8). Our work highlights that the high-pressure topochemical polymerization is a promising method for the precise synthesis of graphane with specific structure and desired properties.
 neutron imaging and diffraction analysis revealing spatial lithiation phase evolution in an ultra-thick graphite electrode
neutron imaging and diffraction analysis revealing spatial lithiation phase evolution in an ultra-thick graphite electrodeStrobl, M.*; Baur, M. E.*; Samothrakitis, S.*; Molamud, F.*; Zhang, X.*; Tung, P. K. M.*; Schmidt, S.*; Woracek, R.*; Lee, J.*; Kiyanagi, Ryoji; et al.
Advanced Energy Materials, p.2405238_1 - 2405238_9, 2025/01
Times Cited Count:0
Jin, H.*; Choi, E. S.*; Wu, H.-C.*; Curro, N. J.*; Nawa, Kazuhiro*; Sato, Taku*; Kiyanagi, Ryoji; Ohara, Takashi; Klavins, P.*; Taufour, V.*
Physical Review B, 111(3), p.035103_1 - 035103_7, 2025/01
Times Cited Count:0 Percentile:0.00(Materials Science, Multidisciplinary)Hattori, Takanori; Komatsu, Kazuki*
Nihon Genshiryoku Gakkai-Shi ATOMO , 66(12), p.618 - 622, 2024/12
, 66(12), p.618 - 622, 2024/12
The high-pressure neutron diffractometer PLANET is the first beamline dedicated to high-pressure neutron experiments in Japan. It was constructed at the Materials and Life Science Experimental Facility (MLF) in the Japan Proton Accelerator Research Complex (J-PARC) located at Tokai-mura in Ibaraki Prefecture. Energy-dispersive data measurement using pulsed neutrons, state-of-the-art optical instruments, and a high-pressure device enable us to analyze the structure of crystals, liquids, and glasses over a wide range of pressure and temperature with unprecedented accuracy. In this paper, we will show how this has been achieved and introduce the recently published results on the symmetrization of hydrogen bonds in ice.
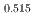 Gd
Gd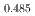 Mn
Mn O
O
Ishii, Yuta*; Sakakura, Terutoshi*; Ishikawa, Yoshihisa*; Kiyanagi, Ryoji; Lustikova, J.*; Aoyama, Takuya*; Ogushi, Kenya*; Wakabayashi, Yusuke*; Kimura, Hiroyuki*; Noda, Yukio*
Physical Review B, 110(18), p.184404_1 - 184404_7, 2024/11
Times Cited Count:0 Percentile:0.00(Materials Science, Multidisciplinary)Qin, T. Y.*; Hu, F. F.*; Xu, P. G.; Zhang, H.*; Zhou, L.*; Ao, N.*; Su, Y. H.; Shobu, Takahisa; Wu, S. C.*
International Journal of Fatigue, 185, p.108336_1 - 108336_13, 2024/08
Times Cited Count:9 Percentile:93.27(Engineering, Mechanical)He, X.*; Kagi, Hiroyuki*; Komatsu, Kazuki*; Iizuka, Riko*; Okajima, Hajime*; Hattori, Takanori; Sano, Asami; Machida, Shinichi*; Abe, Jun*; Goto, Hirotada*; et al.
Journal of Molecular Structure, 1310, p.138271_1 - 138271_8, 2024/08
Times Cited Count:0 Percentile:0.00(Chemistry, Physical)High-pressure responses of the O-D F hydrogen bonds in deuterated magnesium hydroxyfluoride were investigated using neutron powder diffraction and Raman spectroscopy. The Rietveld analysis at ambient conditions revealed a chemical formula of Mg(OD)
F hydrogen bonds in deuterated magnesium hydroxyfluoride were investigated using neutron powder diffraction and Raman spectroscopy. The Rietveld analysis at ambient conditions revealed a chemical formula of Mg(OD)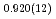 F
F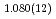 and hydroxyl group/fluorine disorder (OD/F disorder) in the crystal structure, which gave rise to two hydrogen-bonding configurations. The Rietveld analysis showed the hydrogen-bonding geometries remains up to 9.8 GPa, indicating no pressure-induced strengthening of hydrogen bonds. The Raman spectra at ambient conditions showed three hydroxyl stretching bands at 2613, 2694, and 2718 cm
and hydroxyl group/fluorine disorder (OD/F disorder) in the crystal structure, which gave rise to two hydrogen-bonding configurations. The Rietveld analysis showed the hydrogen-bonding geometries remains up to 9.8 GPa, indicating no pressure-induced strengthening of hydrogen bonds. The Raman spectra at ambient conditions showed three hydroxyl stretching bands at 2613, 2694, and 2718 cm . The high frequencies of the O-D stretching modes indicated that the hydroxyls should be involved in weak or none hydrogen-bonding interactions. Up to 20.2 GPa, the mode initially centered at 2694 cm
. The high frequencies of the O-D stretching modes indicated that the hydroxyls should be involved in weak or none hydrogen-bonding interactions. Up to 20.2 GPa, the mode initially centered at 2694 cm displayed a pressure-induced blue shift, revealing no strengthening of hydrogen bonds under compression. We discuss the existence of hydrogen bonds and the causes of the blue-shifting hydroxyls at ambient and at high pressures.
displayed a pressure-induced blue shift, revealing no strengthening of hydrogen bonds under compression. We discuss the existence of hydrogen bonds and the causes of the blue-shifting hydroxyls at ambient and at high pressures.
Fang, W.*; Liu, C.*; Zhang, J.*; Xu, P. G.; Peng, T.*; Liu, B.*; Morooka, Satoshi; Yin, F.*
Scripta Materialia, 249, p.116046_1 - 116046_6, 2024/08
Times Cited Count:2 Percentile:57.76(Nanoscience & Nanotechnology)Zhou, L.*; Zhang, H.*; Qin, T. Y.*; Hu, F. F.*; Xu, P. G.; Ao, N.*; Su, Y. H.; He, L. H.*; Li, X. H.*; Zhang, J. R.*; et al.
Metallurgical and Materials Transactions A, 55(7), p.2175 - 2185, 2024/07
Times Cited Count:3 Percentile:70.74(Materials Science, Multidisciplinary) O ice observed by neutron diffraction
O ice observed by neutron diffractionKomatsu, Kazuki*; Hattori, Takanori; Klotz, S.*; Machida, Shinichi*; Yamashita, Keishiro*; Ito, Hayate*; Kobayashi, Hiroki*; Irifune, Tetsuo*; Shimmei, Toru*; Sano, Asami; et al.
Nature Communications (Internet), 15, p.5100_1 - 5100_7, 2024/06
Times Cited Count:5 Percentile:67.98(Multidisciplinary Sciences)Hydrogen bond symmetrisation is the phenomenon where a hydrogen atom is located at the centre of a hydrogen bond. Theoretical studies predict that hydrogen bonds in ice VII eventually undergo symmetrisation upon increasing pressure, involving nuclear quantum effect with significant isotope effect and drastic changes in the elastic properties through several intermediate states with varying hydrogen distribution. Despite numerous experimental studies conducted, the location of hydrogen and hence the transition pressures reported up to date remain inconsistent. Here we report the atomic distribution of deuterium in D O ice using neutron diffraction above 100 GPa and observe for the first time the transition from a bimodal to a unimodal distribution of deuterium at around 80 GPa. At the transition pressure, a significant narrowing of the peak widths of 110 was also observed, attributed to the structural relaxation by the change of elastic properties.
O ice using neutron diffraction above 100 GPa and observe for the first time the transition from a bimodal to a unimodal distribution of deuterium at around 80 GPa. At the transition pressure, a significant narrowing of the peak widths of 110 was also observed, attributed to the structural relaxation by the change of elastic properties.
Dronskowski, R.*; Br ckel, T.*; Kohlmann, H.*; Avdeev, M.*; Houben, A.*; Meven, M.*; Hofmann, M.*; Kamiyama, Takashi*; Zobel, M.*; Schweika, W.*; et al.
ckel, T.*; Kohlmann, H.*; Avdeev, M.*; Houben, A.*; Meven, M.*; Hofmann, M.*; Kamiyama, Takashi*; Zobel, M.*; Schweika, W.*; et al.
Zeitschrift f r Kristallographie; Crystalline Materials, 239(5-6), p.139 - 166, 2024/06
r Kristallographie; Crystalline Materials, 239(5-6), p.139 - 166, 2024/06
Because of the neutron's special properties, neutron diffraction may be considered one of the most powerful techniques for structure determination of crystalline and related matter. Neutrons can be released from nuclear fission, from spallation processes, and also from low-energy nuclear reactions, and they can then be used in powder, time-of-flight, texture, single crystal, and other techniques, all of which are perfectly suited to clarify crystal and magnetic structures. With high neutron flux and sufficient brilliance, neutron diffraction also excels for diffuse scattering, for in situ and operando studies as well as for high-pressure experiments of today's materials. In this primer, we summarize the current state of neutron diffraction (and how it came to be), but also look at recent advances and new ideas, e.g., the design of new instruments, and what follows from that.
Mori, Yuichiro*; Kagi, Hiroyuki*; Aoki, Katsutoshi*; Takano, Masahiro*; Kakizawa, Sho*; Sano, Asami; Funakoshi, Kenichi*
Earth and Planetary Science Letters, 634, p.118673_1 - 118673_8, 2024/05
Times Cited Count:1 Percentile:41.61(Geochemistry & Geophysics)To investigate silicon effects on the hydrogen-induced volume expansion of iron, neutron diffraction and X-ray diffraction experiments were conducted to examine hcp-Fe Si
Si under high pressures and high temperatures. Neutron diffraction experiments were performed on the deuterated hcp-Fe
under high pressures and high temperatures. Neutron diffraction experiments were performed on the deuterated hcp-Fe Si
Si at 13.5 GPa and 900 K, and at 12.1 GPa and 300 K. By combining the P-V-T equation of state of hcp-Fe
at 13.5 GPa and 900 K, and at 12.1 GPa and 300 K. By combining the P-V-T equation of state of hcp-Fe Si
Si , present results indicate that the hydrogen-induced volume expansion of hcp-Fe
, present results indicate that the hydrogen-induced volume expansion of hcp-Fe Si
Si is 10% greater than that of pure hcp iron. Using the obtained values, we estimated the hydrogen content that would reproduce the density deficit in the inner core, which was 50% less than that without the effect of silicon. Possible hydrogen content,
is 10% greater than that of pure hcp iron. Using the obtained values, we estimated the hydrogen content that would reproduce the density deficit in the inner core, which was 50% less than that without the effect of silicon. Possible hydrogen content, 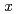 , in the inner core and the outer core was calculated to be 0.07 and 0.12-0.15, respectively, when reproducing the density deficit of the inner core with hcp-Fe
, in the inner core and the outer core was calculated to be 0.07 and 0.12-0.15, respectively, when reproducing the density deficit of the inner core with hcp-Fe Si
Si Hx.
Hx.
Liss, K.-D.*; Han, J.-K.*; Blankenburg, M.*; Lienert, U.*; Harjo, S.; Kawasaki, Takuro; Xu, P. G.; Yukutake, Eitaro*; Kawasaki, M.*
Journal of Materials Science, 59(14), p.5831 - 5853, 2024/04
Times Cited Count:4 Percentile:73.30(Materials Science, Multidisciplinary)Hattori, Takanori; Suzuki, Koji*; Miyo, Tatsuya*; Ito, Takayoshi*; Machida, Shinichi*
Nuclear Instruments and Methods in Physics Research A, 1059, p.168956_1 - 168956_9, 2024/02
Times Cited Count:2 Percentile:43.92(Instruments & Instrumentation)Radial collimators (RC) with a 0.5 mm gauge size (GS) were specially designed for high-pressure neutron diffraction experiments and their performance and efficacy were investigated. The RCs with nominal GS of 0.75 mm, 1.5 mm, and 3.0 mm effectively exhibited GS of 0.50 mm, 1.07 mm, and 2.78 mm, respectively. The transmissions of all three RCs were almost equivalent. The assessment using a P-E press and a DAC revealed that the anvil scattering was considerably minimized and the sample-to-anvil signal ratio reached values of 0.5 and 2.0 for the PE press and DAC, respectively, when using the 0.5 mm-GS RCs. These results indicate that the 0.5mm-GS RCs have been fabricated as intended and exhibit efficacy for the high-pressure-neutron diffraction experiments, specifically those exceeding 30 GPa. Among those ever manufactured for neutron scattering experiments, the RCs display the smallest GS.
Xu, P. G.; Zhang, S.-Y.*; Harjo, S.; Vogel, S. C.*; Tomota, Yo*
Quantum Beam Science (Internet), 8(1), p.7_1 - 7_13, 2024/01
Yamashita, Keishiro*; Nakayama, Kazuya*; Komatsu, Kazuki*; Ohara, Takashi; Munakata, Koji*; Hattori, Takanori; Sano, Asami; Kagi, Hiroyuki*
Acta Crystallographica Section B; Structural Science, Crystal Engineering and Materials (Internet), 79(5), p.414 - 426, 2023/10
Times Cited Count:2 Percentile:41.77(Chemistry, Multidisciplinary)The structure of a recently-found hyperhydrated form of sodium chloride, NaCl 13H(D)
13H(D) O, has been determined by
O, has been determined by  single-crystal neutron diffraction at 1.7 GPa and 298 K. It has large hydrogen-bond networks and some water molecules have distorted bonding features such as bifurcated hydrogen bonds and five-coordinated water molecules. The hydrogen-bond network has similarities to ice VI in terms of network topology and disordered hydrogen bonds. Assuming the equivalence of network components connected by pseudo symmetries, the overall network structure of this hydrate can be expressed by breaking it down into smaller structural units which correspond to the ice VI network structure. This hydrogen-bond network contains orientational disorder of water molecules in contrast to the known salt hydrates. Here, we present an example for further insights into a hydrogen-bond network containing ionic species.
single-crystal neutron diffraction at 1.7 GPa and 298 K. It has large hydrogen-bond networks and some water molecules have distorted bonding features such as bifurcated hydrogen bonds and five-coordinated water molecules. The hydrogen-bond network has similarities to ice VI in terms of network topology and disordered hydrogen bonds. Assuming the equivalence of network components connected by pseudo symmetries, the overall network structure of this hydrate can be expressed by breaking it down into smaller structural units which correspond to the ice VI network structure. This hydrogen-bond network contains orientational disorder of water molecules in contrast to the known salt hydrates. Here, we present an example for further insights into a hydrogen-bond network containing ionic species.
Abeykoon, S.*; Howard, C.*; Dominijanni, S.*; Eberhard, L.*; Kurnosov, A.*; Frost, D. J.*; Boffa Ballaran, T.*; Terasaki, Hidenori*; Sakamaki, Tatsuya*; Suzuki, Akio*; et al.
Journal of Geophysical Research; Solid Earth, 128(9), p.e2023JB026710_1 - e2023JB026710_17, 2023/09
Times Cited Count:2 Percentile:30.17(Geochemistry & Geophysics)Small amounts of iron sulphide minerals are found in most rocks from the Earth's mantle and as inclusions trapped in natural diamonds. Hydrogen may dissolve into iron sulphide minerals under high pressures and temperature, but is most likely lost once pressure and temperature are removed. In this study, we determined deuterium contents in iron sulphide, held under high pressure and temperature conditions, using neutron diffraction measurements with 6-ram multi-anvil press at PLANET, J-PARC. Deuterium contents in iron sulphide were measured at high-P, up to 11.4 GPa and high-T to 1300 K in in situ neutron diffraction experiments. The total deuterium content increases with both P and T. The results are used to estimate hydrogen contents of iron sulphide minerals in the deep continental lithospheric mantle, which are found to be in the range 1700-2700 ppm. This corresponds to approximately 2-3 ppm of hydrogen in the bulk mantle.
 Mg and long-period stacking ordered phases in a Mg-Zn-Y alloy by hot-extrusion with low extrusion ratio
Mg and long-period stacking ordered phases in a Mg-Zn-Y alloy by hot-extrusion with low extrusion ratioHarjo, S.; Gong, W.; Aizawa, Kazuya; Kawasaki, Takuro; Yamasaki, Michiaki*
Acta Materialia, 255, p.119029_1 - 119029_12, 2023/08
Times Cited Count:31 Percentile:96.82(Materials Science, Multidisciplinary)