Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tsutsui, Nao; Ban, Yasutoshi; Suzuki, Hideya*; Nakase, Masahiko*; Ito, Sayumi*; Inaba, Yusuke*; Matsumura, Tatsuro; Takeshita, Kenji*
Analytical Sciences, 36(2), p.241 - 246, 2020/02
Times Cited Count:30 Percentile:83.49(Chemistry, Analytical)To investigate the effective separation of actinides (Ans) from lanthanides (Lns), single-stage batch extraction experiments were performed with a novel extractant, tetradodecyl-1,10-phenanthroline-2,9-diamide (TDdPTDA) with various diluents such as 3-nitrobenzotrifluoride (F-3), nitrobenzene, and  -dodecane for Am, Cm, and Lns. The extraction kinetics with TDdPTDA was rapid enough to perform the actual extraction flow sheet. The slopes of the distribution ratio versus TDdPTDA concentration and the distribution ratio versus nitric acid concentration were similar for F-3 and nitrobenzene systems but different from
-dodecane for Am, Cm, and Lns. The extraction kinetics with TDdPTDA was rapid enough to perform the actual extraction flow sheet. The slopes of the distribution ratio versus TDdPTDA concentration and the distribution ratio versus nitric acid concentration were similar for F-3 and nitrobenzene systems but different from  -dodecane system. These differences were attributed to the characteristics of the diluents. This study reveals high distribution ratios of Am (
-dodecane system. These differences were attributed to the characteristics of the diluents. This study reveals high distribution ratios of Am (
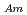 ) and Cm (
) and Cm (
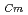 ) for TDdPTDA, with the high separation factors (
) for TDdPTDA, with the high separation factors ( s) of Am from Lns enough for their separation.
s) of Am from Lns enough for their separation.
 ,
, -di(2-ethylhexyl)octanamide toward uranium from nitric acid
-di(2-ethylhexyl)octanamide toward uranium from nitric acidTsutsui, Nao; Ban, Yasutoshi; Sagawa, Hiroshi; Ishii, Sho; Matsumura, Tatsuro
Proceedings of 5th International Conference on Asian Nuclear Prospects 2016 (ANUP 2016) (CD-ROM), 2 Pages, 2016/10
Extraction properties of  ,
, -di(2-ethylhexyl)octanamide (DEHOA) toward uranium from nitric acid were studied using single-stage batch method. The results revealed that DEHOA effectively extracted uranium when the nitrate ion concentration was 2.0-3.0 mol/dm
-di(2-ethylhexyl)octanamide (DEHOA) toward uranium from nitric acid were studied using single-stage batch method. The results revealed that DEHOA effectively extracted uranium when the nitrate ion concentration was 2.0-3.0 mol/dm .
.
 /supercritical CO
/supercritical CO +TBP system
+TBP systemMeguro, Yoshihiro; Iso, Shuichi; Yoshida, Zenko
Proceedings of International Solvent Extraction Conference 2002 (CD-ROM), p.1131 - 1136, 2002/00
no abstracts in English
Tachimori, Shoichi
JAERI 1337, 250 Pages, 1996/05
no abstracts in English
;
PNC TN8420 95-002, 140 Pages, 1995/01
In the field of uranium ore refining and spent fuel reprocessing, a lot of extractants capable of recovering actinides have been innovated, and some of which have been developed in actual industrial scale processes. Extractability against trivalent actinides on each extractants were evaluated in this report. And as for the compounds which showed excellent extractability for Americium, their extraction characteristics for trivalent actinides, such as extraction behavior, mechanism, ligand structure and conformation of complexes, were evaluated, and filed as data-base. Based on these works, some of extractant were newly identified as candidate to apply Americium recovering. The most appropriate compounds for trivalent actinide separation can be generally found in the neutral bidentate organic compound groups: Especially, some derivatives belongs to Diphosphineoxide, Carbamoyl-methylphosphine oxide and Propandiamide were superior to give sufficiently excellent property even in higher nitric acid(1 3M) solutions. Innovative research towards developping new extractants and extraction system seems to be directed to ; (1)investigate new types of extractant sustained by a different kind of complexing mechanism, (2)evaluate synergistic effect of adducts, diluents even including conventional extractants.
3M) solutions. Innovative research towards developping new extractants and extraction system seems to be directed to ; (1)investigate new types of extractant sustained by a different kind of complexing mechanism, (2)evaluate synergistic effect of adducts, diluents even including conventional extractants.
 Co,
Co,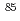 Sr and
Sr and 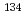 Cs in loess medium
Cs in loess mediumTanaka, Tadao; Ogawa, Hiromichi; Z.Li*; H.Wang*;
JAERI-M 94-077, 17 Pages, 1994/06
no abstracts in English
Tachimori, Shoichi
JAERI 1331, 177 Pages, 1994/03
no abstracts in English
 -TBP-n-dodecane system
-TBP-n-dodecane system; Suzuki, Shinichi; Abe, Hitoshi; Tachimori, Shoichi
JAERI-M 93-014, 40 Pages, 1993/02
no abstracts in English
 Co,
Co, Sr and
Sr and  Cs in a porous tuff; Mechanisms and kinetics
Cs in a porous tuff; Mechanisms and kineticsC.K.Park*; S.I.Woo*; Tanaka, Tadao; Kamiyama, Hideo
Journal of Nuclear Science and Technology, 29(12), p.1184 - 1193, 1992/12
no abstracts in English
 system, II; Characteristics of the DHDECMP/XAD-4 resin on separation of trivalent actinide elements
system, II; Characteristics of the DHDECMP/XAD-4 resin on separation of trivalent actinide elementsKimura, Takaumi; Akatsu, Jokun
Journal of Radioanalytical and Nuclear Chemistry, 149(1), p.25 - 34, 1991/00
Times Cited Count:10 Percentile:71.62(Chemistry, Analytical)no abstracts in English
 system, II; Characteristics of the TBP/XAD-4 resin on separation of actinide elements
system, II; Characteristics of the TBP/XAD-4 resin on separation of actinide elementsKimura, Takaumi
Journal of Radioanalytical and Nuclear Chemistry, 141(2), p.307 - 316, 1990/00
Times Cited Count:12 Percentile:73.82(Chemistry, Analytical)no abstracts in English
;
Analytica Chimica Acta, 93, p.317 - 322, 1976/00
Times Cited Count:1no abstracts in English
; ;
JAERI-M 6284, 33 Pages, 1975/10
no abstracts in English
Ono, Shimpei; Ichige, Yoshiaki; Shibata, Atsuhiro; Sato, Takehiko; Takeuchi, Masayuki
no journal, ,
no abstracts in English
 ,
, -dialkylamides-nitric acid systems by turbidity measurement, 2; The Correlation between turbidity and the distribution ratio of uranium
-dialkylamides-nitric acid systems by turbidity measurement, 2; The Correlation between turbidity and the distribution ratio of uraniumTsutsui, Nao; Ban, Yasutoshi; Hakamatsuka, Yasuyuki; Urabe, Shunichi; Matsumura, Tatsuro
no journal, ,
The turbidity of the agitated solutions of  ,
, -di(2-ethylhexyl)-butanamide (DEHBA) in dodecane and uranyl nitrate in 3 M HNO
-di(2-ethylhexyl)-butanamide (DEHBA) in dodecane and uranyl nitrate in 3 M HNO was measured for 30 min. The variation of the distribution ratio of uranium after stopping the agitation was also measured, and the correlation between the turbidity and the distribution ratio was studied. The turbidity and the distribution ratio of uranium at 3 min after stopping the agitation was more than 1000 FTU and 1.7, respectively, when the initial uranium concentration was 700 mM. The turbidity at 6 min decreased to 363 FTU, while the distribution rate increased to 2.2. Although the turbidity after 6 min decreased gradually, the distribution ratio barely changed. The correlation between the turbidity and the distribution ratio is discussed.
was measured for 30 min. The variation of the distribution ratio of uranium after stopping the agitation was also measured, and the correlation between the turbidity and the distribution ratio was studied. The turbidity and the distribution ratio of uranium at 3 min after stopping the agitation was more than 1000 FTU and 1.7, respectively, when the initial uranium concentration was 700 mM. The turbidity at 6 min decreased to 363 FTU, while the distribution rate increased to 2.2. Although the turbidity after 6 min decreased gradually, the distribution ratio barely changed. The correlation between the turbidity and the distribution ratio is discussed.