Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kusaka, Ryoji
Bunko Kenkyu, 72(4), p.155 - 162, 2023/08
Vibrational sum frequency generation (VSFG) spectroscopy is an optical second-order nonlinear vibrational spectroscopy using ultrashort pulse lasers. Because VSFG spectroscopy is a unique and powerful tool for studying molecular structures of interfaces, it has been widely used in many research fields. However, there still undoubtedly remains some VSFG research areas that have not studied well, partly because VSFG measurements are not so easily performed in comparison with relatively general spectroscopy methods. This review presented recent applications of VSFG spectroscopy to two research topics: (1) chemical reactions on water surfaces, and (2) actinide chemistry.
Kusaka, Ryoji; Watanabe, Masayuki
Journal of Physical Chemistry Letters (Internet), 13(30), p.7065 - 7071, 2022/08
Times Cited Count:10 Percentile:71.30(Chemistry, Physical)Kusaka, Ryoji; Watanabe, Masayuki
Journal of Physical Chemistry B, 125(24), p.6727 - 6731, 2021/06
Times Cited Count:13 Percentile:46.94(Chemistry, Physical)Kusaka, Ryoji
Journal of Nuclear and Radiochemical Sciences (Internet), 20, p.28 - 31, 2020/06
Kusaka, Ryoji
Hosha Kagaku, (41), p.31 - 33, 2020/03
This commentary article introduced researches involved in encouragement award 2019 of the Japan Society of Nuclear and Radiochemical Sciences. Vibrational sum frequency generation (VSFG) spectroscopy and interfacial studies of solvent extraction of lanthanides and actinides using VSFG spectroscopy were described.
Kusaka, Ryoji; Watanabe, Masayuki
Physical Chemistry Chemical Physics, 20(47), p.29588 - 29590, 2018/12
Times Cited Count:20 Percentile:67.81(Chemistry, Physical)Mechanistic understanding of solvent extraction of uranyl ions (UO
 ) by tributyl phosphate (TBP) will help improve the technology for the treatment and disposal of spent nuclear fuels. So far, it has been believed that uranyl ions in the aqueous phase are adsorbed to a TBP-enriched organic/aqueous interface, form complexes with TBP at the interface, and are extracted into the organic phase. Here we show that uranyl-TBP complex formation does not take place at the interface using vibrational sum frequency generation (VSFG) spectroscopy and propose an alternative extraction mechanism that uranyl nitrate, UO
) by tributyl phosphate (TBP) will help improve the technology for the treatment and disposal of spent nuclear fuels. So far, it has been believed that uranyl ions in the aqueous phase are adsorbed to a TBP-enriched organic/aqueous interface, form complexes with TBP at the interface, and are extracted into the organic phase. Here we show that uranyl-TBP complex formation does not take place at the interface using vibrational sum frequency generation (VSFG) spectroscopy and propose an alternative extraction mechanism that uranyl nitrate, UO (NO
(NO )
) , passes through the interface and forms the uranyl-TBP complex, UO
, passes through the interface and forms the uranyl-TBP complex, UO (NO
(NO )
) (TBP)
(TBP) , in the organic phase.
, in the organic phase.
Kusaka, Ryoji; Watanabe, Masayuki
Physical Chemistry Chemical Physics, 20(4), p.2809 - 2813, 2018/01
Times Cited Count:17 Percentile:61.76(Chemistry, Physical)Solvent extraction plays an integral part in the separation and purification of metals. Because extractants generally used as complexing agents for metal extractions, such as di-(2-ethylhexyl)phosphoric acid (HDEHP) for lanthanide extractions, are amphiphilic, they come to the organic/water interface, and the interface plays a crucial role as the site of the formation of metal complexes and subsequent transfer reaction to an organic phase. Despite the importance of the interface for solvent extractions, however, molecular-level structure of the interface is unclear because of experimental difficulty. Here we studied structure of a trivalent europium (Eu ) complex with HDEHP formed at HDEHP monolayer/water interface by heterodyne-detected vibrational sum frequency generation (HD-VSFG) spectroscopy. The study on the HDEHP/water interface enables us to investigate the structure of the interfacial Eu
) complex with HDEHP formed at HDEHP monolayer/water interface by heterodyne-detected vibrational sum frequency generation (HD-VSFG) spectroscopy. The study on the HDEHP/water interface enables us to investigate the structure of the interfacial Eu complex by excluding the migration of Eu
complex by excluding the migration of Eu into an organic phase after the complex formation at the interface. The interface-selective vibrational Im
into an organic phase after the complex formation at the interface. The interface-selective vibrational Im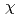
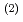 spectra observed by HD-VSFG of HDEHP/Eu(NO
spectra observed by HD-VSFG of HDEHP/Eu(NO )
) aqueous solution interface in the 2800-3500 cm
aqueous solution interface in the 2800-3500 cm region indicate that Eu
region indicate that Eu at the HDEHP/water interface is bonded by HDEHP from the air side and by water molecules from the water side. To the best of our knowledge, such metal complex structures have not been identified in the organic or water solutions.
at the HDEHP/water interface is bonded by HDEHP from the air side and by water molecules from the water side. To the best of our knowledge, such metal complex structures have not been identified in the organic or water solutions.
Kusaka, Ryoji; Schnaars, K.; Watanabe, Masayuki
no journal, ,
To unveil the extraction mechanism of metal elements from the aqueous phase to the organic phase, we have recently suggested that it is useful to clarify the structure and the property of the metal complexes formed on the surface of aqueous solutions dissolving metal ion and extractant (ligand). In the present study, in order to reveal the mechanism of the solvent extraction of lanthanides by di-(2-ethylhexyl)phosphoric acid (HDEHP), we measured vibrational spectra of lanthanides-HDEHP complexes on the aqueous solutions in the P=O stretch region with vibrational sum frequency generation (VSFG) spectroscopy, and investigated the structure and properties of the complexes on the surface of the aqueous solutions.
Kusaka, Ryoji
no journal, ,
no abstracts in English
Kusaka, Ryoji
no journal, ,
no abstracts in English
Nakai, Noa; Urashima, Shuhei; Watanabe, Masayuki; Kusaka, Ryoji
no journal, ,
no abstracts in English
Kusaka, Ryoji; Watanabe, Masayuki
no journal, ,
In general solvent extraction, an aqueous phase where metal ions are dissolved and an organic phase where an extractant is dissolved are brought into contact with each other, and the metal is extracted from the aqueous phase to the organic phase as a metal complex. Since the extractant used is poorly soluble in water and surface active, it is considered that complex formation between metal and extractant takes place at the interface and then the metal is extracted into the organic phase. However, due to experimental difficulties, the phase transfer mechanism occurring at the interface is unknown. One of the reasons for the difficulties to observe the metal complex at the interface is that the metal complex moves to the organic phase after the complex formation at the interface. In order to observe the metal complex at the interface, we removed the organic phase so that we can trap the metal complex at the extractant/water interface (the surface of aqueous solution). The structure of the metal complex at the interface is investigated using sum frequency generation (VSFG) spectroscopy to elucidate the solvent extraction mechanism.
Kusaka, Ryoji; Watanabe, Masayuki
no journal, ,
no abstracts in English
Kusaka, Ryoji; Watanabe, Masayuki
no journal, ,
We study extraction mechanism of metal elements from the aqueous phase to the organic phase to improve separation technology by solvent extraction. In the present presentation, we comprehensively show recent results about transfer mechanism of metal elements, studied by Vibrational Sum Frequency Generation (VSFG) spectroscopy. Based on the results, we propose a novel way for improvement of solvent extraction technology.
Kusaka, Ryoji; Watanabe, Masayuki
no journal, ,
Liquid-liquid solvent extraction of uranyl using tributyl phosphate (TBP) is a significantly important technique in treatments of spent nuclear fuels. In the solvent extraction, TBP comes to an organic/aqueous interface and forms an organic/TBP monolayer/aqueous interface because of the surface activity of TBP. At the interface, it is believed that the complex formation between uranyl and TBP takes place, and subsequently the uranyl-TBP complex transfers to an organic phase. However, the structure of the uranyl-TBP complex formed at the interface is controversial. This is partly because of no experimental studies on molecular-level structure at interfaces pertaining to the solvent extraction of uranyl. Recently, we proposed that an extractant/aqueous interface, formed on the surface of an aqueous solution, is a good model of an organic/extractant/aqueous interface, to investigate the structure of interfacial metal complexes using vibrational sum frequency generation (VSFG) spectroscopy. In the present study, we investigated the structure of theuranyl-TBP complex at the TBP/uranyl aqueous solution interface by VSFG spectroscopy. Surprisingly, interface-selective vibrational spectra obtained by VSFG suggests that uranyl does not form a complex with TBP at the TBP/uranyl aqueous solution interface. This result proposes that we should reconsider the mechanism of the solvent extraction of uranyl by TBP.
Kusaka, Ryoji; Watanabe, Masayuki
no journal, ,
The solvent extraction utilizing the complex formation between uranyl and tributyl phosphate (TBP) is important as a method for treating spent nuclear fuel. Since TBP is surface active, it is believed that the uranyl-TBP complex is formed at the organic/TBP/water interface, and the complex is extracted into the organic phase. However, the structure of the uranyl-TBP complex formed at the interface is controversial. In this study, the structure of the uranyl-TBP complex was investigated by heterodyne-detected vibrational sum frequency generation spectroscopy at the surface of an aqueous solution of uranyl and TBP (TBP molecular layer/aqueous solution interface). Surprisingly, it is found that uranyl-TBP complex does not form at the interface.
Kusaka, Ryoji
no journal, ,
This presentation is the award-winning lecture of the Japan Society of Nuclear and Radiochemical Sciences 2019 Encouragement Award. It will be presented that the research achievements on the interface chemistry of lanthanide and actinide, which have been obtained by using vibrational sum frequency generation spectroscopy in the 4th research building of JAEA.
Kusaka, Ryoji
no journal, ,
The interfaces of oil and aqueous phases in solvent extractions are the gates for metals to transfer between the two phases. It would be that something special occurs at the interfaces because extractants, which are ligand reagents that facilitate metal transfer into the oil phase by surrounding and stabilizing metals in the oil phase, are surfactants tending to form layers at the interfaces. We have investigated the interfaces of lanthanide and actinide solvent extractions using vibrational sum frequency generation spectroscopy both in homodyne and heterodyne detection methodologies. Vibrational signals of coordinating chemical groups of extractants and interfacial water OH stretch revealed molecular structures of lanthanide and actinide complexes with extractant forming at the interfaces, providing some aspects of phase transfer mechanism of lanthanide and actinide between the oil and aqueous phases.