Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tochiyama, Osamu*
JNC TJ8400 2000-044, 53 Pages, 2000/02
To estimate the polyelectrolyte effect and the effect of the heterogeneous composition of humic acids, the complex formation constants of Eu(III) and Ca(II) with Aldrich humic acid and polyacrylic acid were obtained, for Eu(10 to 10
to 10 M) by solvent extraction with TTA and TBP in xylene, for Ca (10
M) by solvent extraction with TTA and TBP in xylene, for Ca (10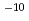 M) with TTA and TOPO in cyclohexane and for Ca(10
M) with TTA and TOPO in cyclohexane and for Ca(10 M) by using ion-selective electrode. By defining the apparent formation as
M) by using ion-selective electrode. By defining the apparent formation as  = [MR
= [MR ]/([M][R]), where [R] denotes the concentration of dissociated functional group, [M] and [MR
]/([M][R]), where [R] denotes the concentration of dissociated functional group, [M] and [MR ] denote the concentration of free and bound metal ion and pcH is defined as-log[H], the values of log
] denote the concentration of free and bound metal ion and pcH is defined as-log[H], the values of log have been obtained at pcH 4.8 - 5.5 in 0.1 - 1.0M NaClO
have been obtained at pcH 4.8 - 5.5 in 0.1 - 1.0M NaClO and NaCl. Log
and NaCl. Log of Eu-humate varied from 5.0 to 9.3 and that of Ca-humate from 2.0 to 3.4..For both humate and polyacrylate, log
of Eu-humate varied from 5.0 to 9.3 and that of Ca-humate from 2.0 to 3.4..For both humate and polyacrylate, log increased with pcH or with the degree of dissociation. The increase in the ionic strength O.1 to 1.0 M decreased the log
increased with pcH or with the degree of dissociation. The increase in the ionic strength O.1 to 1.0 M decreased the log , the decrease in log
, the decrease in log of Eu(III)-humate is 1.6, that of Eu(III), polyacrylate 0.7, that of Ca(II)-humate 1.9 and that of Ca(II)-polyacrylate 1.2. While the increase in the metal ion produced no effect on log
of Eu(III)-humate is 1.6, that of Eu(III), polyacrylate 0.7, that of Ca(II)-humate 1.9 and that of Ca(II)-polyacrylate 1.2. While the increase in the metal ion produced no effect on log of polyacrylate, log
of polyacrylate, log of humate decreased. Depending on the concentration of Eu(III), the coexistence of Ca(II) reduced log
of humate decreased. Depending on the concentration of Eu(III), the coexistence of Ca(II) reduced log  of humate by 0 to 0.8. The dependence of log
of humate by 0 to 0.8. The dependence of log of humate on the metal ion concentration suggests the coexistence of strong and weak binding sites in the hmnic acid.
of humate on the metal ion concentration suggests the coexistence of strong and weak binding sites in the hmnic acid.
Sasamoto, Hiroshi; Yui, Mikazu; Savage, D.*; Bille, B.*
JNC TN8400 99-025, 32 Pages, 1999/06
Groundwater data used for modelling site or repository evolution need to be assessed for their quality and whether they are "fit for purpose", prior to utilization. This report discuss factors and issues which impinge upon the quality of such data. It is recommended that geochemical modelleres : (1)are aware of how groundwater samples were collected, whether during drilling, during hydraulic testing, or thereafter, by in-situ measurement, pumped from boreholes, or by pressurised sampler ; (2)are aware of what procedures (if any) were used to "correct" samples for drill fluid contamination and what errors were associated with those methods ; (3)are aware of whether samples were subject to de-pressurisation during sampling, and whether geochemical modelling techniques were applied to correct the compositions of samples for that process ; (4)request different measures of redox activity (e.g., electrode measurements of Eh, concentrations of different redox-sensitive aqueous species) to be applied to key groundwater samples to investigate the extent of redox equilibrium ; (5)are aware of how groundwater samples were filtered and preserved for off-site analysis ; (6)ensure that adequate methods of groundwater filtration ( 0.1
0.1 m) and chemical analysis are applied to ensure accurate and reproducible analyses for dissolved aluminum at low levels of concentration (generally less than 0.2 mg/L) ; (7)are aware of elemental errors and detection limits in chemical analysis of groundwater samples and assess the quality of groundwater analyses via ion exchange balances and via a comparison of measured and calculated values for total dissolved solids contents. (8)ensure that detailed mineralogical analysis is carried out on rock samples from locations where key groundwater samples have been extracted.
m) and chemical analysis are applied to ensure accurate and reproducible analyses for dissolved aluminum at low levels of concentration (generally less than 0.2 mg/L) ; (7)are aware of elemental errors and detection limits in chemical analysis of groundwater samples and assess the quality of groundwater analyses via ion exchange balances and via a comparison of measured and calculated values for total dissolved solids contents. (8)ensure that detailed mineralogical analysis is carried out on rock samples from locations where key groundwater samples have been extracted.
Morishima, Hiroshige*; Koga, Taeko*
PNC TJ1630 97-001, 37 Pages, 1997/03
no abstracts in English
Ikeda, Yasuhiro*; Ishihara, Yoshinao*; Nakahara, Yutaka*; Sugisaki, Takayoshi*; Doi, Hideo*; Hamamoto, Hiroshi*; Mukai, Satoru*; Hachiya, Naoyuki*
PNC TJ1214 91-003, 529 Pages, 1991/05
None
Genshiryoku Kogyo, 17(8), p.32 - 36, 1972/00
no abstracts in English